Preterm Newborn
Key Points
- A preterm newborn is born before 37 weeks and has risk severity strongly linked to gestational age.
- In the US, preterm birth affects about 1 in 10 infants and disparity burden remains higher in some racial and ethnic groups.
- Major morbidity domains are respiratory immaturity, thermoregulation failure, infection risk, neurologic injury, and feeding instability.
- Preterm birth is a leading contributor to infant death and long-term neurodevelopmental disability risk.
- Early surveillance and protocolized NICU care reduce mortality and long-term disability.
- Family education and ongoing developmental follow-up are essential components of preterm care.
- Early-course complications include RDS, fluid-electrolyte instability, PDA/hypotension, NEC, IVH, thermoregulation failure, metabolic instability, and sepsis risk.
- Long-term follow-up priorities include neurodevelopment, vision/hearing, chronic lung disease, feeding, and behavioral-health outcomes.
Pathophysiology
Prematurity interrupts organ maturation across pulmonary, neurologic, gastrointestinal, and immune systems. Immature lungs, fragile vasculature, poor thermal reserve, and limited metabolic buffering drive many high-risk complications.
Complications often cluster: respiratory instability increases infection and neurologic injury risk, while feeding intolerance and illness worsen growth and developmental trajectory.
Because rapid organ maturation occurs in late gestation (especially lung, brain, and liver development), earlier delivery substantially increases short-term and long-term morbidity risk.
Respiratory distress syndrome (RDS) is strongly linked to surfactant deficiency and alveolar collapse in infants born before about 34 weeks. In severe disease, prolonged oxygen and ventilatory support can contribute to chronic lung injury patterns such as bronchopulmonary dysplasia.
Classification
- Late preterm: 34 to 36 weeks.
- Moderately preterm: 32 to 34 weeks.
- Very preterm: Less than 32 weeks.
- Extremely preterm: 28 weeks or earlier.
- Moderate-to-late preterm (alternate grouping): 32 to less than 37 weeks.
Nursing Assessment
NCLEX Focus
Priority questions emphasize trend-based assessment: a subtle change in breathing, temperature, perfusion, or feeding can indicate severe deterioration.
- Assess respiratory pattern and support needs for acute-respiratory-distress-syndrome, apnea of prematurity, MAS overlap, PPHN, and chronic lung risk.
- Assess for RDS signs: tachypnea, retractions, nasal flaring, expiratory grunting, and central cyanosis (distinguish from benign acrocyanosis).
- Assess respiratory diagnostics and trajectory, including chest X-ray evidence of low lung volumes with diffuse ground-glass appearance in RDS.
- Assess for pulmonary-transition failure (PPHN) with severe hypoxemia, escalating oxygen need, and poor response to routine transition support.
- Assess apnea patterns: apnea episodes of about 15 to 20 seconds or longer, or shorter pauses with bradycardia/desaturation, are concerning for apnea of prematurity.
- Consider gestational-age prevalence context for apnea of prematurity: it is common in infants below 28 weeks and much less frequent after about 34 weeks.
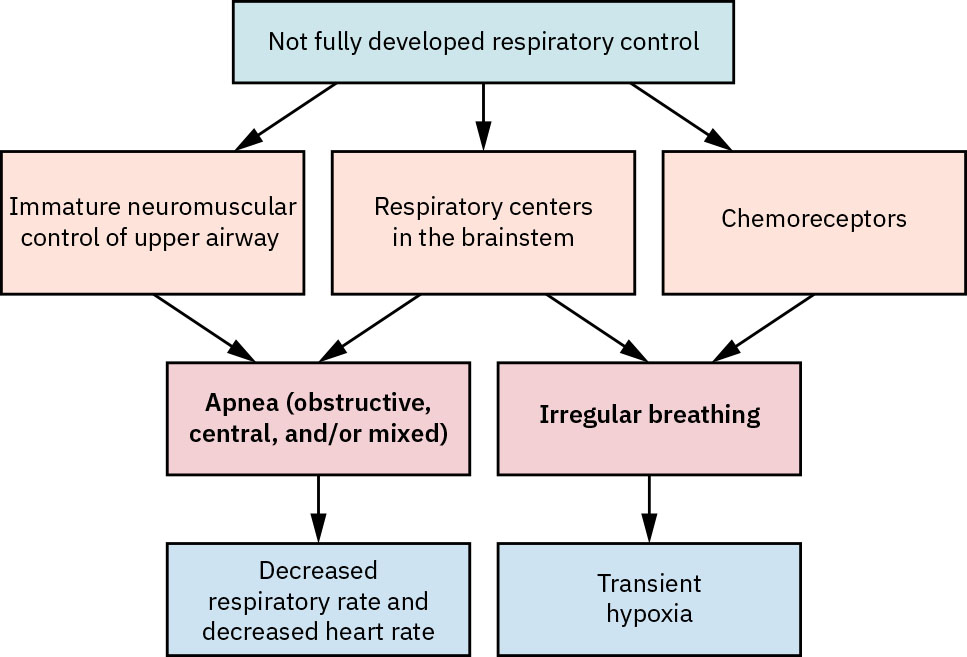 Illustration reference: OpenStax Maternal-Newborn Nursing Ch.25.4.
Illustration reference: OpenStax Maternal-Newborn Nursing Ch.25.4.
- Assess for TTN pattern in early transition (tachypnea, mild retractions/grunting, delayed lung-fluid clearance risk after cesarean or rapid delivery).
- Assess MAS overlap when meconium exposure is present, including distress with hyperinflation features, worsening oxygenation, and air-leak risk.
- Assess physical prematurity cues such as low body mass with proportionally larger head, decreased subcutaneous fat with sharper facial contour, and lanugo.
- Assess thermal stability, glucose risk, and feeding tolerance progression.
- Assess fluid-electrolyte status with strict I&O, diaper-weight urine tracking, daily weight trends, sodium trends, and fontanel/perfusion findings for dehydration or overhydration.
- Assess for neurologic complications (IVH, seizures, hypoxic-ischemic injury) using exam and ordered imaging trends.
- For IVH risk (especially very/extremely preterm infants), assess for apnea, hypotonia, weak suck, lethargy, and obtain ordered cranial-ultrasound surveillance (early life and near term-equivalent age).
- For IVH imaging surveillance, expect routine head ultrasound in infants born before about 30 weeks during the first 2 weeks and again near corrected term age.
- For neonatal seizures, assess subtle seizure cues (for example lip smacking or focal twitching) and recognize that EEG-only seizure activity can occur without obvious clinical signs.
- Assess infection risk and sepsis indicators with culture/lab pathway awareness.
- Differentiate sepsis timing context: early onset (first week), late onset (after early period to first month), and very late onset in prolonged NICU courses.
- Assess GI tolerance and early warning signs of Preterm Newborn.
- Assess NEC red flags: abrupt feeding intolerance, abdominal distention, bloody stool/diarrhea, vomiting, lethargy, temperature instability, and cardiorespiratory instability.
- Treat bilious (dark-green) emesis, rectal bleeding, and abrupt abdominal tenderness/distention as high-priority NEC escalation cues.
- Correlate suspected NEC with abdominal imaging findings such as intramural bowel gas (pneumatosis intestinalis) and trend serial films for progression.
- Recognize concurrent bacteremia risk in NEC and maintain heightened sepsis surveillance when NEC signs appear.
- Assess maternal and pregnancy risk-profile context (for example prior preterm birth, diabetes, hypertensive disorders, interpregnancy interval below about 18 months, substance exposure, infection burden, and limited prenatal care) to guide anticipatory surveillance.
- Include PPHN-risk context in respiratory assessment (for example maternal diabetes/obesity, advanced maternal age, in utero SSRI exposure, and meconium-exposure pathways).
- Assess for BPD trajectory in oxygen-dependent preterm infants, including ongoing oxygen need at 36 weeks’ postmenstrual age and failure of oxygen weaning.
- Assess for hypoxic-ischemic brain injury using structured neurologic exams and continuous EEG when encephalopathy or subclinical seizure risk is present.
- Assess longer-term complication risk during follow-up planning, including retinopathy risk with oxygen exposure, hearing loss, and developmental-delay surveillance.
- Assess skin integrity frequently because preterm skin is fragile and injury-prone.
Nursing Interventions
- Provide respiratory support escalation/de-escalation with continuous monitoring and frequent reassessment.
- Use stepwise RDS support: supplemental oxygen, CPAP, and escalation to intubation/mechanical ventilation when oxygenation remains inadequate.
- Anticipate RDS progression over the first 2 to 3 days of life, with expected improvement as endogenous surfactant production increases.
- Titrate oxygen and saturation targets carefully to balance hypoxemia prevention with hyperoxia complications (including retinopathy risk).
- In severe hypoxemia, coordinate escalation for pulmonary vasodilation/critical-care pathways (for example inhaled nitric oxide or ECMO candidacy in refractory PPHN).
- Use caffeine-based apnea management when indicated in prematurity-related respiratory-control instability.
- In meconium-exposure respiratory failure, avoid routine suctioning but support tracheal intubation/suction when airway obstruction is suspected.
- Maintain strict thermoregulation and glucose-support strategies.
- Regulate NICU environment (noise/light reduction) and cluster care to reduce stress burden and support sleep/growth.
- Implement infection-prevention bundles and timely antibiotic administration when sepsis is suspected.
- Before starting antibiotics for suspected sepsis, support timely culture collection from appropriate sites when clinically feasible.
- Use gentle skin-protection strategies, moisture barriers, and handling minimization to prevent skin breakdown.
- Optimize nutrition pathways (enteral/parenteral) and monitor growth trajectory closely.
- Include NEC-risk reduction practices, including close feeding-intolerance surveillance and breast-milk-forward feeding plans when feasible.
- When NEC is suspected, support bowel rest (NPO), IV fluids/TPN pathways, NG decompression, prescribed antibiotics, and serial abdominal reassessment.
- Prepare for surgical escalation when severe NEC or bowel necrosis/perforation is suspected.
- For IVH pathways, provide grade-severity monitoring, supportive care for anemia/hemodynamic needs, and neurosurgical escalation when posthemorrhagic hydrocephalus develops.
- For seizure pathways, support rapid cause-directed workup (for example hypoxia, hemorrhage, metabolic/electrolyte/infectious causes) and timely anticonvulsant treatment to reduce long-term neurologic injury.
- For moderate-to-severe hypoxic-ischemic encephalopathy, support therapeutic hypothermia initiation within about 6 hours of birth per protocol.
- Integrate developmental and bonding supports (including skin-to-skin and parent involvement in care) as physiologic status allows.
- Use neonatal pain-assessment workflows and treat pain proactively during procedures/care.
- Verify weight-based medication dosing and monitor adverse effects closely to prevent medication-error harm.
- Provide safe-sleep counseling before discharge because prematurity and low birth weight increase SIDS vulnerability.
- Prepare family for prolonged care course, high-acuity warning signs, and developmental follow-up needs.
- Integrate emotional-support and resource linkage for family stress and financial burden related to prolonged NICU care.
- Coordinate interprofessional care with neonatology, respiratory therapy, rehabilitation services, and case-management support for continuity across NICU and discharge phases.
- Reinforce nosocomial-infection prevention with strict hand hygiene, human-milk support (including donor-milk pathways), and antibiotic-stewardship workflows.
Rapid Deterioration Potential
Preterm infants can decompensate quickly from subtle respiratory, infectious, or neurologic changes; delayed escalation increases severe outcome risk.
Pharmacology
| Drug Class | Examples | Key Nursing Considerations |
|---|---|---|
| surfactants | Beractant context | Supports immature lungs and can reduce severity of respiratory distress. |
| methylxanthines | Caffeine citrate context | Used in apnea-of-prematurity pathways; monitor heart rate, feeding tolerance, and response trend. |
| Pulmonary vasodilators | Inhaled nitric oxide context | Used for selected severe PPHN pathways to reduce pulmonary vascular resistance. |
| chronic-lung-disease support agents | Diuretics, bronchodilator, inhaled corticosteroid contexts | Used in selected BPD pathways; monitor fluid balance, airway response, and adverse effects. |
| antibiotics | Ampicillin plus aminoglycoside context | Early empiric therapy for suspected neonatal sepsis (sepsis) while cultures are pending. |
| anticonvulsants (antiepileptics) | Levetiracetam context | Used for neonatal seizure control with close neurologic monitoring. |
| neonatal-seizure adjuncts | Phenobarbital, phenytoin, benzodiazepine contexts | Selected seizure pathways may require escalation beyond first-line antiepileptic strategy. |
| antenatal corticosteroids (maternal pathway) | Betamethasone, dexamethasone | If preterm delivery risk is high before about 34 weeks, maternal ACS helps improve newborn pulmonary readiness. |
Clinical Judgment Application
Clinical Scenario
A very preterm infant develops increasing tachypnea, recurrent desaturation, temperature instability, feeding intolerance, and intermittent apnea.
- Recognize Cues: Multi-system instability in a high-risk gestational-age infant.
- Analyze Cues: Possible overlap of respiratory immaturity, infection, and metabolic stress.
- Prioritize Hypotheses: Immediate priorities are oxygenation, perfusion, and sepsis exclusion.
- Generate Solutions: Escalate respiratory support, initiate sepsis workup/treatment pathway, optimize thermal and nutrition support.
- Take Action: Implement NICU protocol bundle with close serial reassessment.
- Evaluate Outcomes: Stabilized oxygenation, improved perfusion/temperature, and clearer diagnostic direction.
Related Concepts
- newborn-resuscitation - Many preterm infants require advanced transition support at birth.
- preterm-labor - Maternal preterm-labor management directly affects newborn prematurity risk and severity.
- physiological-adaptation-and-transition - Prematurity magnifies normal transition challenges.
- neutral-thermal-environment - Thermal instability is a major contributor to preterm morbidity.
- care-of-common-problems-in-the-newborn - Jaundice, hypoglycemia, and infection overlap with prematurity risk.
- discharge-planning-for-high-risk-newborns - Preterm discharge requires structured readiness and follow-up.
Self-Check
- Which complications are most strongly associated with decreasing gestational age?
- Why are respiratory, thermal, and nutritional interventions tightly linked in preterm care?
- Which subtle bedside findings should trigger immediate escalation in a preterm infant?