Deep Vein Thrombosis
Key Points
- Deep vein thrombosis (DVT) is thrombus formation in deep veins, most commonly in the lower extremities.
- Virchow triad (venous stasis, endothelial injury, and hypercoagulability) explains major DVT risk pathways.
- Common assessment cues include unilateral leg swelling, warmth, tenderness, erythema, and dilated superficial veins.
- Homan sign is no longer a reliable diagnostic criterion and should not guide decision making.
- Suspected pulmonary embolism (PE) is time-critical and requires immediate escalation.
- Acute PE support prioritizes oxygenation, upright positioning, and rapid escalation to higher-acuity monitoring when unstable.
- Pregnancy and the postpartum period increase venous thromboembolism risk and require pregnancy-specific anticoagulation planning.
- Core management includes anticoagulation, mobility and compression strategies when appropriate, and prevention teaching for recurrence reduction.
Pathophysiology
DVT develops when blood flow in deep venous channels becomes slow or disrupted, clotting activity rises, and local endothelial integrity is compromised. The resulting thrombus can partially or completely obstruct venous return and cause local inflammation, edema, and pain.
Virchow triad provides a practical framework for bedside reasoning:
- Venous stasis: immobility, prolonged sitting, postoperative recovery, heart failure, and reduced calf-muscle pump activity.
- Endothelial injury: trauma, surgery, vascular instrumentation, and inflammation.
- Hypercoagulability: inherited thrombophilia, malignancy, pregnancy/postpartum state, hormone therapy, dehydration, and smoking-associated vascular risk.
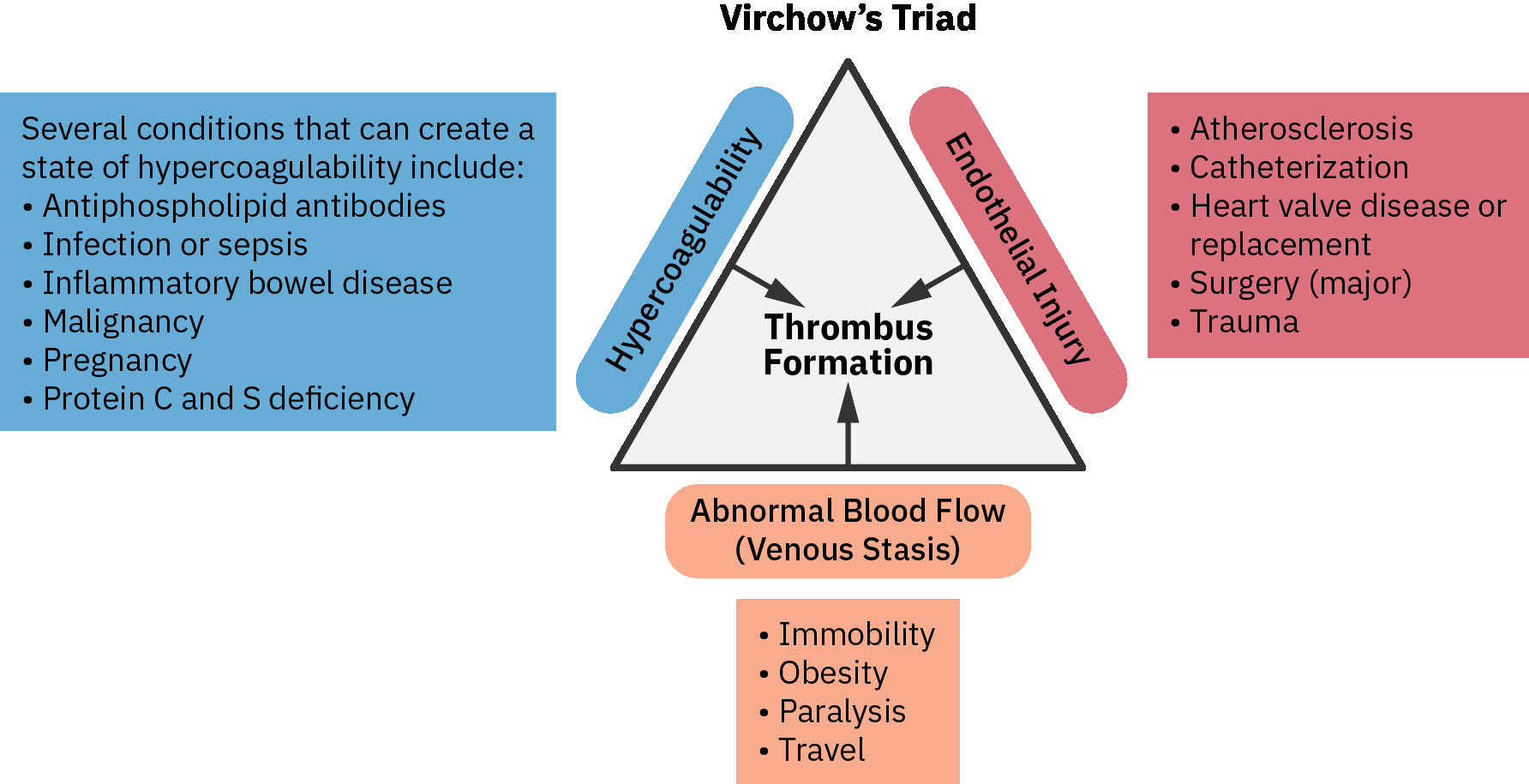 Illustration reference: OpenStax Pharmacology Ch.20.1.
Illustration reference: OpenStax Pharmacology Ch.20.1.
Thrombus fragments can embolize to the pulmonary circulation and cause PE, which can rapidly worsen oxygenation and hemodynamics. Untreated DVT can progress to PE and become immediately life-threatening when embolic obstruction is not rapidly treated.
Inherited Thrombophilia Cues
- Factor V Leiden (F5 mutation): Produces activated-protein-C resistance and increases venous clot recurrence risk.
- Prothrombin G20210A (F2 mutation): Raises prothrombin levels and amplifies coagulation-cascade activity.
- Unprovoked or recurrent DVT, especially with family clustering, should raise suspicion for inherited thrombophilia and may prompt genetic-risk evaluation.
- Risk expression can increase with estrogen exposure (oral contraceptives or hormone therapy) and pregnancy/postpartum physiology.
Nursing Assessment
NCLEX Focus
Prioritize early recognition of unilateral venous findings and immediate escalation for any suspected PE indicators.
- Assess unilateral lower-extremity swelling, warmth, tenderness, erythema, and calf or thigh discomfort.
- Treat unilateral/asymmetric edema as a high-value thrombotic cue when compared with bilateral or gravity-dependent edema patterns.
- Compare bilateral limb size, color, temperature, and focal calf tenderness when screening for lower-extremity DVT.
- Assess symptom onset context (recent surgery, immobility, long travel, trauma, malignancy treatment, pregnancy/postpartum status).
- Assess for inherited thrombosis cues: personal/family history of recurrent VTE, early-age unprovoked clotting events, and estrogen-associated event timing.
- Assess for asymmetric limb circumference and progression of edema burden over time.
- Assess for PE warning cues: sudden dyspnea, pleuritic chest pain, tachypnea, tachycardia, anxiety/restlessness, hemoptysis, syncope, and oxygen desaturation.
- Recognize PE-risk amplification when prior PE/DVT history, recent surgery/prolonged bedrest, active cancer/chemotherapy exposure, inherited clotting mutations, or prothrombotic medication exposure are present.
- Avoid using Homan sign as a diagnostic rule because it lacks reliability and may delay evidence-based escalation.
Diagnostic Testing Cues
- Anticipate D-dimer testing as a rule-out support tool in lower-risk contexts; elevated values are nonspecific.
- A D-dimer level below about 0.50 can support exclusion in low-suspicion pathways; elevated values alone do not confirm VTE.
- In pregnancy and postpartum assessment pathways, a low D-dimer level can support exclusion, but elevated D-dimer values do not confirm DVT.
- Anticipate duplex venous ultrasound as the primary noninvasive DVT imaging test.
- Anticipate CT pulmonary angiography when PE is suspected and clinical urgency is high.
- Trend coagulation labs (PTT, PT, INR) and bleeding-risk-relevant studies according to the ordered anticoagulation pathway.
Nursing Interventions
- Escalate suspected DVT promptly for diagnostic confirmation and treatment initiation.
- Escalate suspected PE immediately as an emergency condition.
- In suspected/confirmed DVT, avoid rubbing or massaging the affected extremity to reduce embolization risk.
- Perform focused neurovascular reassessment (6 Ps and capillary refill trends) at least each shift and more frequently after clot-directed procedures.
- In acute high-risk DVT phases, follow provider protocol for temporary activity restriction or bed rest before progressive mobilization.
- Administer and monitor ordered anticoagulants with focused bleeding surveillance and medication-safety teaching.
- First-line acute VTE treatment commonly uses IV heparin infusion, subcutaneous LMWH, or DOAC pathways when clinically appropriate.
- In pregnancy and postpartum DVT pathways, anticipate LMWH as the common first-line anticoagulation when not contraindicated.
- Avoid warfarin and most oral anticoagulants during pregnancy; reassess labor and delivery timing carefully because peripartum anticoagulation increases bleeding risk.
- When anticoagulation is contraindicated in pregnancy/postpartum DVT, prepare for invasive prophylaxis pathways such as IVC filter placement.
- For acute PE respiratory burden, support oxygenation and use High Fowler positioning to improve ventilation.
- Significant PE burden may require step-down/ICU care with telemetry and frequent hemodynamic monitoring.
- Coordinate dysrhythmia surveillance and treatment because atrial rhythm disorders can coexist with thromboembolic risk pathways.
- Reinforce safe mobility, hydration, and calf-muscle activation to reduce stasis risk once clinically appropriate.
- Implement and teach prescribed compression strategies and limb-care practices per provider guidance.
- Use postoperative SCD prophylaxis as ordered, but do not apply SCDs when unilateral DVT signs are present until nurse/provider reassessment.
- Coordinate peri-procedural monitoring when invasive options (for example, IVC filter placement, thrombectomy, or embolectomy) are used.
- Reinforce expected treatment duration planning (commonly at least about 3 months for confirmed pregnancy/postpartum DVT) and strict follow-up adherence.
- Provide recurrence-prevention education at transitions of care, including medication adherence, follow-up reliability, and red-flag symptom reporting.
- Counsel on combined thrombotic risk from smoking plus estrogen exposure (oral contraceptives or hormone-replacement therapy) and discuss safer alternatives when indicated.
- In inherited thrombophilia pathways (for example, Factor V Leiden with recurrent events), reinforce that extended or lifelong anticoagulation may be required per specialist plan.
- In long-term therapy planning, align regimen choice with monitoring burden, administration practicality, and cost/access factors to improve adherence.
Evaluation of Outcomes
- Trend respiratory recovery markers: reduced dyspnea, improving respiratory rate, and oxygen saturation normalization.
- Trend local thrombotic symptom improvement: reduced unilateral pain, edema, warmth, and erythema.
- Reassess mobility trajectory and tolerance as acute stabilization progresses.
- Evaluate ongoing bleeding/infection risk while on anticoagulation and after invasive interventions.
- Confirm the patient can explain personal risk factors and demonstrates adherence intent for anticoagulation and follow-up.
Pulmonary Embolism Escalation
New dyspnea, pleuritic chest pain, oxygen desaturation, syncope, or hemodynamic instability in a patient with possible DVT should be treated as an immediate emergency.
Pharmacology
| Drug Class | Examples | Key Nursing Considerations |
|---|---|---|
| anticoagulants | heparin, LMWH, warfarin, DOAC classes | Prevent thrombus extension and recurrence; monitor bleeding signs and ordered labs. |
| pregnancy-adapted anticoagulation pathway | LMWH-first approach during pregnancy/postpartum contexts | In pregnancy, avoid warfarin and many oral agents; postpartum agent choice should align with lactation goals and specialist guidance. |
| thrombolytic therapy | class-based PE/DVT rescue contexts | Used selectively in severe clot burden or hemodynamic compromise; monitor bleeding risk. |
| analgesic adjunct use | context-based pain control | Support comfort while preserving ongoing reassessment of perfusion and respiratory status. |
Clinical Judgment Application
Clinical Scenario
A postoperative patient develops unilateral calf swelling, warmth, and tenderness, then reports sudden pleuritic chest pain and shortness of breath.
- Recognize Cues: New unilateral venous findings plus acute respiratory symptoms indicate possible DVT with PE progression.
- Analyze Cues: The immediate threat is cardiopulmonary instability from embolic obstruction.
- Prioritize Hypotheses: PE is the highest-priority hypothesis requiring emergency escalation.
- Generate Solutions: Activate urgent response, support oxygenation, and prepare for definitive imaging and anticoagulation/thrombus-directed treatment.
- Take Action: Communicate concise trend data and implement ordered emergency interventions without delay.
- Evaluate Outcomes: Oxygenation and hemodynamic trends stabilize, and definitive treatment pathway is initiated.
Related Concepts
- peripheral-vascular-system-and-insufficiency-patterns - Venous insufficiency and thrombotic disease share risk pathways and lower-extremity assessment patterns.
- cardiovascular-and-peripheral-vascular-nursing-assessment - Structured vascular assessment supports early recognition and escalation.
- immobility-complications - Immobility is a major venous stasis driver and recurrence risk amplifier.
- postoperative-pacu-priorities-and-complication-surveillance - Postoperative surveillance is critical for early thromboembolic detection.
- emergency-situations-and-rapid-response - Suspected PE requires immediate emergency-level response.
- peripartum-cardiomyopathy - Pregnancy and postpartum cardiopulmonary symptoms require overlap assessment for thromboembolic causes.
Self-Check
- Which findings support likely DVT versus nonthrombotic leg discomfort?
- Which symptom cluster should trigger immediate escalation for possible PE?
- How does Virchow triad guide prevention teaching for high-risk patients?