Peripheral Vascular System and Insufficiency Patterns
Key Points
- The peripheral vascular system includes arteries, veins, arterioles, venules, and capillary networks that maintain tissue perfusion.
- The peripheral vascular system supports whole-body homeostasis by distributing oxygen/nutrients and removing metabolic waste.
- Arteriosclerosis refers to arterial wall thickening and stiffening, while atherosclerosis refers to inflammatory plaque buildup (often LDL-rich) within arterial walls.
- Arterial insufficiency and venous insufficiency present with distinct pain, skin, pulse, and edema patterns.
- Intermittent claudication is a key cue for peripheral arterial disease (PAD) and should trigger focused vascular follow-up.
- Acute limb perfusion decline is screened with the 6 P findings: pain, paralysis, paresthesia, pulselessness, pallor, and poikilothermia.
- PAD with persistent ischemia can progress to nonhealing wounds, amputation risk, and increased myocardial infarction and stroke risk.
- Chronic venous insufficiency reflects ineffective lower-extremity venous return with dependent pooling that commonly worsens over the day.
- Venous-insufficiency burden is higher in older adults, people with obesity, and females.
- PAD burden is high globally and rises with older age, smoking exposure, diabetes, and coexisting cardiovascular disease.
Pathophysiology
Peripheral vessels distribute oxygen, nutrients, hormones, and heat-regulation capacity to tissues while returning deoxygenated blood and metabolic waste to central circulation. Flow dynamics depend on vessel tone, valve function, and pressure gradients.
Systemic flow follows an ordered vascular path: aorta → arteries → arterioles → capillaries → venules → veins. This sequence frames bedside interpretation of where perfusion loss or venous return failure is most likely occurring.
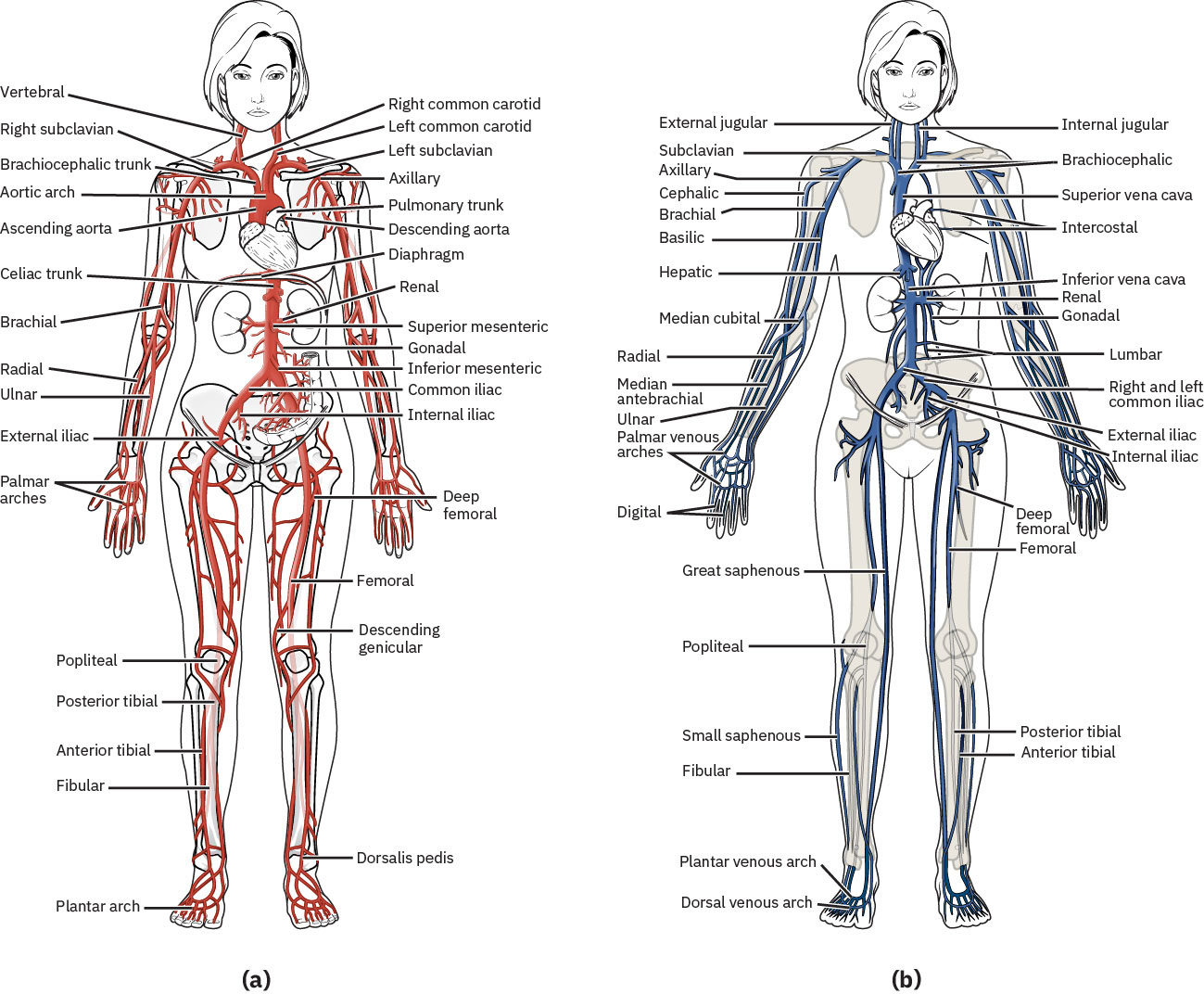 Illustration reference: OpenStax Clinical Nursing Skills Ch.24.2.
Illustration reference: OpenStax Clinical Nursing Skills Ch.24.2.
Peripheral arterial disease (PAD) most often affects lower-extremity arteries, where progressive atherosclerotic narrowing reduces oxygenated blood delivery during exertion and can progress to ischemic tissue injury. Carotid-artery stenosis is a high-yield occlusive vascular pattern because cerebral hypoperfusion and embolic risk increase stroke probability; clinically significant carotid narrowing is also a marker for broader systemic arterial disease. Both males and females over age 60 are affected by PAD, and coexisting CAD, aortic disease, or carotid disease should heighten suspicion for peripheral-arterial involvement.
Occlusive processes (such as plaque or thrombus) and functional dysregulation (abnormal vasoconstriction or vasodilation) can both reduce effective perfusion and produce ischemic injury. Arteriosclerosis and atherosclerosis commonly coexist; arterial stiffening reduces vascular adaptability, and plaque progression further narrows lumen diameter and worsens flow. When carotid disease is identified, concurrent aortic and renal-artery disease burden should be considered during risk stratification and follow-up planning.
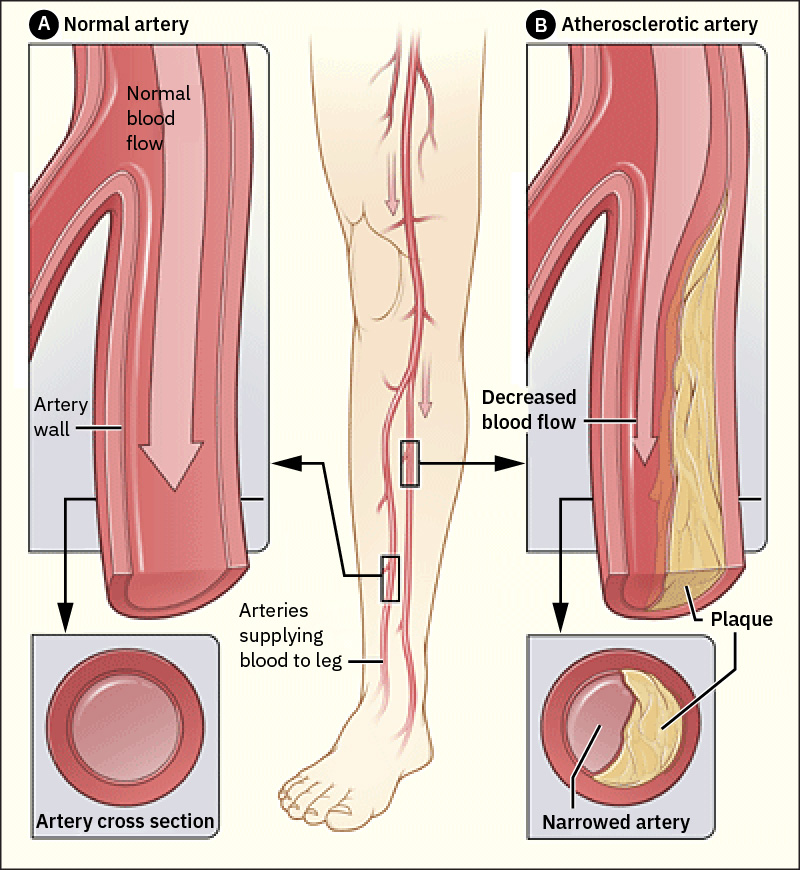 Illustration reference: OpenStax Clinical Nursing Skills Ch.24.2.
Illustration reference: OpenStax Clinical Nursing Skills Ch.24.2.
Peripheral vascular dysfunction also affects other care domains, including wound-healing trajectory, perioperative risk, and chronic disease management plans.
Arteries are smooth-muscle, elastic vessels that expand during systole and recoil during diastole; this pressure-wave transmission is what is palpated as the peripheral pulse. Arteries also support nutrient/hormone delivery and thermoregulation in addition to oxygen transport.
Arterioles are a major resistance site and are tightly controlled by sympathetic signaling and vasoactive mediators. Catecholamine effects and circulating/local mediators (for example angiotensin II and prostaglandins) alter arteriolar diameter and therefore tissue perfusion distribution.
Veins are thin-walled, low-pressure, high-capacitance vessels that depend on skeletal-muscle contraction and respiratory pressure changes to move blood toward the heart. Intraluminal venous valves help maintain one-way forward flow and limit backward pooling. When valve competence declines, venous reflux can drive retrograde pooling in lower-extremity circulation and accelerate chronic venous-insufficiency symptom progression.
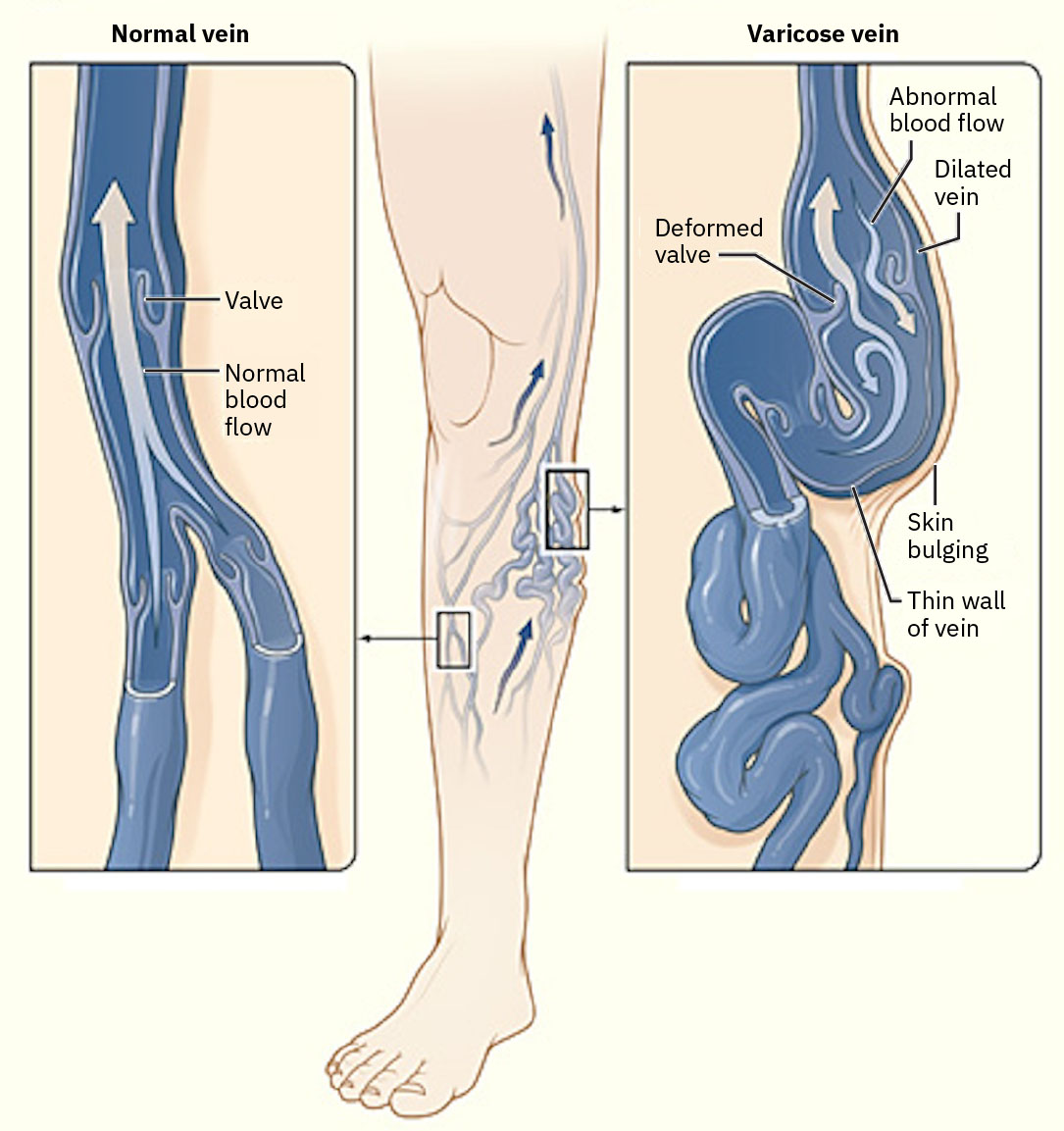 Illustration reference: OpenStax Medical-Surgical Nursing Ch.12.7.
Illustration reference: OpenStax Medical-Surgical Nursing Ch.12.7.
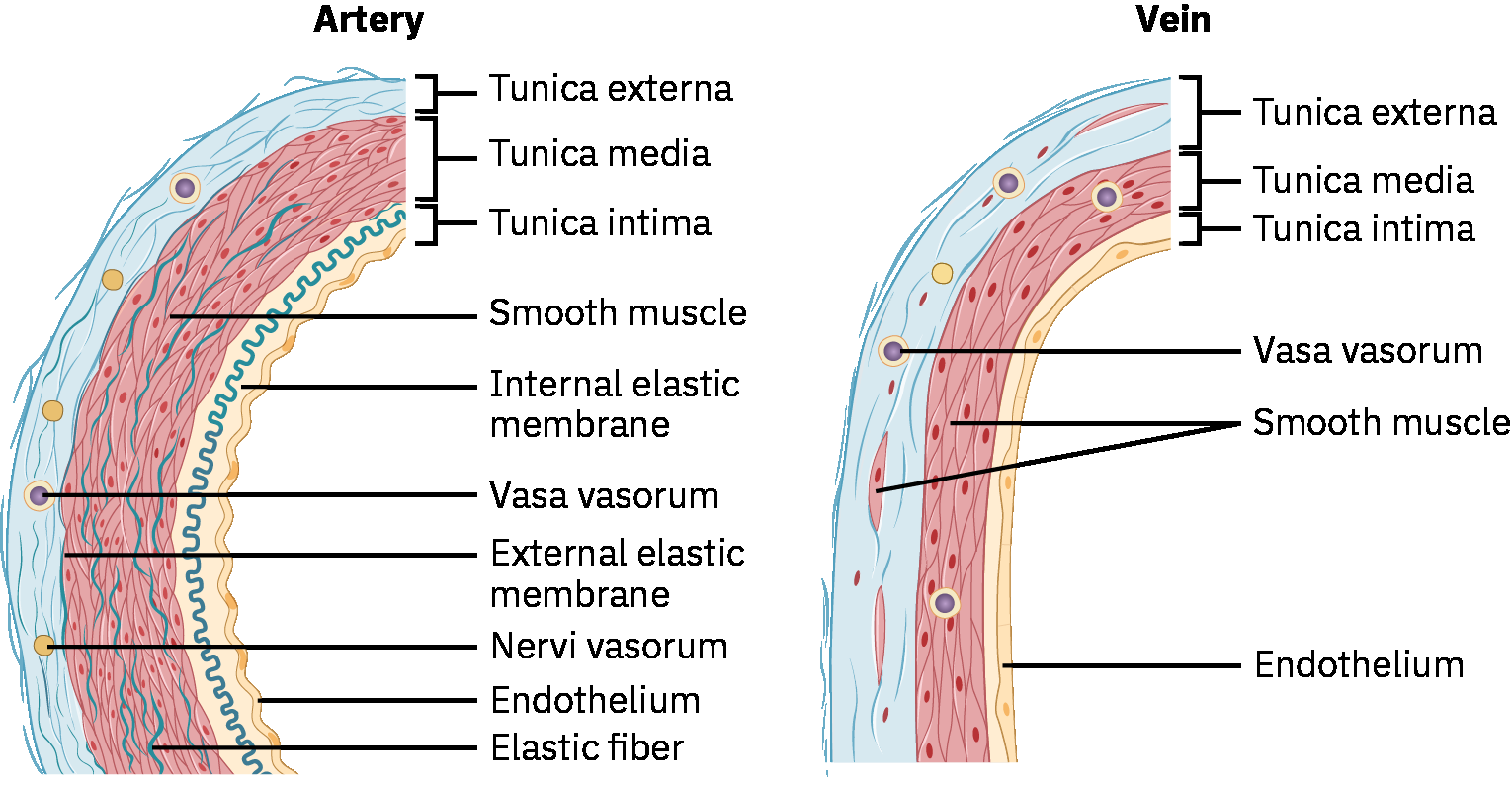 Illustration reference: OpenStax Pharmacology Ch.18.1.
Illustration reference: OpenStax Pharmacology Ch.18.1.
Venules provide the transition from capillary exchange to venous return. Postcapillary venules are highly reactive to inflammation and permit plasma-protein and leukocyte exit during inflammatory or infectious responses.
Pulmonary circulation is an important exception to usual oxygen-content patterns: the pulmonary artery carries deoxygenated blood to the lungs, and the pulmonary veins return oxygenated blood to the heart.
Classification
- Vessel classes: Arteries/arterioles for forward oxygenated flow, capillaries for exchange, and venules/veins for return flow.
- Disorder classes: Occlusive disorders versus functional vascular disorders.
- Insufficiency pattern classes: Arterial insufficiency and venous insufficiency.
- Common occlusive examples: Atherosclerosis, Buerger disease, carotid artery disease, DVT, and lymphedema-related outflow obstruction patterns.
- Common functional examples: Chronic venous insufficiency, Raynaud phenomenon, and varicose veins.
- Common peripheral-assessment disorder groups: Venous stasis dermatitis, vasculitis, PAD, and mixed venous-arterial insufficiency.
Nursing Assessment
NCLEX Focus
Compare bilateral lower-extremity findings and trend exertional pain patterns rather than relying on a single isolated cue.
- Assess exertional calf/thigh/buttock pain relieved by rest (intermittent claudication pattern).
- Assess temperature, color, edema distribution, skin texture, and ulcer location patterns.
- Assess for broader peripheral abnormality clusters including edema, cellulitis-like changes, temperature asymmetry, color change, reduced pulses, and ulceration.
- If peripheral findings coexist with systemic inflammatory symptoms (fatigue, fever, diffuse aches, appetite/weight loss), consider vasculitis-pattern escalation.
- Assess peripheral pulse quality and symmetry with focused attention to diminished unilateral findings.
- Assess for acute severe ischemia using the 6 P findings and escalate rapidly when present.
- Assess new exertional unilateral calf/thigh/buttock pain relieved by rest as claudication-pattern ischemia and trend severity/distance-to-onset over time.
- Clarify whether exertional leg pain is intermittent and rest-relieved versus persistent at rest, because progression toward rest pain suggests worsening ischemia.
- Assess risk factors such as smoking history, hypertension, dyslipidemia, systemic inflammation, older age, diabetes, prior arterial injury/trauma, and activity limitation.
- Include lifestyle-pattern screening for tobacco exposure, saturated-fat dominant diet, and physical inactivity because these factors accelerate plaque-mediated vascular progression.
- Assess venous-insufficiency contributors such as prior DVT, varicose veins, obesity, pregnancy history, and prolonged sitting or standing exposure.
- Assess for cross-system manifestations of reduced perfusion, including angina, TIA/stroke-like symptoms, reduced urine output, delayed wound healing, and vision changes.
- Assess for trophic skin changes such as hair loss on lower extremities, brittle or thickened nails, and nonhealing toe/foot ulcers.
- Assess for arterial-insufficiency findings including cool temperature, pale color, exertional pain, and ischemic-ulcer patterns; monitor closely for black necrotic tissue as a limb-threat cue.
- Assess for arterial skin-color pattern changes (pallor with elevation and dusky/red dependent color) and capillary refill delayed beyond three seconds.
- Assess for venous-pattern findings including progressive ankle/lower-leg edema, leg heaviness or aching, itching, dark/discolored skin, thick brown stasis changes, dry or scaly texture, tortuous superficial veins, and weeping venous ulcer development in severe disease.
- Venous-insufficiency discomfort commonly worsens with prolonged standing and may ease with rest/elevation; include this positional pattern in symptom triage.
- In chronic venous disease, document nail changes (for example thickened, discolored, or raised nail-bed appearance) alongside skin/hair findings.
- Differentiate thermal patterns: cold/clammy lower-extremity skin supports arterial insufficiency, whereas warm ankles with edema support venous insufficiency.
- Differentiate ulcer patterns: arterial ulcers are often painful, distal (toes/feet), round/smooth, dry or black-eschar dominant, and associated with weak/absent pulses; venous ulcers are often medial-ankle/low-leg, irregular with drainage/slough, and associated with edema and preserved pulses.
- In lower-leg ulcer triage, note location cues: venous ulcers often cluster in the medial calf/ankle “gaiter area,” while arterial ulcers are more common at toes/ankle pressure points.
- Venous-stasis ulcers commonly appear along medial or lateral distal lower-leg regions and can be shallow, irregular, brown-yellow periwound lesions that are minimally painful.
- Yellow periwound tissue in venous-ulcer beds can indicate necrotic/slough burden and should be tracked in wound-status documentation.
- Map pain location to likely arterial territory when possible (for example thigh/buttock/aortoiliac distribution, calf/femoropopliteal distribution, foot/tibial-peroneal distribution).
- Treat edema as a sign rather than a standalone diagnosis; asymmetric unilateral edema should raise concern for local venous thrombosis.
Diagnostic Testing Cues
- Review lipid profile and inflammatory markers (for example, C-reactive protein) in overall atherosclerotic-risk interpretation.
- Anticipate Doppler ultrasound to identify arterial narrowing and flow reduction in affected limbs.
- Anticipate segmental pressure measurements to localize arterial narrowing by comparing pressures across thigh, calf, and ankle levels.
- Anticipate ankle-brachial index (ABI) testing; lower ABI values support reduced lower-extremity arterial perfusion.
- Exercise ABI protocols may compare arm-leg pressures at rest and after graded treadmill walking; postactivity pressure decline with claudication symptoms supports flow-limitation diagnosis.
- Anticipate carotid ultrasound when carotid flow limitation is suspected.
- In carotid-disease pathways, duplex velocity findings with stenosis greater than about 50% often trigger pharmacologic or procedural treatment planning.
- Carotid stenosis less than about 50% is often managed with medical monitoring, while higher-grade disease is evaluated for revascularization candidacy.
- Anticipate coronary angiography or coronary CT angiography when coronary plaque burden/stricture assessment is needed.
- Anticipate venous Doppler/ultrasound flow studies to evaluate venous directionality and reflux patterns; anticipate venography with contrast when more detailed localization of restricted venous segments is required.
- Anticipate ankle-brachial index (ABI) workflow comparing brachial pressure to dorsalis pedis/posterior tibial ankle pressures; accuracy depends on correct cuff/probe technique.
- For disease-burden stratification, trend lipid elevation and hemoglobin A1C patterns because hyperlipidemia and diabetes burden commonly coexist with PAD/PVD progression.
Nursing Interventions
- Escalate acute ischemic or thromboembolic concern signs immediately.
- Reinforce risk-factor modification including tobacco cessation, activity progression, and follow-up adherence.
- Use structured secondary-prevention targets when ordered in advanced vascular disease (for example LDL below about 70 mg/dL, A1C below about 7%, and controlled blood-pressure trends).
- Teach prescribed walking therapy progression (walk to pain threshold, rest, then resume) to support collateral circulation development.
- Coordinate ordered vascular diagnostics and structured reassessment for symptom progression.
- Reinforce heart-healthy nutrition, blood-pressure control, lipid management, weight optimization, and diabetes blood-glucose control.
- Teach patients which new or worsening symptoms require immediate provider notification.
- Teach limb-protection strategies: avoid constrictive clothing and leg crossing; guide leg positioning based on perfusion and edema status (including dependent positioning for arterial flow support and avoiding elevation above heart level when edema/perfusion concerns coexist).
- Reinforce daily foot and skin inspection with prompt reporting of injury or nonhealing breakdown.
- After revascularization procedures, monitor surgical site integrity and distal pulses; report decreased perfusion, possible bypass compromise, or suspected bypass rupture immediately.
- In carotid disease pathways, CEA candidacy commonly includes symptomatic stenosis around 50-99% and selected asymptomatic severe stenosis around 70-99% per vascular-team criteria.
- Reinforce venous-return strategies: consistent use of correctly fitted graduated compression stockings, scheduled leg elevation above heart level, calf-muscle activation exercises, and avoidance of prolonged immobility.
- For suspected arterial-ulcer patterns, avoid compression until ABI or equivalent perfusion testing confirms safe arterial inflow.
- For chronic venous insufficiency, coach frequent position changes and avoidance of prolonged standing; when swelling worsens, reinforce pillow-supported leg elevation and hydration to reduce pooling risk.
- Coordinate evaluation for procedural venous interventions (for example, sclerotherapy or vein ligation/stripping) when conservative symptom-control measures are insufficient.
- In painful varicose-vein disease despite conservative care, escalate for wound-care or vascular-specialist referral and discuss procedural options (for example venous stripping) per team plan.
- For venous stasis dermatitis patterns, reinforce limb elevation, progressive muscle activation, and prescribed static/stocking compression plans while differentiating chronic stasis change from acute bacterial cellulitis.
- For PAD patterns, reinforce supervised exercise progression, smoking cessation, lipid/risk-factor control, wound-care adherence, and routine foot inspection.
- In symptomatic claudication phases, pace ambulation to individual tolerance and avoid prolonged overexertion while preserving structured walking-program goals.
- For arterial-ulcer care, coordinate ordered wound dressings and debridement pathways and monitor for infection progression.
- In mixed venous-arterial insufficiency, use ABI-guided planning, monitor closely for wound infection, and coordinate revascularization/wound-specialist pathways as ordered.
- In severe arterial insufficiency not controlled medically, anticipate angiography with possible endovascular repair or arterial-bypass referral.
Limb-Threatening Progression
Untreated progressive insufficiency can lead to critical ischemia, tissue necrosis, debridement/amputation requirement, and loss of limb function.
Pharmacology
| Drug Class | Examples | Key Nursing Considerations |
|---|---|---|
| coronary-artery-disease (antilipemics) | Statin therapy contexts | Lowers LDL burden and reduces plaque progression risk. |
| antiplatelets | Aspirin-class therapy contexts | Reduces thrombosis risk in atherosclerotic arterial disease. |
| antihypertensives | Blood-pressure control contexts | Reduces arterial strain and downstream ischemic complications. |
| anticoagulants | Anticoagulant therapy contexts | Use focused bleeding surveillance and monitor perfusion changes. |
| vasodilators | Vasodilator therapy contexts | Monitor blood pressure response and symptom relief with perfusion trends. |
| cholesterol absorption inhibitors | Ezetimibe-class contexts | Supports additional lipid reduction when LDL targets are not met. |
| thrombolytic therapy | Acute limb clot contexts | Reserve for acute obstruction protocols with close bleeding and perfusion monitoring. |
Clinical Judgment Application
Clinical Scenario
A patient reports calf cramping after short walking distance that resolves with rest; right foot pulse is weaker and cooler than left.
- Recognize Cues: Exertional unilateral pain with pulse and temperature asymmetry.
- Analyze Cues: Pattern is consistent with peripheral arterial insufficiency.
- Prioritize Hypotheses: Immediate priority is preventing progression to critical ischemia.
- Generate Solutions: Initiate focused vascular follow-up and reinforce risk-reduction plan.
- Take Action: Escalate findings and document bilateral comparison clearly.
- Evaluate Outcomes: Diagnostic workup and treatment plan are initiated without delay, and symptom progression does not advance to critical limb ischemia.
Related Concepts
- cardiovascular-system - Central cardiac function and peripheral flow are tightly linked.
- impaired-skin-and-tissue-integrity - Links perfusion-pattern findings to tissue-injury and healing consequences.
- hypertension-assessment-and-management - Chronic pressure burden and vascular injury progression frequently overlap.
- capillary-filtration - Microvascular exchange determines tissue-level delivery and clearance.
- fluid-volume-overload-hypervolemia - Volume status influences edema burden and vascular findings.
- cardiovascular-and-peripheral-vascular-nursing-assessment - Assessment sequence and escalation thresholds for bedside care.
Self-Check
- Which features best distinguish arterial from venous insufficiency at bedside?
- Why is bilateral pulse and skin comparison essential in peripheral vascular assessment?
- When should intermittent claudication symptoms trigger urgent follow-up escalation?