Cardiovascular and Peripheral Vascular Nursing Assessment
Key Points
- Cardiovascular assessment integrates symptom history with inspection, palpation, percussion, and auscultation.
- High-priority findings include chest pain progression, dyspnea, JVD, new murmurs, pulse asymmetry, and edema changes.
- Peripheral findings such as unilateral warmth/swelling, reduced pulses, and delayed capillary refill require rapid risk stratification.
- Acute peripheral-perfusion decline is screened with the 6P cluster: pain, pallor, poikilothermia, pulselessness, paresthesia, and paralysis.
Pathophysiology
Cardiovascular assessment identifies how well the heart generates forward flow and how effectively peripheral vessels distribute and return blood. Dysfunction can appear first as subtle changes in perfusion, effort tolerance, or fluid distribution.
Cardiac output (CO = stroke volume x heart rate) reflects interaction among preload, afterload, and contractility. Assessment trends should consider that early tachycardia may transiently support output, but persistent rapid rate can reduce filling and worsen forward flow.
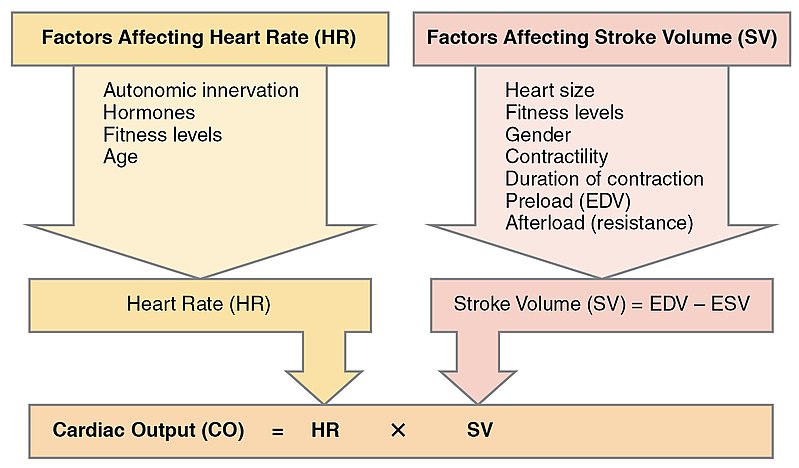 Illustration reference: OpenRN Health Alterations Ch.5.2.
Illustration reference: OpenRN Health Alterations Ch.5.2.
Because cardiopulmonary and vascular systems are interdependent, assessment requires simultaneous interpretation of central signs (heart sounds, rhythm, JVD) and peripheral signs (pulses, edema, skin changes).
Classification
- Subjective domains: Chest pain characteristics, dyspnea pattern, palpitations, risk factors, medication history.
- Objective domains: Inspection, palpation, percussion, auscultation, and pulse assessment.
- High-risk finding groups: Ischemic pain patterns, dysrhythmia cues, venous congestion, thromboembolic signs, and perfusion deficits.
Nursing Assessment
NCLEX Focus
Correlate heart sounds and pulse findings with symptoms and perfusion cues before concluding stability.
- Assess chest discomfort using structured symptom questions and trend changes over time.
- Start with immediate-threat clarifiers first (for example severe chest pain, severe dyspnea, syncope progression) before routine history expansion.
- If severe active ischemic or other critical symptoms are identified during interview, stop routine assessment and activate immediate emergency assistance per policy.
- Include fatigue, dyspepsia-like discomfort, leg swelling, and unexplained rapid weight gain when screening for possible cardiovascular decompensation.
- Use focused clarifying question sets for palpitations, dyspnea (including orthopnea/PND), edema progression, dizziness/syncope, and poor peripheral circulation.
- Use structured chest-pain interview logic (for example PQRSTU) to define provocation, quality, region/radiation, severity, timing, and patient interpretation.
- Include complete medication inventory (prescription, OTC, vitamins, herbals/supplements) because stimulant-containing products can provoke palpitations or rhythm changes.
- Include family-history detail for premature cardiovascular events (relationship and age at event) and integrate social-risk context (smoking/vaping, alcohol/drug use, activity, diet, stress, sleep, travel/occupation exposure).
- Inspect for color, warmth, movement, sensation changes, cyanosis/pallor, JVD, and bilateral extremity asymmetry.
- Treat JVD in upright positioning or with head-of-bed elevation around 30-45 degrees as an abnormal finding requiring follow-up.
- During bilateral extremity inspection, include superficial distended veins, hair-distribution changes, and presence/location/size of skin ulcers.
- Treat unilateral calf warmth, redness, swelling, and tenderness as urgent thromboembolic cues requiring rapid escalation.
- Inspect nails for clubbing as a possible chronic oxygenation/perfusion abnormality cue that may coexist with cardiopulmonary disease.
- Palpate capillary refill, edema grade, pulse quality and symmetry, and tenderness suggestive of DVT.
- Record baseline manual blood pressure and full 60-second apical pulse, then correlate with peripheral perfusion findings.
- Assess capillary refill after blanching (typically
<=2seconds in adults; up to<=3seconds may be acceptable in some pediatric contexts). - In darker skin-tone assessment contexts, great-toe capillary-refill checks can improve peripheral-perfusion screening sensitivity when discoloration cues are subtle.
- In pediatric assessments, prioritize apical pulse for heart-rate measurement (radial pulse is generally more reliable in adolescence) and interpret expected age-related heart-rate decline toward adult ranges by about school age.
- Use age-banded newborn baselines when trending instability (for example preterm HR about 120-170 and BP about 40-75/30-35; term neonate HR about 120-160 and BP about 60-75/45-55).
- In infants with possible CHD, ask focused feeding-history questions: weak suck, prolonged feeds, sleep before feed completion, diaphoresis or cyanotic color change with feeds/crying, and poor daily weight-gain trajectory.
- Grade pitting edema consistently (
0absent to4+very deep/rebound delayed) and trend by location/time-of-day. - Distinguish pitting versus nonpitting edema during palpation (preferably over a bony area such as the tibia) and document pattern.
- Use edema-grade detail when available (
+1trace to+4severe) with indentation depth and rebound time to improve trend precision.
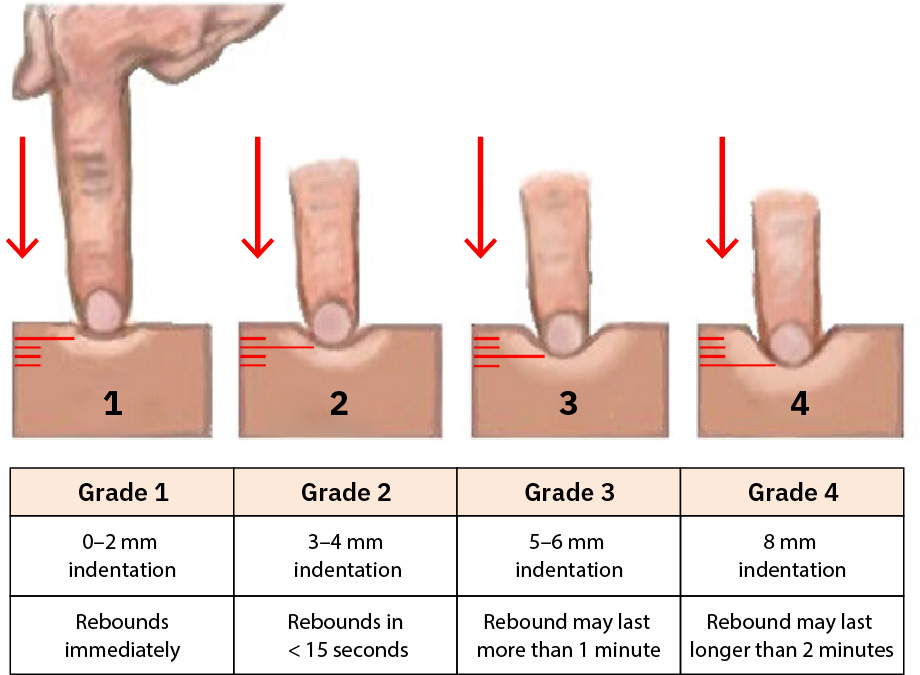 Illustration reference: OpenStax Clinical Nursing Skills Ch.24.2.
Illustration reference: OpenStax Clinical Nursing Skills Ch.24.2.
- Consider sudden weight increase as a possible fluid-overload companion cue when edema burden changes quickly.
- Assess delayed venous-filling patterns, cool skin, and abnormal dependent/elevated color changes as peripheral insufficiency cues.
- Recognize dependent edema as an early venous dysfunction cue, especially in nonambulatory patients or prolonged sitting/standing states.
- Assess new exertional unilateral leg pain relieved by rest (intermittent claudication pattern) and trend progression.
- Clarify whether leg pain remains exercise-linked or persists at rest; progression to rest pain suggests advanced ischemic burden.
- Use pain location and pulse/temperature findings together to localize likely arterial involvement (for example aortoiliac versus femoropopliteal versus tibial-peroneal distribution).
- Reassess JVD with repositioning when needed and integrate skin moisture/temperature findings (warm-dry vs cool-clammy) into perfusion interpretation.
- In pediatric CHD concern, compare upper-versus-lower extremity color, pulse quality, and skin temperature; significant arm-leg differences suggest possible flow restriction and require prompt escalation.
- Use known cardiac orientation landmarks when localizing heart sounds (the cardiac apex is typically left of the sternum near the fourth-fifth intercostal region).
- Percussion can screen for gross tissue-density changes but has limited cardiovascular specificity; prioritize high-quality auscultation and correlation with other findings.
- Use the
APE To Mansequence (aortic, pulmonic, Erb point, tricuspid, mitral) to standardize valve-area auscultation coverage. - Auscultate for S1/S2 integrity, extra sounds (S3/S4), murmurs, friction rub, and vascular bruits when indicated.
- Treat a new high-pitched, blowing/whooshing systolic sound at the cardiac apex as a possible mitral-regurgitation cue and escalate in clinical context.
- Expect
S2to be louder thanS1in aortic/pulmonic areas; use this pattern to confirm landmark placement. - Include femoral-artery bruit assessment when lower-extremity ischemia or pulse asymmetry is suspected.
- Auscultate carotids for bruits when clinically indicated; new turbulence can reflect atherosclerotic narrowing.
- Compare peripheral pulse
rate/rhythm/qualitybilaterally at carotid, radial, brachial, posterior tibial, and dorsalis pedis sites. - Palpate carotid pulses one side at a time to avoid compromising cerebral perfusion; avoid prolonged or forceful pressure over known diseased carotids because syncope risk rises.
- Use pulse-quality grading for documentation consistency:
0absent,+1weak,+2normal,+3strong,+4full/bounding. - Follow organizational pulse-grading convention consistently, because some settings use a
0-3scale while others use0-4. - If a pulse is not palpable, determine whether this is new versus chronic and verify flow with Doppler when available; treat new absent pulse as urgent.
- Treat absent pulse not detectable by Doppler and capillary refill greater than about 3 seconds as critical peripheral-perfusion findings requiring immediate escalation.
- If a pulsatile abdominal mass is present, avoid deep or forceful palpation and escalate promptly for vascular evaluation.
- Use the diaphragm first and then the bell at standard auscultation areas, and count apical pulse for a full 60 seconds when rhythm or rate clarity is needed.
- Count apical pulse for full
60seconds before many cardiac-medication administrations. - For irregular peripheral pulse, use a full
60-second count;30 x 2is acceptable only with regular rhythm. - Interpret
S1as systolic onset andS2as systolic end/diastolic onset when mapping timing of additional heart sounds. - Recognize
S3andS4context during interpretation:S3may occur with fluid-overload states (and may be physiologic in pregnancy), whereasS4often reflects reduced ventricular compliance. - In newborns, a transient murmur may be present during early postnatal ductus arteriosus closure; correlate with age, perfusion, oxygenation, and clinical stability.
- In infant CHD contexts, treat tachypnea, retractions, nasal flaring, lethargy/limpness, loss of consciousness, or asymmetrical movement as urgent findings and escalate immediately.
- In neonatal tachycardia states, remember blood pressure can remain near age norms while perfusion worsens; treat hypotension as a late decompensation sign.
- Assess suspected pericardial friction rub with the patient leaning forward and breath held while auscultating with the diaphragm at the apex/left lower sternal border.
- Integrate diagnostic context when available: echocardiography ejection fraction (normal about 55-70%), stress-test tolerance/symptom response, and post-procedure trend changes.
Diagnostic and Procedure-Focused Assessment Cues
- For echocardiography, correlate ejection fraction with dyspnea, edema burden, activity tolerance, and heart-failure trajectory.
- For broad cardiovascular diagnostics, prepare for ECG/echocardiography, stress testing (exercise or pharmacologic), electrophysiology studies, cardiac catheterization/angiography, and advanced imaging (MRI/MRA/CT/PET) based on clinical indication.
- Before contrast-related imaging/catheter procedures, verify allergy/adverse-reaction history and the patient’s ability to tolerate test positioning/protocol demands.
- Integrate relevant laboratory data with exam findings: coagulation profile, electrolytes, cardiac biomarkers (troponin/CK-MB), BNP, D-dimer, and lipid trends.
- For cardiac stress testing, monitor chest discomfort, dyspnea, rhythm changes, and hemodynamic tolerance; recognize that pharmacologic stress may use agents such as dobutamine or adenosine.
- For coronary angiography/cardiac catheterization, complete pre-procedure checks (informed consent, allergy/contrast risk, renal baseline, anticoagulant review, NPO status, IV access) and post-procedure surveillance (access-site bleeding, distal perfusion, urine output, and chest-pain recurrence).
- In ICU-level instability, integrate invasive trends (for example arterial-line blood pressure and selected pulmonary-artery-catheter data) with bedside exam cues rather than interpreting isolated values.
- For peripheral vascular workup, anticipate ABI comparison of brachial pressure with ankle pressures (dorsalis pedis/posterior tibial) and ensure standardized technique for valid interpretation.
- During initial objective assessment, verify level of consciousness and immediate physiologic stability before detailed exam progression.
- In acute respiratory/cardiac decompensation, anticipate ABG support for PaO2/PaCO2 and acid-base interpretation when pulse oximetry alone is insufficient.
Nursing Interventions
- Escalate severe or worsening chest pain, unstable rhythm signs, respiratory compromise, and thromboembolic cues immediately.
- Treat symptomatic tachycardia at rest (
HR >100 bpm), symptomatic bradycardia (HR <60 bpm), or hypotension (SBP <100 mm Hg) as urgent abnormal findings. - Escalate orthostatic blood-pressure changes, new dysrhythmia, new extra heart sounds (murmur,
S3,S4), new calf pain, or worsening shortness of breath without delay. - Reassess after interventions and document objective trend changes to support rapid team decisions.
- In critical abnormal findings, implement immediate-response workflow: rapid communication, continuous monitoring, symptom relief, and interdisciplinary coordination.
- In emergency pathways, escalate to rapid-response or higher-acuity activation when symptoms or perfusion trends indicate immediate deterioration risk.
- Administer ordered anti-ischemic/analgesic therapies (for example nitrates and opioid adjuncts) with close reassessment of pain, hemodynamics, and adverse effects.
- Reinforce risk-factor education, medication adherence, and follow-up care for chronic cardiovascular disease management.
- Provide clear education, advocacy, and discharge-follow-up coordination after abnormal findings or procedures.
Rapid Decompensation Risk
New chest pain with dyspnea, abnormal heart sounds, or acute perfusion deficits can precede cardiac arrest or embolic events.
Pharmacology
| Drug Class | Examples | Key Nursing Considerations |
|---|---|---|
| antihypertensive therapy | ACE inhibitor or beta-blocker contexts | Monitor perfusion response, blood pressure trends, and symptom burden. |
| anticoagulant therapy | Heparin or DOAC contexts | Use focused bleeding and thrombosis surveillance during assessment cycles. |
Clinical Judgment Application
Clinical Scenario
A patient has chest pressure, tachycardia, new bibasilar crackles, and 2+ bilateral pitting edema.
- Recognize Cues: Combined ischemic concern, congestion signs, and perfusion stress.
- Analyze Cues: Pattern suggests evolving cardiopulmonary decompensation.
- Prioritize Hypotheses: Immediate priority is stabilization and rapid diagnostic escalation.
- Generate Solutions: Trigger urgent provider response and initiate ordered monitoring/interventions.
- Take Action: Communicate objective findings and reassessment trend clearly.
- Evaluate Outcomes: Symptoms stabilize with timely treatment adjustment.
Related Concepts
- cardiovascular-system - Core structure and function framework for exam interpretation.
- peripheral-vascular-system-and-insufficiency-patterns - Peripheral insufficiency patterns refine lower-extremity findings.
- vital-sign-indicators-of-physiologic-functioning-and-homeostasis - Links assessment cues to global physiologic status.
- documenting-and-reporting-data - Objective, trend-based charting supports safe escalation.
Self-Check
- Which cardiovascular findings require immediate escalation regardless of prior history?
- How do S3/S4 or new murmur findings change nursing priority setting?
- Why should pulse, edema, and capillary refill always be interpreted bilaterally?