Cardiovascular System
Key Points
- The cardiovascular system includes the heart, blood, and blood vessels that deliver oxygen/nutrients and remove wastes.
- Blood composition (red cells, white cells, platelets, plasma) links oxygen transport, host defense, and clotting support to cardiovascular stability.
- The heart sits in the mediastinum; right-sided chambers receive and send deoxygenated blood to lungs, while the left ventricle is the primary systemic pump.
- Arteries, capillaries, and veins form the transport network linking tissue metabolism and organ function.
- Veins are lower-pressure return vessels and larger veins use valves to support one-way flow toward the heart.
- Systemic veins also function as blood reservoirs; venoconstriction can mobilize venous reserve toward central circulation when demand rises.
- Sympathetic stimulation increases heart rate and contractility, whereas parasympathetic input lowers heart rate.
- Local tissue autoregulation and endocrine pathways (including RAAS and ADH signaling) adjust arteriolar tone and blood pressure.
- Arteriosclerosis and atherosclerosis increase vascular stiffness and luminal narrowing, raising afterload and long-term perfusion risk.
- Aging and atherosclerotic change reduce perfusion reserve and increase risk for falls, edema, stroke, and myocardial events.
- Cardiovascular disease is the leading global cause of death, with more than 19 million deaths annually.
Pathophysiology
Cardiovascular function depends on coordinated cardiac pumping and vessel integrity to maintain tissue perfusion and waste removal. Oxygenated blood is distributed through arteries to capillaries, where exchange occurs, then returned via veins for recirculation and organ-based filtration. This transport role is integrated with other organ systems: lungs support gas exchange, the gastrointestinal system supports nutrient availability, kidneys influence volume and pressure regulation, and lymphatic pathways return interstitial fluid to circulation.
The heart has four chambers (right/left atria and right/left ventricles) and layered structure (endocardium, myocardium, and outer epicardial-pericardial interface). Atrial contraction fills ventricles, then ventricular contraction drives pulmonary and systemic circulation. Coronary arteries supply myocardium, and coronary perfusion occurs primarily during diastole. Anatomically, the cardiac base aligns near the third costal cartilage and the apex lies left of the sternum near the fourth-fifth intercostal region, which helps guide precise precordial auscultation. Cardiac valves preserve unidirectional flow: AV valves (tricuspid right, mitral left) open from atria to ventricles and close during ventricular systole to limit regurgitation, while semilunar valves (pulmonic right, aortic left) open during ventricular ejection and close in relaxation. Flow sequence is right atrium → tricuspid valve → right ventricle → pulmonic valve → lungs → left atrium → mitral valve → left ventricle → aortic valve → systemic circulation.
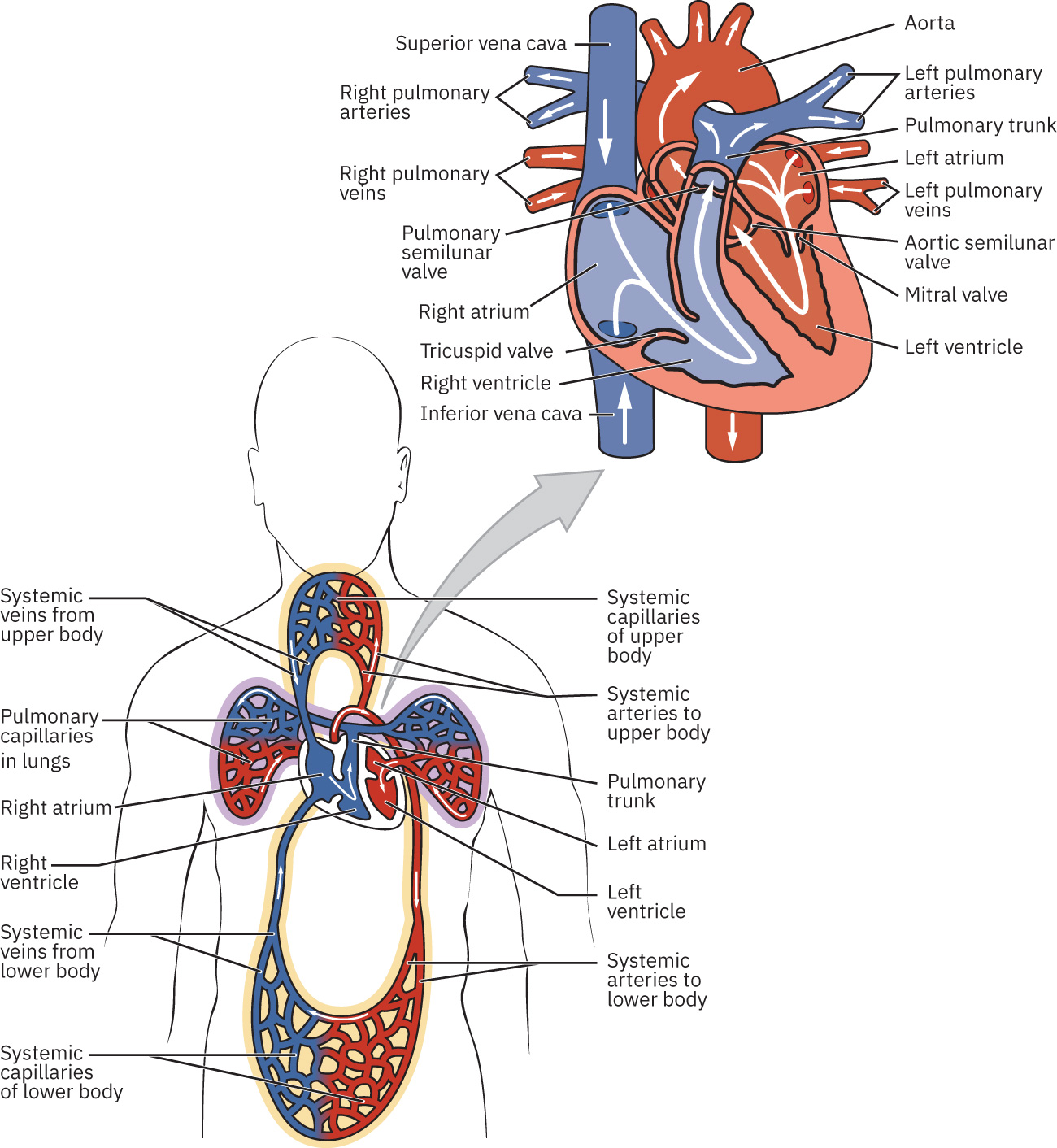 Illustration reference: OpenStax Clinical Nursing Skills Ch.18.2.
Illustration reference: OpenStax Clinical Nursing Skills Ch.18.2.
Pericardial structure includes fibrous pericardium and serous pericardium (parietal plus visceral layer/epicardium). The pericardial cavity between layers contains lubricating fluid (often around 15-50 mL in referenced adult anatomy framing) that reduces friction during cardiac motion. Heart-wall layers from superficial to deep are epicardium, myocardium, and endocardium. The myocardium is the thickest layer and the primary contractile tissue; the endocardium is endothelial lining continuous with vascular endothelium and also covers cardiac valves. Myocardial fiber orientation is not linear; spiral and figure-eight muscle architecture supports efficient chamber compression and forward ejection. The left ventricular wall is normally thicker and more muscular than the right because it must generate higher pressure to overcome systemic vascular resistance (roughly near 90 mm Hg arterial context), whereas the right ventricle pumps into the lower-resistance pulmonary circuit (roughly 8-20 mm Hg context).
Systemic circulation and pulmonary circulation are linked loops: pulmonary flow sends blood to lungs for gas exchange, and systemic flow distributes oxygenated blood to tissues before venous return. Veins hold a major portion of circulating volume at baseline and can shift reserve blood centrally through venoconstriction. In physiology framing, systemic veins hold about 64% of blood volume at baseline, with about 21% of venous blood in liver, bone-marrow, and integumentary venous networks. Vascular flow pathway follows arteries → arterioles → capillaries → venules → veins. Fluids move along pressure gradients, so chamber and vessel pressure changes during the cardiac cycle direct blood forward. During diastole, venous return fills atria and blood moves passively into ventricles; atrial systole augments ventricular filling, and ventricular systole then ejects blood into the pulmonary trunk and aorta.
Neurohormonal control continuously adjusts perfusion. Sympathetic stimulation increases heart rate and contractility, while parasympathetic activity decreases heart rate. Endocrine signals also influence vascular tone and circulating volume: epinephrine and norepinephrine support short-term redistribution of blood flow, and RAAS/ADH signaling increases vasoconstriction and fluid retention when perfusion pressure drops. Catecholamine signaling also supports coronary vasodilation in demand states, improving myocardial blood flow. Cardiovascular homeostasis also includes dynamic resource allocation: active tissues (for example exercising muscle) receive increased flow while organs such as the brain maintain strong baseline perfusion. Blood transport supports acid-base/water balance by delivering oxygen, nutrients, and hormones and by removing metabolic by-products including carbon dioxide. Protective roles are embedded in circulating components: WBCs support host defense and platelets with coagulation proteins form fibrin-rich hemostatic barriers after vessel injury. Thermoregulation is partly vascular: cutaneous vasodilation promotes heat loss in warm states, while diversion of blood away from skin in cold states preserves core temperature but raises extremity frostbite risk. The adult heart retains intrinsic electrical automaticity. Fastest pacemaker activity, usually from the sinoatrial node, sets sinus rhythm and propagates through the AV node, His bundle, bundle branches, and Purkinje fibers. AV nodal delay allows atrial contraction to complete ventricular filling before ventricular depolarization. Rapid Purkinje conduction then supports coordinated apex-to-base ventricular contraction for efficient ejection into the aorta and pulmonary trunk. Cardiac conductive-cell auto-rhythmicity depends on cyclic ionic flux: slow sodium entry (prepotential) to threshold, calcium-driven depolarization, and potassium-mediated repolarization back toward baseline. Cardiac contractile cells differ from pacemaker cells: they maintain a more stable resting potential, then undergo rapid depolarization, a calcium-mediated plateau, and repolarization. The plateau and prolonged refractory periods support effective mechanical ejection and help prevent premature re-excitation that would compromise perfusion. Calcium has dual functional roles in this process: it supports the plateau/refractory behavior and directly enables actin-myosin interaction through troponin-tropomyosin regulation during contraction. Excitation-contraction coupling includes L-type calcium influx and sarcoplasmic-reticulum calcium release (via ryanodine receptors), which amplifies cytoplasmic calcium and triggers actin-myosin sliding force. Pacemaker hierarchy matters clinically: SA node usually drives rhythm fastest, with slower fallback pacing from AV node and downstream His-Purkinje structures if upstream signaling fails. Electrolyte disruption, especially hypokalemia, increases dysrhythmia risk and should be corrected quickly when conduction instability appears. Concurrent low sodium, calcium, or magnesium can further destabilize conduction and weaken mechanical contraction, worsening perfusion. When clinically significant bradycardia causes instability, immediate priorities include respiratory and cardiovascular support, rapid monitoring, and urgent provider escalation while reversible causes are evaluated.
Intravascular volume is a core determinant of blood pressure and perfusion. Advanced hemodynamic relationships include CO = HR x SV, CI = CO/BSA, and SV = EDV - ESV; these trends are especially relevant in critical-care monitoring when compensation is unstable. Mean arterial pressure is linked to flow and resistance (MAP rises as CO and/or systemic vascular resistance increase), so heart-rate or stroke-volume shifts can directly alter perfusion pressure. Common reference ranges used in this context include CO about 4-8 L/min, CI about 2.5-4 L/min/m^2, and resting HR about 60-100 bpm.
Stroke volume is shaped by preload, contractility, and afterload. Ejection fraction (EF = SV/EDV) describes the proportion of ventricular blood ejected per beat and helps classify pump function; broadly, normal is about 50-70%, preserved function is typically above 50%, and clearly reduced systolic function is often seen at or below about 40% with compatible clinical findings.
The cardiac cycle includes coordinated atrial and ventricular systole/diastole phases that repeat continuously; efficient sequencing is required to preserve forward flow and organ perfusion.
During early ventricular filling, most preload enters passively with AV valves open and semilunar valves closed; atrial systole then provides additional filling before ventricular contraction.
Ventricular systole proceeds from isovolumetric pressure rise to ejection after semilunar-valve opening. Ventricular relaxation then includes isovolumetric relaxation before AV valves reopen, with arterial dicrotic-notch signaling after semilunar closure.
Ectopic impulse initiation outside the SA node can occur transiently; persistent ectopy may progress to clinically significant arrhythmia or fibrillation depending on trigger and substrate.
Cardiovascular regulation is integrated in the medulla through cardioaccelerator, cardioinhibitory, and vasomotor control pathways. Baroreceptors (aortic/carotid stretch sensing) and chemoreceptors (oxygen, carbon dioxide, and pH sensing) provide reflex input that continuously adjusts heart rate, vascular tone, and blood pressure.
With age, reduced vascular elasticity and myocardial reserve can impair blood-pressure regulation and oxygen distribution. Additional plaque burden narrows vessels and worsens flow, increasing risk of thrombosis, ischemia, and cardiorespiratory compromise. Dyslipidemia, diabetes, and tobacco exposure accelerate this process, and long-standing hypertension both results from and further worsens vascular stiffening. Diet patterns high in saturated fat and cholesterol can sustain LDL elevation, accelerate plaque accumulation, and increase long-term afterload stress that contributes to heart-failure progression. Perfusion instability can also occur at the opposite extreme with hypovolemia: volume loss from bleeding or dehydration lowers pressure and may produce confusion, cool clammy skin, oliguria, and rising creatinine as organ perfusion drops.
Pediatric cardiovascular physiology also has high-yield differences: major transitional circulation changes occur in the first days to weeks after birth, sympathetic cardiac innervation is less mature in newborns (bradycardia vulnerability), relatively small absolute blood loss can represent a large circulating-volume percentage, and higher metabolic rate increases myocardial workload. Pediatric vascular access can be more challenging because of smaller veins and proportionally greater subcutaneous tissue.
Modifiable lifestyle factors, including tobacco exposure, low physical activity, and high-risk dietary patterns, contribute substantially to cardiovascular disease burden. Early nursing health teaching and prevention counseling are key protective interventions.
Classification
- Core system components: Heart pump function, circulating blood volume/content, and vascular network.
- Blood-content domain: Red cells, white cells, platelets, and plasma functions that support oxygen carriage, immunity, and hemostasis.
- Cardiac structural units: Two atria and two ventricles with myocardial pumping and coronary circulation support.
- Valve and flow-control units: AV valves (tricuspid/mitral) and semilunar valves (pulmonic/aortic) that coordinate forward blood movement and minimize backflow.
- Transport pathways: Arterial delivery, capillary exchange, venous return.
- Circulation loops: Pulmonary circuit for oxygen uptake and carbon-dioxide unloading; systemic circuit for tissue delivery and venous return.
- Perfusion-control systems: Local autoregulation, autonomic input, and endocrine pathways (catecholamines, RAAS, ADH).
- Intrinsic electrical-control system: SA node, AV node, AV bundle, bundle branches, and Purkinje network.
- Hemodynamic metrics: HR, SV, EDV, ESV, CO, and CI for trend-based perfusion interpretation.
- Pump-performance metrics: EF, preload, contractility, and afterload for systolic/diastolic performance framing.
- Risk progression states: Reduced elasticity, plaque accumulation, and thromboembolic risk.
Nursing Assessment
NCLEX Focus
Priority questions test recognition of cardiovascular red flags requiring immediate nurse notification or emergency activation.
- Observe for chest pain, dyspnea, cyanosis, acute dizziness, or sudden activity intolerance.
- Confirm auscultation landmarks and precordial orientation when heart sounds are difficult to localize (apex is typically left of sternum near the fourth-fifth intercostal region).
- Identify edema pattern changes, rapid weight gain trends (especially more than about 2-3 lb in 24 hours), and unilateral leg findings suggestive of DVT.
- Monitor for fatigue, palpitations, and symptomatic blood-pressure instability cues.
- Assess electrical and mechanical function together: telemetry/ECG activity without a palpable pulse indicates pulseless electrical activity and requires emergency escalation.
- In monitored settings, trend hemodynamic values and compensation patterns; very fast HR can lower ventricular filling and reduce stroke volume despite tachycardic compensation.
- Watch for low-perfusion clusters during volume loss (for example confusion, cool clammy skin, oliguria, rising creatinine) and escalate quickly when pattern worsens.
- Report potential PE signs urgently (sudden shortness of breath, chest pain, hemoptysis).
Nursing Interventions
- Encourage activity as tolerated and mobility measures to reduce stasis-related clot risk.
- Support heart-healthy habits: low-fat/low-cholesterol and low-sodium intake, smoking cessation, and weight management.
- Implement prescribed compression supports and monitor tolerance/safety.
- In postoperative DVT prevention, use SCDs only as ordered and do not apply them when unilateral DVT signs are present.
- Escalate emergent cardiopulmonary symptoms immediately per setting protocol.
- Anticipate hemodynamic effects of vasoactive therapy; for example, venous and arterial vasodilation can reduce preload and afterload.
Acute Perfusion Threat
New chest pain, severe dyspnea, or cyanosis can signal life-threatening cardiovascular compromise and requires immediate escalation.
Pharmacology
| Drug Class | Examples | Key Nursing Considerations |
|---|---|---|
| antihypertensives | Blood-pressure management context | Support adherence and monitor for dizziness/fall risk with position changes. |
| anticoagulants | DVT/AF-related context | Observe bleeding risk and promptly report signs of clot progression or PE. |
Clinical Judgment Application
Clinical Scenario
A resident with edema and heart-failure history develops rapid weight gain, increasing dyspnea, and perioral cyanosis.
- Recognize Cues: Worsening fluid overload indicators and oxygenation concern.
- Analyze Cues: Perfusion/respiratory status is deteriorating and may progress quickly.
- Prioritize Hypotheses: Immediate priority is acute cardiopulmonary compromise prevention.
- Generate Solutions: Obtain urgent nurse support, position safely, and prepare ordered oxygen/interventions.
- Take Action: Escalate rapidly with objective symptom/timing report.
- Evaluate Outcomes: Breathing and oxygenation stabilize or emergency pathway is activated.
Related Concepts
- heart-failure - Reduced pump performance commonly drives edema and dyspnea progression.
- hypertension-assessment-and-management - Chronic pressure burden accelerates vascular damage and cardiac strain.
- systematic-ecg-interpretation-and-dysrhythmia-triage - Rhythm instability increases stroke and clot risk.
- deep-vein-thrombosis - Venous clot can progress to pulmonary embolism.
- peripheral-iv-therapy-complications - Vascular monitoring and prompt escalation principles overlap.
Self-Check
- Which cardiovascular findings require immediate escalation rather than routine monitoring?
- How do age-related vessel changes increase fall and fatigue risk?
- Why is unilateral leg swelling with tenderness a high-priority report?