Congenital Heart Defects Acyanotic and Cyanotic Patterns
Key Points
- Congenital heart defects (CHDs) are structural abnormalities present at birth that alter blood-flow pathways.
- Acyanotic defects are usually left-to-right shunts that increase pulmonary and right-sided volume load.
- Cyanotic defects often include right-to-left shunting or severe outflow obstruction, causing systemic hypoxemia.
- Pulse-ox CCHD screening at about 24 hours helps detect severe lesions early.
- Hypercyanotic episodes (Tet spells) are emergencies requiring rapid positioning, oxygen support, and escalation.
- Progressive complications include heart-failure and pulmonary-hypertension-in-congenital-heart-disease-and-pphn in unrepaired or hemodynamically significant lesions.
Pathophysiology
CHDs alter normal neonatal circulation after fetal-shunt closure. Defects may create abnormal communication between chambers/vessels, vessel transposition, or obstructive outflow pathways. The resulting hemodynamics determine oxygenation status, ventricular workload, and progression to heart failure.
Acyanotic pathways usually shunt oxygenated blood from high-pressure left heart to lower-pressure right heart. This does not initially cause cyanosis but can produce pulmonary overcirculation, pulmonary hypertension, and right-sided failure over time.
Cyanotic pathways reduce effective systemic oxygen delivery by moving deoxygenated blood into systemic circulation or by limiting pulmonary blood flow. Chronic hypoxemia may drive polycythemia, hyperviscosity, thrombosis risk, and long-term cardiopulmonary strain.
Classification
- Acyanotic left-to-right shunt defects: patent-ductus-arteriosus, atrial-septal-defect, patent-foramen-ovale, and ventricular-septal-defect.
- Cyanotic mixed-flow defects: tetralogy-of-fallot and transposition-of-the-great-arteries.
- Oxygenation cue split: Acyanotic patterns usually remain around 90% or higher oxygen saturation, whereas cyanotic right-to-left pathways commonly present below 90%.
- Single-ventricle severe-flow defect: hypoplastic-left-heart-syndrome.
- Left-sided obstructive defect patterns: coarctation-of-the-aorta and congenital-mitral-stenosis.
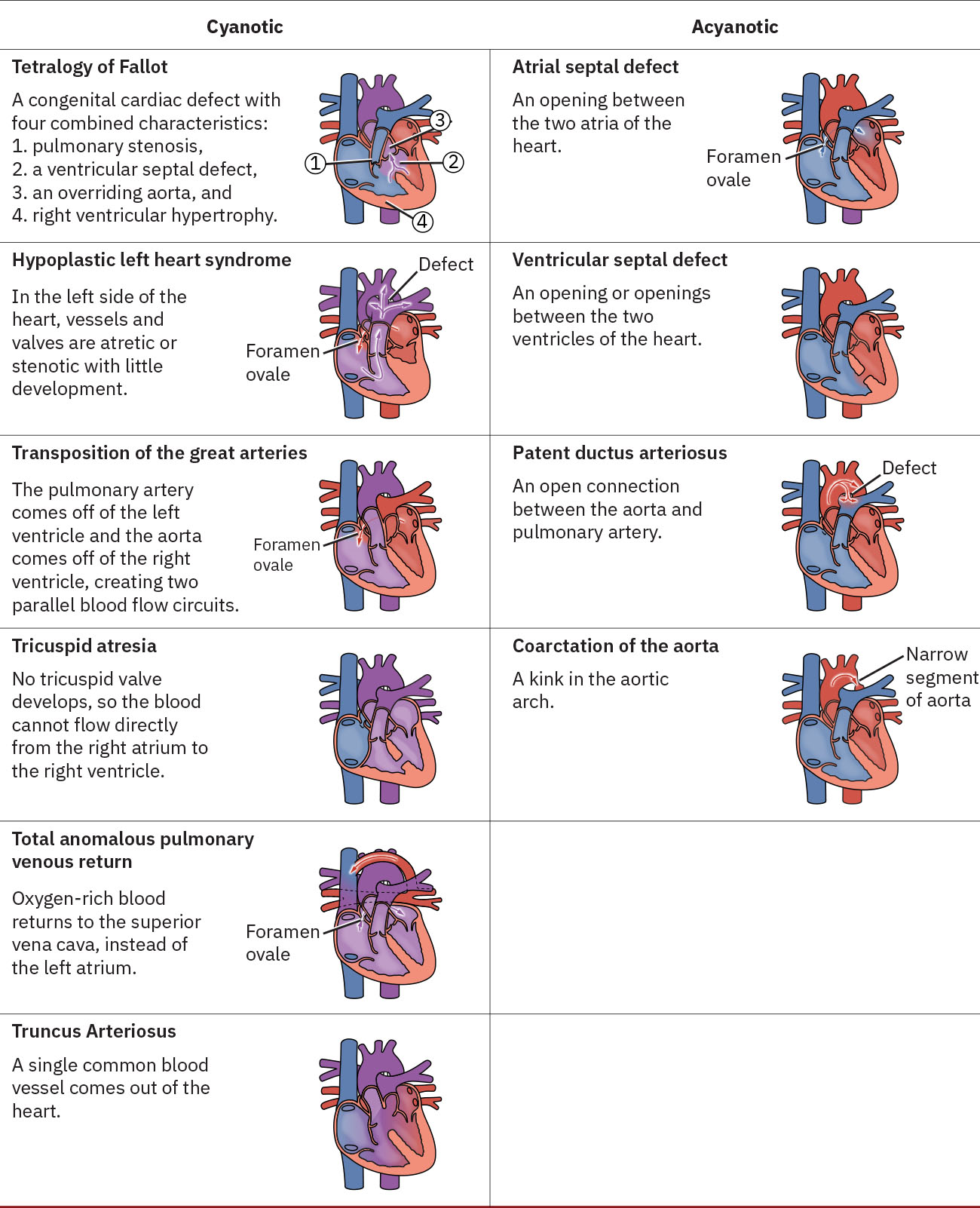 Illustration reference: OpenStax Maternal-Newborn Nursing Ch.25.2.
Illustration reference: OpenStax Maternal-Newborn Nursing Ch.25.2.
Nursing Assessment
NCLEX Focus
Distinguish pulmonary-overcirculation acyanotic patterns from hypoxemic cyanotic patterns, then prioritize deterioration cues.
-
Review prenatal and perinatal risk context (family CHD history, maternal hyperglycemia, smoking, alcohol, infection, and selected medication exposure).
-
Verify CCHD screening approach at about 24 hours: right hand (preductal) and foot (postductal) pulse oximetry, with concern when saturation is under 95% or pre/postductal difference exceeds 3%.
-
For abnormal CCHD screening, ensure repeat testing pathway is followed (up to three readings about one hour apart) and escalate persistent positives for diagnostic echocardiography.
-
In PDA concern, assess tachypnea, feeding difficulty, poor weight gain, tachycardia, continuous machine-like murmur, cardiomegaly, and widened pulse pressure.
-
In ASD/PFO/VSD concern, assess murmur, feeding fatigue, growth trajectory, pulmonary congestion signs, and right-sided volume-load progression.
-
In undiagnosed-infant concern, ask caregiver-focused feeding-history questions (weak suck, prolonged feeding, early fatigue, diaphoresis/cyanotic color change with feeding or crying, and poor weight-gain trend).
-
In cyanotic CHD concern, assess persistent central cyanosis and hypoxemia that may show limited response to routine oxygen delivery.
-
Assess for polycythemia-related risk (viscosity increase, thrombosis/embolic risk) and potential iron-deficiency anemia with chronic cyanotic physiology.
-
In postoperative or high-risk CHD contexts, monitor for thrombosis cues linked to blood stasis, arrhythmia burden, central-line/shunt exposure, and hyperviscosity.
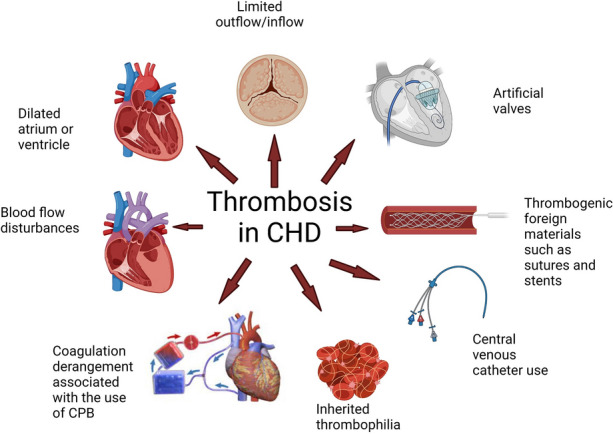 Illustration reference: OpenRN Nursing Health Promotion Ch.17.5.
Illustration reference: OpenRN Nursing Health Promotion Ch.17.5. -
In hypercyanotic (Tet-spell) concern, assess sudden irritability, hyperventilation, worsening cyanosis, lethargy, syncope, and shock progression.
-
In coarctation concern, compare upper and lower extremity blood pressures and pulses, and monitor renal-perfusion cues including urine-output decline.
-
Compare upper-versus-lower extremity color and temperature, and escalate significant perfusion asymmetry as possible flow-restriction evidence.
-
In congenital mitral stenosis concern, assess dyspnea, feeding intolerance, poor growth, murmur, and signs of left-sided heart failure or pulmonary hypertension.
-
Assess for complication progression cues across lesion types: worsening tachypnea/dyspnea, fatigue, syncope, hypotension, edema, hepatomegaly/abdominal distension, and declining perfusion.
-
Treat lethargy, limpness, altered consciousness, or asymmetrical movement as emergency findings that may indicate shock or stroke-level hypoperfusion.
Nursing Interventions
- Trend oxygenation, perfusion, feeding tolerance, growth, and heart-failure signs with frequent reassessment and early pediatric-cardiology escalation.
- Support energy-conserving feeding plans and hydration monitoring in infants with high cardiopulmonary workload.
- Strictly monitor intake and output trends, including hourly urine output when acutely unstable.
- Trend daily weight on the same scale/conditions; rapid gain (about 0.5 kg in 24 hours) can indicate fluid retention.
- Maintain thermal stability and treat fever promptly to reduce oxygen consumption and cardiac workload.
- Cluster care and maintain a calm environment with rest periods to reduce metabolic demand.
- Use high-calorie feeding plans as ordered: offer at least every three hours, upright positioning, and target feed duration around 20 minutes to limit cardiopulmonary strain.
- Escalate feeding strategies (including gavage pathway per order) when feeds exceed about 30 minutes or intake/weight gain are inadequate.
- During hypercyanotic episodes, calm the infant/child, position knees to chest (or allow squatting in older children), provide high-concentration oxygen, and use blow-by method if mask distress worsens agitation.
- Prepare for ordered medications and advanced support during persistent hypercyanotic episodes; escalate rapidly for altered consciousness or worsening perfusion.
- Administer supplemental oxygen only as needed in left-to-right shunt physiology because excess oxygen can lower pulmonary vascular resistance and worsen pulmonary overcirculation.
- Reinforce diagnostic pathways (echocardiography as key confirmation tool) and communicate trend changes in murmur, saturation, and perfusion.
- Anticipate intervention pathways by lesion severity: monitoring for small asymptomatic defects versus catheter-based closure or surgical repair for significant lesions.
- Support family teaching on staged surgical plans when relevant (for example HLHS Norwood/Glenn/Fontan sequence) and long-term follow-up for arrhythmia or heart-failure risk.
- Escalate for suspected pulmonary-hypertension burden and support advanced pathways (oxygen, pulmonary vasodilator plans, defect correction, or ECMO) based on deterioration severity.
Cyanotic Deterioration Risk
Cyanosis with respiratory distress, poor perfusion, or decreased responsiveness is a pediatric emergency even when blood pressure is not yet severely reduced.
Pharmacology
| Drug Class | Examples | Key Nursing Considerations |
|---|---|---|
| nsaids | Indomethacin/ibuprofen context for PDA closure | Used in selected PDA pathways to reduce prostaglandin-mediated ductal patency; avoid in ductal-dependent critical lesions before definitive repair. |
| prostaglandin E1 pathway | Ductal-patency infusion bridge | Maintains ductal flow in critical lesions until catheter/surgical intervention is possible. |
| opioids | Morphine in hypercyanotic episodes | May reduce oxygen demand and agitation when prescribed for Tet-spell management. |
| antihypertensive and antidysrhythmic therapies | Lesion-specific pediatric regimens | Used to control pressure/rhythm burden in selected obstructive or symptomatic pathways. |
| antibiotics | Procedure-prophylaxis context | Some high-risk valve lesions may need prophylaxis planning before selected procedures. |
Clinical Judgment Application
Clinical Scenario
A 2-month-old infant has poor feeding, diaphoresis during feeds, tachypnea, and a new loud murmur. Pulse oximetry is borderline and weight gain is below expected trend.
- Recognize Cues: Murmur with feeding intolerance, respiratory effort, and growth faltering.
- Analyze Cues: Pattern suggests hemodynamically significant CHD with evolving heart-failure risk.
- Prioritize Hypotheses: Immediate concern is inadequate oxygen delivery and cardiopulmonary workload.
- Generate Solutions: Escalate cardiology evaluation, optimize feeding/oxygenation strategy, and monitor perfusion closely.
- Take Action: Implement ordered monitoring and supportive interventions, communicate trend changes rapidly.
- Evaluate Outcomes: Oxygenation and feeding stability improve, and lesion-specific treatment plan is initiated.
Related Concepts
- physiological-adaptation-and-transition - Fetal-to-neonatal circulation transition frames postnatal CHD physiology.
- congenital-genetic-and-acquired-complications - Broader high-risk newborn context for multisystem congenital conditions.
- basic-newborn-care - Includes universal pulse-ox screening for critical CHD detection.
- cardiovascular-and-peripheral-vascular-nursing-assessment - Murmur, perfusion, and blood-pressure comparison assessment framework.
- heart-failure - Common complication pathway in unrepaired or residual CHD burden.
- pulmonary-hypertension-in-congenital-heart-disease-and-pphn - CHD complications include pulmonary vascular pressure overload and refractory hypoxemia pathways.
- anticoagulants - Selected high-risk CHD pathways require thrombosis-prevention planning with bleeding-risk surveillance.
Self-Check
- How do acyanotic and cyanotic CHDs differ in primary hemodynamic problem?
- Which cues indicate a hypercyanotic episode requiring emergency escalation?
- Why are upper-versus-lower extremity BP comparisons important in coarctation screening?