Systematic ECG Interpretation and Dysrhythmia Triage
Key Points
- Reliable ECG interpretation starts with a fixed sequence: rate, rhythm regularity, and waveform-interval assessment.
- A practical rate method is counting R waves in a 6-second strip (30 large boxes) and multiplying by 10.
- Normal interval references include PR 0.12-0.20 s, QRS 0.06-0.12 s, and QT about 0.36-0.44 s.
- Normal sinus rhythm is typically 60-100/min with regular R-R intervals and ordered P→QRS→T sequence.
- Long-QT concern increases when QT exceeds about 0.47 s in males or 0.48 s in females.
- New abnormal rhythms require immediate bedside assessment for signs of decreased cardiac output.
- Ventricular tachycardia, ventricular fibrillation, and third-degree AV block are emergency rhythms.
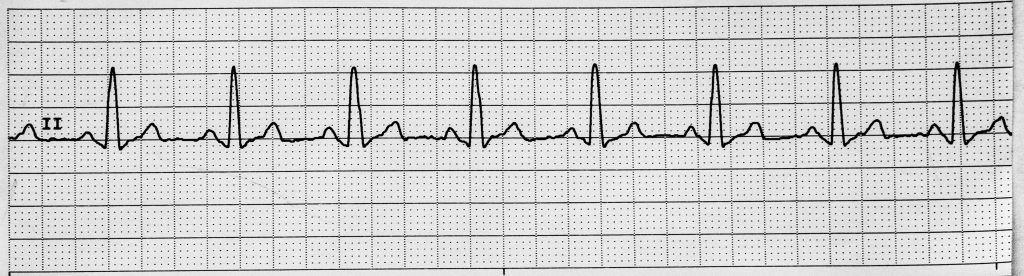 Illustration reference: OpenRN Health Alterations Ch.5.3.
Illustration reference: OpenRN Health Alterations Ch.5.3.
Pathophysiology
Cardiac rhythm interpretation reflects conduction through the intrinsic pacing system. The sinoatrial-node is the primary pacemaker (60-100 beats/min), the atrioventricular-node can pace at 40-60 beats/min if needed, and distal backup pacing can fall to 20-40 beats/min. Rhythm disruption at any level can impair coordinated ventricular contraction and reduce effective forward flow.
Normal conduction proceeds from the SA node through internodal pathways to the AV node, then through the bundle of His, right and left bundle branches, and Purkinje fibers. Sinus-rhythm framing includes SA-node impulse origin, rate usually 60-100/min, and SA-to-AV conduction arrival in about <=0.12 seconds without excessive delay.
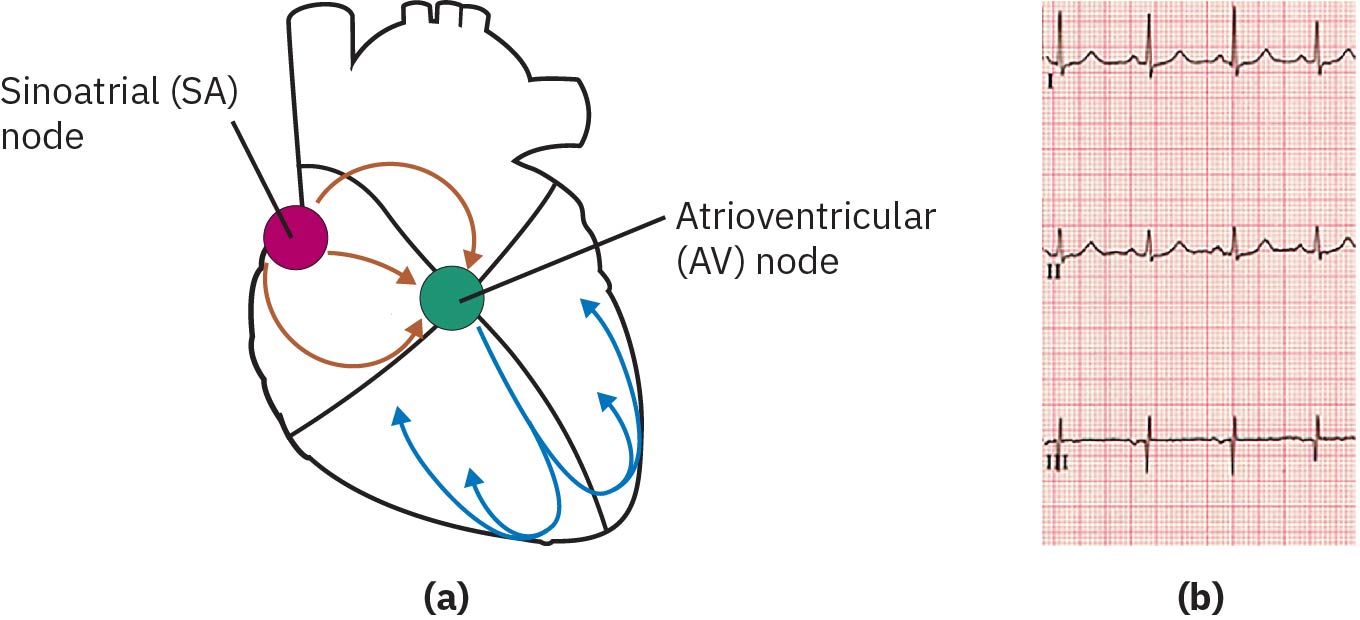 Illustration reference: OpenStax Clinical Nursing Skills Ch.18.2.
Illustration reference: OpenStax Clinical Nursing Skills Ch.18.2.
If SA-node firing fails, junctional AV-node pacing can provide a slower backup rhythm (often 40-60/min). This may transiently preserve blood pressure but can still produce low-output symptoms from reduced rate and filling dynamics.
Waveform timing represents depolarization and repolarization events. On ECG paper, one small box equals 0.04 seconds and one large box equals 0.2 seconds. Key measurements include PR interval (0.12-0.20 seconds), QRS duration (0.06-0.12 seconds), and QT interval (about 0.36-0.44 seconds), with QT behavior varying by rate and often trended as corrected QT. Cardiac-cycle mapping supports bedside interpretation: P wave aligns with atrial depolarization, QRS with ventricular depolarization and systolic onset, and T wave with ventricular repolarization/relaxation.
Ectopic beats can arise from foci outside the SA node and may present as premature atrial, junctional, or ventricular contractions. Common contributors include ischemia, medication effects (including agents such as digitalis), electrolyte or acid-base disturbances, hypoxia, autonomic stimulation shifts, stimulant exposure (for example cocaine, methamphetamine, or excess caffeine), structural/infectious cardiac injury, and blunt chest trauma patterns. Hypokalemia is a high-yield electrolyte trigger for dysrhythmia because repolarization stability is potassium dependent.
Classification
- Sinus rhythms: Impulse origin in SA node; may be normal, bradycardic, or tachycardic.
- Atrial rhythms: Electrical activity originates above ventricles but outside normal sinus pattern.
- Ventricular rhythms: Impulse origin in ventricles, often hemodynamically unstable.
- Conduction blocks: Delay or interruption in conduction pathways, including AV block patterns.
- Regularity patterns: Regular, irregularly irregular, regularly irregular, and occasionally irregular R-R spacing patterns.
Common Pattern Clusters
- Sinus tachycardia: Rate >100/min in adults, usually with regular PR and QRS intervals; common triggers include stress/exertion, dehydration, stimulants (including caffeine and cocaine), hypovolemia, anemia, and fever/infection.
- Sinus bradycardia: Rate <60/min in adults; may be physiologic (for example conditioned athletes) or symptomatic with low-output features.
- Atrial fibrillation: Irregularly irregular rhythm with absent distinct P waves, indeterminate PR interval, and usually narrow QRS (<0.12 s); prioritize low-output assessment and stroke-risk awareness. Rate-controlled AF is commonly 60-100/min, while sustained rates >100/min suggest rapid-ventricular-response physiology.
- Atrial flutter: Sawtooth atrial activity (often around 240-300/min) with non-1:1 ventricular response; PR interval is often not measurable.
- AF/flutter nutritional-risk context: Excess caffeine or alcohol intake and chronic cardiometabolic burden (for example high cholesterol and hypertension) can coexist with AF/flutter triggers and symptom worsening.
- PVC spectrum: Wide premature ventricular beats in sinus background; recognize bigeminy, trigeminy, couplets, R-on-T risk patterns, and ventricular-tachycardia run patterns (commonly >3 consecutive PVCs).
- Pause-pattern distinction: PVCs are often followed by a compensatory pause, while PAC-related pauses are commonly noncompensatory/shorter.
- SVT: Narrow-complex rapid rhythm (often >160/min) with difficult-to-identify P waves.
- SVT trigger context: Excess caffeine/alcohol exposure and broader cardiovascular disease burden can precipitate recurrent episodes.
- Ventricular tachycardia: Usually wide-complex rhythm around 150-200/min with no clear P waves/PR interval and high risk for hemodynamic collapse.
- Torsades de pointes: Polymorphic VT with twisting morphology, often associated with prolonged QT.
- Long-QT syndrome context: Nutritional-electrolyte deficits (especially low potassium, magnesium, or calcium) can prolong QT and raise torsades risk.
- Ventricular fibrillation: Chaotic baseline often >200/min with no discernible P/PR/QRS complexes and no organized cardiac output.
- Common VT/VF trigger set: Acute myocardial infarction, heart-failure decompensation, electrolyte disturbance, illicit substance exposure, shock states, and medication-related proarrhythmia.
- Asystole/PEA context: Asystole is electrical silence; PEA shows electrical activity without a palpable pulse.
- AV blocks: First-degree (PR >0.20 seconds), second-degree type I (progressive PR prolongation then dropped QRS), second-degree type II (fixed PR with dropped QRS and higher progression risk), third-degree (complete AV dissociation, often with slow ventricular escape around 20-40/min, wide QRS, and low-perfusion symptoms such as dizziness, syncope, or hypotension).
- Bundle branch block context: Intraventricular conduction delay with widened QRS (>0.12 seconds).
- Paced rhythms: Pacer spikes before paced chamber depolarization (atrial spike before P, ventricular spike before wide QRS, dual spikes in AV pacing).
- ST-segment abnormality: Elevation or depression can indicate acute ischemic/infarction patterns and requires immediate escalation.
- ACS subtype cueing: Unstable angina may show ischemic ST/T changes without biomarker rise; NSTEMI often has troponin rise without persistent ST elevation; STEMI shows ST elevation in contiguous leads with urgent reperfusion priority.
- Morphology warning clues: Marked P-wave amplitude change (atrial enlargement or potassium disturbance), absent P waves/irregular baseline (atrial fibrillation pattern), pathologic/enlarged Q-wave changes (possible infarction or ventricular enlargement), flattened T waves (possible impaired myocardial oxygen delivery), and ST elevation/depression shifts (acute injury or ischemia/hypoxia context).
- U-wave prominence cue: New or prominent U waves commonly suggest hypokalemia and can accompany tachycardia or other conduction instability.
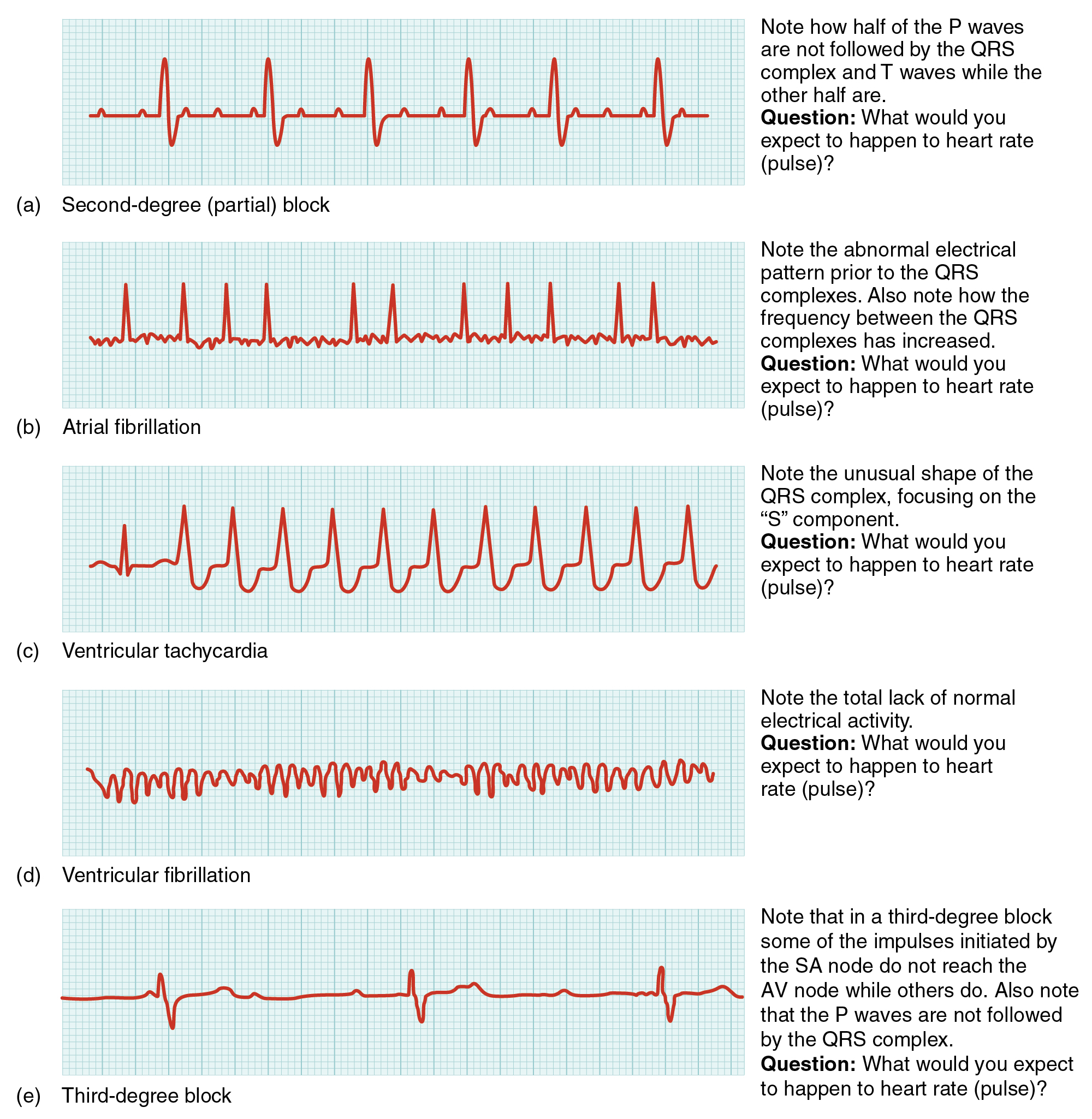 Illustration reference: OpenRN Nursing Pharmacology 2e Ch.6.3.
Illustration reference: OpenRN Nursing Pharmacology 2e Ch.6.3.
Nursing Assessment
NCLEX Focus
Priority is not only identifying the strip pattern but deciding whether perfusion is unstable now.
- Calculate atrial and ventricular rates using a consistent method, then compare for concordance.
- Treat machine-calculated rate as preliminary when tracing quality or rhythm complexity is poor and verify manually using strip-based counting.
- Use rhythm-favorable leads (commonly I, II, aVF, and V1) when available to improve rhythm-origin interpretation.
- Assess rhythm regularity with P-P and R-R interval patterns and inspect P-wave relationship to QRS.
- Confirm expected P-to-QRS association (normally 1:1 in sinus rhythm) when evaluating rhythm origin and conduction continuity.
- Use calipers or consistent box-count methods to verify whether P-P and R-R intervals march out regularly.
- Measure PR, QRS, and QT/QTc trends for conduction delay, widening, or repolarization risk.
- Check potassium (and related electrolytes such as magnesium) in new dysrhythmia patterns, and escalate hypokalemia promptly.
- Compare T-wave direction against dominant QRS direction; clear discordance can indicate repolarization abnormality requiring context-based escalation.
- Recognize interval clues: PR >0.20 seconds suggests AV delay/block; short PR can occur with accelerated AV conduction (for example pre-excitation patterns).
- Treat wide QRS as high-risk until explained (for example ventricular origin, bundle-branch conduction delay, or severe electrolyte effect such as hyperkalemia).
- Correlate strip findings with bedside status: blood pressure, pulses, capillary refill, chest pain, dyspnea, dizziness, confusion, or syncope.
- In frequent ectopy patterns, assess symptom burden trend (palpitations/flutter sensation, dizziness, chest pain, dyspnea) because occasional ectopy may be asymptomatic but recurrent ectopy can reduce tolerance.
- In AF/flutter presentations, do not rely on symptoms alone to separate rhythm subtype; trend telemetry and vital signs continuously and reassess oxygenation support need as rate increases.
Nursing Interventions
- Use a standardized interpretation workflow for every new strip to reduce misclassification and delay.
- For symptomatic sinus bradycardia, hold rate-lowering cardiac medications when pulse is below ordered threshold (commonly near 60/min), keep the patient in a safe low-activity position, and escalate promptly.
- For sinus tachycardia without immediate instability, support rest, fluids, and treatment of the underlying trigger while continuing hemodynamic reassessment.
- In symptomatic ectopy without collapse physiology, reduce adrenergic load (calm setting, coached deep breathing), then reassess rhythm and symptoms.
- Verify monitor artifact versus true dysrhythmia by reassessing the client and lead quality before escalation (for example poor electrode contact or client motion can mimic ventricular ectopy).
- Include ST/J-point review in unstable presentations; ST shift of about 1 mm or more can be clinically significant in the right context and lead pattern.
- Treat unstable VT/VF/complete heart block patterns as immediate emergency rhythms; use pulse status to separate synchronized cardioversion pathways from defibrillation/CPR pathways.
- For complete heart block with symptoms, maintain continuous telemetry, support oxygenation, keep the patient on bed rest, trend potassium/magnesium, and hold AV-node-slowing agents (for example beta-blockers or other antiarrhythmic pathways) pending urgent provider direction.
- In symptomatic second-degree AV block (especially Mobitz II), trend blood pressure and symptoms (for example dizziness, chest pain, worsening hypotension), administer atropine per protocol, and prepare for transcutaneous pacing as a temporary bridge with early provider escalation.
- When pacing dependence is expected, prepare for permanent pacemaker planning and reinforce teaching: carry device identification for security screening, clearly report pacemaker status before MRI, and follow device-safety precautions around direct household electromagnetic exposure.
- Escalate urgently for collapse-risk rhythms, especially ventricular tachycardia, ventricular fibrillation, and complete AV block.
- Match device and mode to setting and rhythm goal: AED pathways are designed for bystander automated analysis/defibrillation, while manual defibrillators support monitored rhythm viewing, synchronized cardioversion, and external pacing when indicated.
- During in-hospital arrest response, support role clarity and closed-loop communication, and anticipate compressor rotation about every
1-2minutes to preserve CPR quality. - Follow unit protocols for rapid response, defibrillation/cardioversion pathways, and continuous reassessment after interventions.
- In new ectopy clusters, check and correct trigger labs including potassium, calcium, and magnesium, and reinforce caffeine/alcohol trigger reduction.
- Do not assume ECG confirms effective pumping; integrate pulse and perfusion assessment and treat pulseless electrical activity as a no-output emergency.
- If ischemic concern remains despite nondiagnostic ECG, continue escalation for further testing (for example echocardiography or other imaging/biomarker pathways) because not all infarction patterns are captured on initial tracing.
- Use concrete outcome targets after stabilization: improved BP/heart-rate perfusion profile, oxygen saturation commonly above 92%, improved activity tolerance, and teach-back on rhythm-management self-care (for example smoking cessation, diet/weight modification, and medication adherence).
Perfusion-First Response
An abnormal ECG without symptoms still requires prompt evaluation, but any sign of decreased cardiac output mandates immediate emergency escalation.
Clinical Judgment Application
Clinical Scenario
A telemetry client develops a rapid wide-complex rhythm. The monitor alarm triggers, blood pressure drops, and the client reports severe light-headedness.
- Recognize Cues: Wide-complex tachycardia, hypotension, and acute symptoms indicate unstable rhythm physiology.
- Analyze Cues: Electrical instability is causing reduced ventricular filling and poor cardiac output.
- Prioritize Hypotheses: Highest priority is unstable ventricular dysrhythmia with imminent arrest risk.
- Generate Solutions: Activate emergency response, prepare rhythm-directed therapy, and support oxygenation/perfusion.
- Take Action: Implement protocol-based emergency management while continuously reassessing hemodynamics.
- Evaluate Outcomes: Rhythm stabilizes, perfusion improves, and symptoms resolve or decrease.
Related Concepts
- ecg-waveform-basics-and-12-lead-application - Accurate lead setup and waveform fundamentals reduce misclassification.
- heart-failure - Clinical syndrome that determines rhythm urgency at bedside.
- cardiovascular-system - Conduction pathways and cardiac-cycle physiology that anchor ECG interpretation.
- cardiovascular-and-peripheral-vascular-nursing-assessment - Perfusion-first bedside cues that determine rhythm urgency.
Self-Check
- Which ECG intervals are priority measurements in a systematic rhythm interpretation sequence?
- Why does combining strip analysis with perfusion assessment improve triage safety?
- Which rhythm findings should trigger immediate emergency escalation even before full diagnostic workup?