Heart Failure
Key Points
- Heart failure (HF) is decreased cardiac output from ineffective pumping, often causing dyspnea, fatigue, edema, and congestion.
- Common causes include coronary artery disease, myocardial infarction, hypertension, valvular disease, and cardiomyopathy.
- Cardiac output depends on both heart rate and stroke volume, and stroke volume is determined by preload, afterload, and contractility.
- HFrEF is the most common HF phenotype and is commonly linked to CAD/MI injury and long-standing afterload stress from uncontrolled hypertension.
- EF-based phenotype triage uses common thresholds: HFrEF ⇐40%, HFmrEF 41-49% with symptoms, and HFpEF >=50% with symptoms.
- NYHA functional class (I-IV) guides expected baseline findings, progression risk, and care intensity.
- Acute decompensated heart failure (ADHF) presents with sudden worsening and requires rapid escalation.
- Peripartum cardiomyopathy is a pregnancy-associated cause of systolic heart failure that requires maternal-fetal safety adaptations.
- Core management combines medication therapy, sodium and fluid strategies, symptom tracking, and coordinated follow-up.
- Secondary prevention nutrition should pair sodium control with adequate protein intake to reduce hypoalbuminemia-related worsening edema risk.
- In HF, medication burden plus low diet variety can contribute to micronutrient deficits (for example calcium, folate, magnesium, zinc, and vitamins C, D, E, and K), so nutrition-surveillance and dietitian follow-up are safety-relevant.
- In infants and children, congenital heart defects are a major HF driver and often present as feeding intolerance, diaphoresis, growth delay, and congestion signs.
Pathophysiology
HF develops when myocardial pump function cannot meet tissue oxygenation and perfusion demands. Reduced forward flow and compensatory fluid-retention pathways produce fatigue, pulmonary congestion, and peripheral edema. As disease progresses, clients move along a continuum from asymptomatic risk to end-stage disease.
Cardiac output is the product of heart rate and stroke volume (CO = HR x SV), with typical resting adult output around 4-5 L/min. Stroke volume is shaped by preload (ventricular filling), afterload (resistance to ejection), and contractility (myocardial squeeze strength). HF develops when these relationships can no longer maintain adequate systemic perfusion.
Etiologies include ischemic injury from coronary-artery-disease and MI, pressure overload from chronic hypertension-assessment-and-management, structural valve dysfunction, and inherited or acquired cardiomyopathy. These mechanisms impair ventricular filling and/or ejection and reduce effective perfusion reserve.
Population risk patterns in HF include high burdens of hypertension, ischemic heart disease, atrial fibrillation, and diabetes, with higher observed occurrence in some Black and Hispanic populations. This supports early risk-trend screening during routine cardiovascular assessment.
In HFrEF pathways, reduced contractility lowers ejection performance and forward flow. CAD/MI can reduce myocardial oxygen delivery to myocytes, and prolonged uncontrolled hypertension can drive compensatory left-ventricular hypertrophy that later decompensates, producing systolic-failure patterns.
HFpEF pathways are driven by ventricular stiffness and impaired filling. In these patterns, both the amount of blood filling the ventricle and the amount ejected can decline proportionally, so measured EF may remain in the preserved range even when cardiac output is reduced.
In late pregnancy and early postpartum periods, peripartum-cardiomyopathy can present with new systolic dysfunction and signs that may overlap with expected pregnancy discomforts. This overlap can delay diagnosis unless dyspnea, orthopnea, pulmonary congestion, and hemodynamic trends are assessed carefully.
Compensatory stretch can briefly increase stroke volume (Frank-Starling response), but chronic pressure and volume overload eventually become maladaptive. Over time, ventricles may hypertrophy or dilate with reduced recoil and weaker contractility, driving persistent HF symptom burden.
In pediatric populations, congenital structural defects can produce early pressure or volume overload that progresses to pulmonary congestion, systemic venous congestion, and reduced cardiac-output reserve. Clinical decline may appear first during feeding and crying because energy demand rises quickly in infants.
Classification
- Left-sided HF: Predominant pulmonary congestion pattern (dyspnea, orthopnea, crackles, reduced ejection fraction trends).
- Right-sided HF: Predominant systemic venous congestion pattern (peripheral edema, ascites, hepatomegaly, JVD).
- HF by ejection fraction context: HFrEF is commonly ⇐40%; HFmrEF is commonly 41-49% with symptoms; HFpEF is commonly >=50% with symptoms.
- ACC/AHA/HFSA Stage A-D framework: Stage A = at risk without structural damage/symptoms; Stage B = structural damage without symptoms; Stage C = structural damage with symptoms; Stage D = advanced symptomatic disease with major functional limitation/hospitalization risk.
- NYHA Class I-IV: Functional status classification by symptom limitation from no limitation to symptoms at rest.
- Acute decompensated HF (ADHF): Sudden or gradual worsening that requires urgent outpatient escalation or hospitalization.
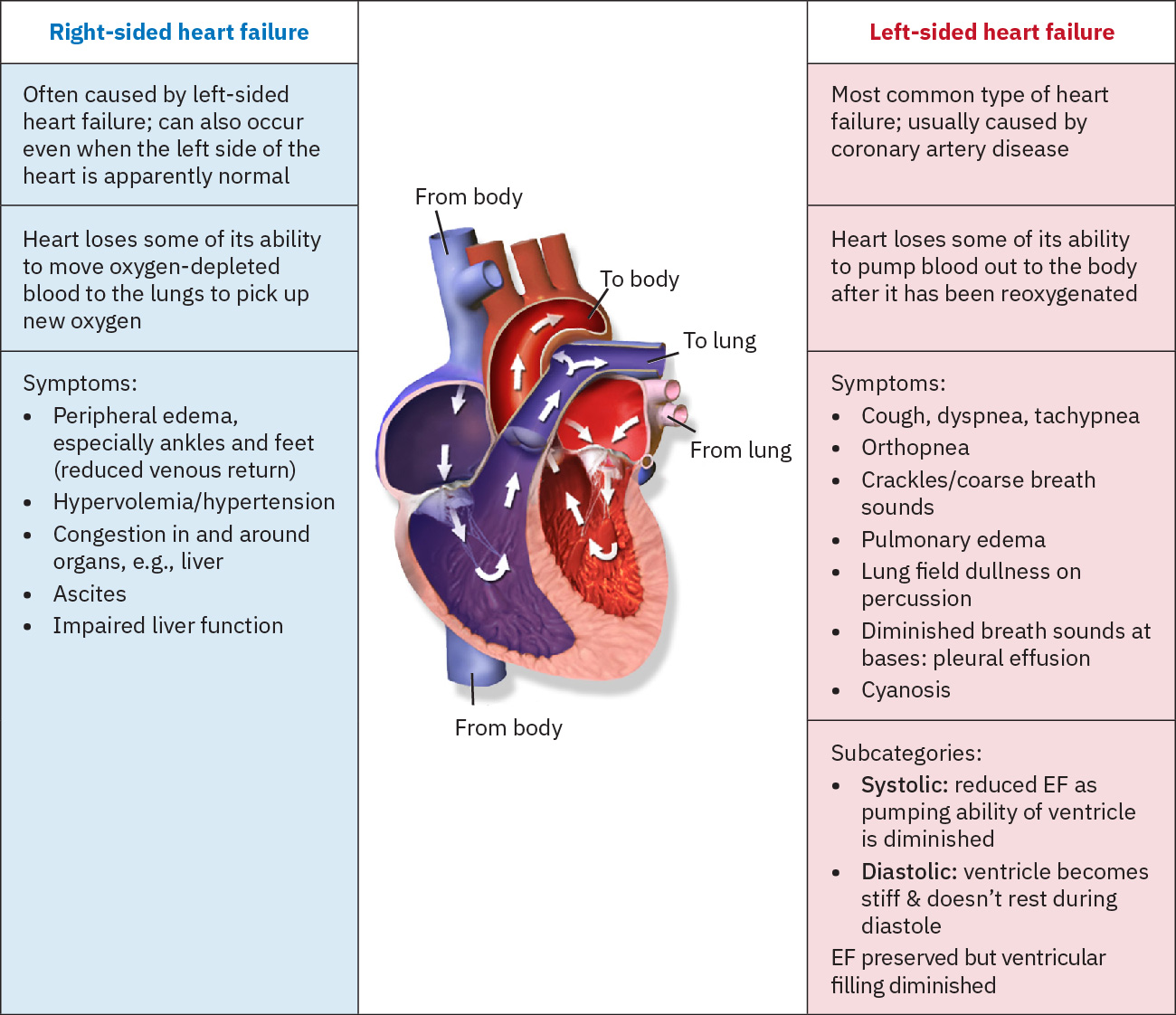 Illustration reference: OpenStax Clinical Nursing Skills Ch.18.2.
Illustration reference: OpenStax Clinical Nursing Skills Ch.18.2.
Nursing Assessment
NCLEX Focus
Questions prioritize distinction between expected baseline findings and acute decompensation requiring immediate provider notification.
- Assess baseline versus current status for edema burden, lung sounds, mental status, and kidney function.
- Recognize that patients may underidentify early HF symptoms; use focused history plus targeted physical exam to close this gap.
- Start with general survey cues: patient position, breathing pattern, skin color/pallor, and baseline mentation.
- Assess left-sided and right-sided symptom patterns, including orthopnea/PND, edema distribution, ascites, and JVD.
- Characterize respiratory and cardiovascular findings by location and quality: adventitious lung sounds, cough/sputum color-consistency-amount, S1/S2 changes, murmurs, rhythm irregularity, and peripheral pulse quality.
- Grade peripheral edema trend when present (for example pitting depth scale from 0 to 4+) and compare with baseline limb findings.
- Assess for abdominal fluid-congestion findings (ascites, hepatomegaly, hepatosplenomegaly), especially in right-sided failure patterns.
- Assess NYHA functional class to contextualize expected tolerance and detect meaningful decline.
- Assess for ADHF cues: worsening dyspnea, increased edema, new/worse crackles, confusion, and oliguria.
- Weigh under consistent conditions (ideally morning post-void, same clothing) and treat rapid gain as decompensation cue (commonly around 3 lb/day or 5 lb/week).
- Treat orthopnea progression (for example needing more than two pillows or sleeping upright in a recliner) as worsening pulmonary-congestion evidence.
- Escalate frothy pink sputum or oxygen saturation below about 92% with crackles as acute pulmonary-edema concern.
- In pediatric congenital-HF contexts, assess feeding fatigue, diaphoresis during feeds/crying, poor growth, diminished peripheral pulses, hepatomegaly/abdominal distension, edema, and extra S3 gallop pattern.
- Escalate pallor, lethargy, bradycardia, apnea, or minimal spontaneous movement as impending cardiopulmonary collapse findings.
- Use structured chest-pain assessment (for example PQRST) when ischemic contribution is suspected in HF deterioration.
- Trend diagnostics used in HF workup and monitoring: BNP (normal commonly <100 pg/mL), chest X-ray pulmonary congestion/cardiomegaly, echocardiography ejection fraction (normal commonly around 55-60%, hyperdynamic >70% context with diastolic dysfunction concern, and <40% with higher serious-dysrhythmia risk), and ECG rhythm findings.
- Interpret BNP with serial trend plus clinical context because ventricular stretch from hypervolemia generally raises BNP with worsening congestion risk.
- Follow adjunct laboratory trends for severity and comorbidity impact: potassium, BUN/creatinine, hemoglobin A1C, liver enzymes, and TSH.
- Assess appetite, early satiety, and food-variety pattern because repeated low-variety intake in HF can increase micronutrient-deficiency risk.
- Use EF context with ventricular-volume trends when available (
SV = EDV - ESV,EF = SV/EDV) to interpret contractile performance over time.
Nursing Interventions
- Prioritize oxygenation in acute worsening: place in High Fowler position and apply ordered supplemental oxygen to maintain saturation at or above 92%.
- Administer and monitor HF medication classes safely and watch for adverse effects, especially electrolyte imbalance from diuretic therapy.
- Use frequent hemodynamic checks when administering beta-blockers, ACE inhibitors/ARBs, diuretics, and nitrates; hold and escalate when ordered safety parameters are not met (for example pulse <60 bpm).
- In severe low-output decompensation (for example post-MI or post-cardiac-surgery contexts), prepare for temporary intra-aortic-balloon-pump support and monitor femoral-access bleeding plus distal pulse changes.
- Reconcile RAAS-modifying regimens carefully; avoid concurrent ACE inhibitor + ARB + ARNI combinations unless a specialist transition protocol explicitly directs timing and substitution.
- Teach symptom self-management and urgent reporting thresholds to reduce preventable ADHF admissions.
- Reinforce low-sodium dietary strategy (target commonly <2 g/day), daily weights, blood-pressure logging, and follow-up adherence.
- Teach practical sodium-control skills: read nutrition labels, track daily intake, and reduce processed/fast/canned high-sodium foods.
- For patients on RAAS-modifying and potassium-sparing pathways, reinforce avoidance of high-potassium diets/salt substitutes when potassium risk is elevated.
- Teach fluid-limiting plans when prescribed (often less than about 1.5-2 L/day in advanced HF or pulmonary edema contexts).
- For clients with fatigue or dyspnea-limited intake, use small nutrient-dense meals and lower-effort meal-preparation strategies with dietitian collaboration.
- In persistent poor-intake or low-variety patterns, coordinate micronutrient-focused nutrition review and consider provider-directed supplementation planning.
- Perform/assist daily or weekly weights as ordered, monitor intake/output, and apply compression stockings if prescribed.
- Escalate immediately for sudden chest pain, severe shortness of breath, or new white/pink foamy sputum.
- Teach discharge risk-modification priorities: smoking cessation, reduced or no alcohol intake, gradual aerobic activity progression, and adherence to follow-up plans.
- Reinforce recommended vaccines (influenza, COVID-19, and pneumococcal) to reduce infection-triggered HF recurrence risk.
- Encourage activity-rest balance, elevated head-of-bed/sleep positioning for orthopnea, smoking cessation, and participation in cardiac-rehabilitation-across-care-transitions.
- Cluster care and scheduled rest periods to reduce fatigue and cardiac workload.
- Support psychosocial coping and refer to mental-health and support-group resources when disease burden affects adherence and quality of life.
- In pediatric congenital-HF pathways, prepare for class-based medication plans (loop/thiazide diuretics, ACE inhibitor therapy, aldosterone antagonist, beta-blocker, inotrope, antiarrhythmic) with close rhythm and electrolyte surveillance.
- For pediatric digoxin pathways, count apical pulse for one full minute before dosing, hold and notify provider when infant apical heart rate is under 100 bpm (or below ordered age-specific parameters), and monitor for nausea, vomiting, bradycardia, or dysrhythmia toxicity cues.
- Track potassium and magnesium closely in children receiving digoxin plus diuretic therapy because electrolyte depletion increases toxicity risk.
- Coordinate frequent follow-up and escalation when congenital-defect correction or transplant-level evaluation is needed for end-stage deterioration.
- Use SMART-oriented evaluation targets: oxygen saturation >=92%, ability to complete clustered ADLs with less dyspnea, edema ⇐2+, stable weight/fluid status, and medication reconciliation aligned with active orders.
ADHF Escalation Risk
Worsening dyspnea, edema progression, confusion, and falling urine output can indicate acute decompensation requiring urgent escalation.
Pharmacology
| Drug Class | Examples | Key Nursing Considerations |
|---|---|---|
| Diuretic therapy | class-based HF use | Reduces congestion; monitor daily weight, urine output, potassium, BUN/creatinine, and hemodynamic tolerance. Give IV furosemide slowly to reduce ototoxicity risk. |
| ACE inhibitor, ARB, and ARNI therapy | class-based HF use | Modify RAAS pathways to lower pressure load and adverse remodeling; monitor blood pressure, renal function, and potassium trends. |
| Beta-blocker therapy | metoprolol (beta-1 selective), carvedilol (nonselective) | Slows rate and may improve ventricular filling; monitor for intolerance, bronchospasm risk in reactive-airway disease, and worsening status. |
| Aldosterone-antagonist therapy | class-based HF use | Supports fluid reduction; monitor electrolyte and kidney-function trends. |
| sodium-glucose-cotransporter-2-inhibitors (SGLT2 inhibitor therapy) | class-based HFrEF use | Originally diabetes-focused class now used in HF pathways; monitor volume status and tolerability. |
| ivabradine and hydralazine/isosorbide adjunct pathways | selected symptomatic HFrEF use after first-line optimization | Target heart-rate reduction or afterload reduction when symptoms persist; monitor for bradycardia and hypotension. |
| Inotrope therapy | digoxin and class-based acute/advanced use | Supports contractility in selected cases; requires close hemodynamic monitoring. |
Clinical Judgment Application
Clinical Scenario
A patient with chronic HF baseline NYHA Class II reports 3-lb overnight weight gain, worsening dyspnea, and new ankle edema with diminished urine output.
- Recognize Cues: Rapid weight increase, respiratory worsening, edema progression, and oliguria indicate fluid-retention acceleration.
- Analyze Cues: Findings suggest transition from chronic baseline toward ADHF.
- Prioritize Hypotheses: Immediate priority is preventing respiratory and perfusion deterioration.
- Generate Solutions: Escalate promptly, intensify monitoring, and implement ordered oxygen/diuretic interventions.
- Take Action: Communicate trend data and reassessment findings to the provider without delay.
- Evaluate Outcomes: Dyspnea improves with oxygen saturation >=92%, edema trends toward ⇐2+, urine output improves, and weights stabilize.
Related Concepts
- coronary-artery-disease - Ischemic myocardial injury is a common HF cause and progression driver.
- cardiovascular-system - Core cardiovascular physiology explains forward-flow failure and congestion patterns.
- cardiovascular-and-peripheral-vascular-nursing-assessment - Structured exam findings support early decompensation recognition.
- fluid-volume-overload-hypervolemia - Fluid-retention pathways overlap with edema and pulmonary-congestion management.
- angiotensin-receptor-neprilysin-inhibitors - ARNI pathways are guideline-directed HFrEF therapy options with dedicated safety monitoring.
- sodium-glucose-cotransporter-2-inhibitors - SGLT2 inhibitors support HF outcomes through osmotic diuresis and ventricular workload reduction.
- ivabradine - If-current inhibition can be used as adjunct heart-rate strategy in selected stable HFrEF pathways.
- congenital-heart-defects-acyanotic-and-cyanotic-patterns - Pediatric structural lesions are common upstream causes of infant HF.
- pulmonary-hypertension-in-congenital-heart-disease-and-pphn - Pulmonary vascular pressure overload can coexist with and worsen HF trajectories.
- digoxin - High-alert inotrope pathway in selected pediatric heart-failure plans.
- peripartum-cardiomyopathy - Pregnancy-associated systolic dysfunction can present as acute or progressive HF.
- readmission-reduction-programs-hrrp-and-value-based-purchasing - Discharge and follow-up reliability strongly affect 30-day HF outcomes.
Self-Check
- Which findings distinguish expected chronic HF from acute decompensated HF?
- How does NYHA classification change nursing assessment expectations and escalation thresholds?
- Which home-monitoring cues should trigger same-day provider contact in HF self-management?