Neurological System
Key Points
- The neurological system processes sensory input, coordinates movement, and supports cognition and behavior.
- The CNS (brain and spinal cord) serves as interpretation and transmission control, and the PNS connects these centers to the periphery.
- ANS divisions (sympathetic and parasympathetic) provide complementary involuntary control for stress response and recovery.
- Neuronal signaling depends on neurotransmitters and adequate metabolic fuel, especially glucose.
- Brain tissue is highly perfusion dependent: glucose is not stored, and brief oxygen-delivery interruption can cause rapid neuronal injury.
- Neurotransmitters are synthesized from amino-acid building blocks, so adequate protein intake supports neurosensory function.
- Age-related slowing increases fall risk and functional vulnerability, requiring proactive safety support.
Pathophysiology
The central nervous system (brain and spinal cord) integrates sensory signals and generates adaptive motor and cognitive responses, while the peripheral nervous system carries cranial and spinal nerve communication between these centers and target tissues. Rapid electrochemical transmission allows reflexes, balance control, communication, and executive functions needed for daily living.
The brain has a high metabolic demand relative to body size and depends on continuous cerebral perfusion for oxygen and nutrient delivery. Because neurons do not store meaningful glucose reserves, sustained intake and circulation are required to support function; prolonged hypoperfusion or hypoxemia can trigger rapid toxic-metabolite accumulation and irreversible neuronal damage.
Neural control includes both automatic regulation (for example breathing and cardiovascular patterning) and conscious processing (sensation, learning, memory, and emotional response). Dysregulation can therefore produce combined physical and cognitive-behavioral dysfunction.
Neurologic performance can decline with age, metabolic instability, and progressive neurodegenerative disease. Slower signal transmission, sensory deficits, and impaired cognition increase risk for injury, communication breakdown, and loss of independence.
In pediatric clients, neurologic-development differences affect risk patterns: thermoregulation is less mature in newborns/infants (especially with head exposure), motor development is incomplete and increases falls, head-to-body proportion is larger, and cranial bones are thinner. Rapid brain growth in infancy also increases sensitivity to oxygen- and glucose-supply compromise.
Neurologic impairment can result from infection, trauma, degeneration, neoplasm, ischemic-flow disruption, structural defects, and autoimmune disease. Effects may be multisystem and include concurrent physical, cognitive, and psychosocial dysfunction.
Functional brain specialization supports integrated behavior. The cerebellum coordinates fine motor control, the hypothalamus contributes autonomic and endocrine homeostatic regulation, and the thalamus relays sensory and motor signals to cortical networks.
The cerebrum and cerebral cortex are the largest brain components and interact with the diencephalon, brain stem, and cerebellum as an integrated network. Cerebral hemispheric lateralization contributes to dominant-language/logical processing (commonly left) and creative-intuitive processing (commonly right), with the corpus callosum serving as a major interhemispheric communication pathway. The cortical surface is described by gyri and sulci, and the longitudinal fissure separates left and right hemispheres.
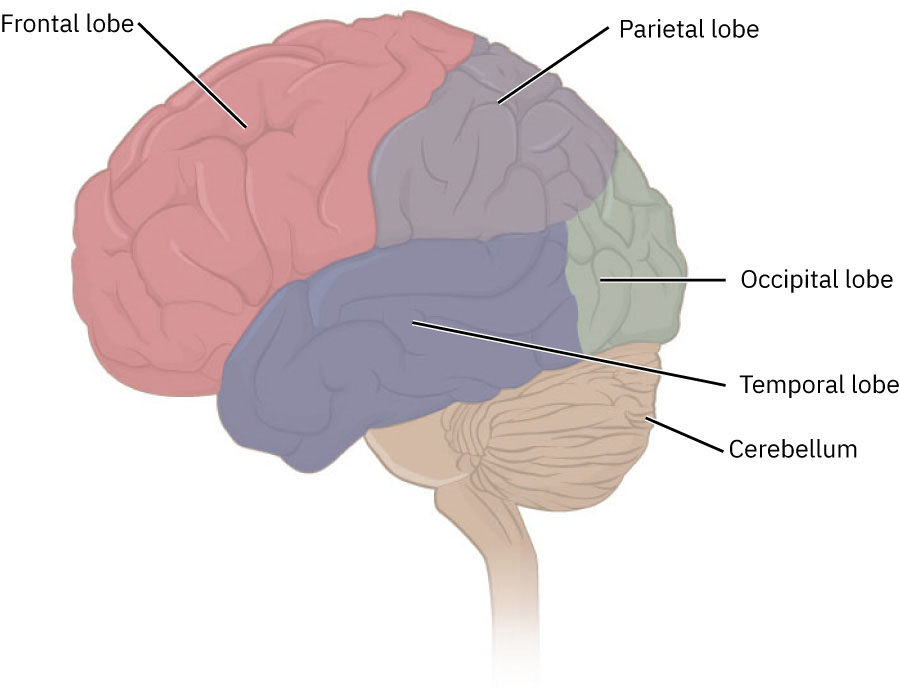 Illustration reference: OpenStax Clinical Nursing Skills Ch.26.1.
Illustration reference: OpenStax Clinical Nursing Skills Ch.26.1.
CNS protective and support structures include ventricles that produce cerebrospinal fluid and meninges (pia, arachnoid, and dura) that envelop neural tissue. Brain stem pathways support autonomic cardiopulmonary control and protective reflexes such as swallowing, coughing, and vomiting.
The brain stem (midbrain, pons, medulla) links higher cortical centers with lower pathways. Midbrain circuits support visual-auditory reflex integration; pons pathways contribute to cerebellar communication and respiration modulation; medullary centers regulate respiration, heart rate, blood pressure, swallowing, sneezing, and vomiting reflexes.
Cerebellar function extends beyond fine-motor coordination to postural sense and proprioceptive integration through continuous muscle-joint sensory feedback loops.
At the cellular level, neurons generate electrical impulses and chemical signaling, while glial cells provide structural and functional support. Axons transmit outgoing impulses, dendrites receive input, and synapses permit neurotransmitter-based communication across neural junctions; myelin sheaths from glial cells improve conduction efficiency.
Nutrition status influences neurologic recovery trajectories. Malnutrition, dysphagia-related intake delay, and inflammatory-catabolic states are associated with poorer outcomes in stroke, traumatic brain injury, and neurodegenerative conditions, while balanced macronutrient intake and targeted micronutrient sufficiency (for example B-complex vitamins, iron, magnesium, zinc, and iodine) support neurotransmission, myelin integrity, and cerebral metabolism.
Abnormal impulse transmission and neurotransmitter imbalance contribute to many neurologic and mental health disorders. Neurotransmission context helps explain why different disorders present with movement, mood, cognition, pain, or seizure changes.
Classification
- Core function domains: Sensory processing, motor control, cognition, and language.
- Core processing sequence: Sensation (input), integration (processing/interpretation), and response (voluntary or involuntary output).
- Core structural domains: CNS (brain and spinal cord) and PNS (cranial nerves and spinal nerves).
- Hemispheric control pattern: Left and right cerebral hemispheres communicate through callosal fibers and largely control contralateral body motor output.
- Cranial nerve organization: Twelve cranial nerves (I-XII) primarily support sensory and motor function of head and neck, with vagal influence extending into thoracic and upper abdominal organs.
- Cranial nerve modality model: Individual cranial nerves are sensory, motor, or mixed pathways; deficit interpretation should match modality.
- Cranial nerve modality distribution: Three cranial nerves are primarily sensory, five are primarily motor, and four are mixed sensory-motor pathways.
- Spinal nerve organization: Mixed sensory/motor spinal nerve pairs include C1-C8, T1-T12, L1-L5, S1-S5, and one coccygeal pair.
- Segmental innervation landmarks: C1-C5 supports upper-cervical/head-neck function, C3-C5 supports phrenic-diaphragmatic breathing, C5-T1 supports brachial plexus/upper-limb function, thoracic roots support trunk/abdominal wall pathways, L1-L5 supports lumbar plexus and anterior-limb sensory-motor pathways, and L4-S4 contributes to sacral plexus including gluteal/foot pathway context.
- Cell signaling architecture: Neuron cell body, axon, dendrites, synapse, and glial support.
- Synapse signaling types: Electrical synapses use direct cell-to-cell conduction; chemical synapses use neurotransmitter release across a synaptic cleft.
- Neurochemical transmission model: Neurotransmitters released into synapses bind specific neuroreceptors on target neurons, muscle fibers, or other effector tissues.
- Major neurotransmitter patterns:
- Acetylcholine dysregulation is associated with neuromuscular transmission disorders such as myasthenia-gravis.
- Dopamine deficiency is associated with parkinsons-disease and movement/cognition change.
- Serotonin imbalance is associated with mood and sleep dysregulation, including depression contexts.
- Reduced GABA inhibitory effect increases seizure vulnerability.
- Glutamate excess can contribute to excitotoxic neuronal injury and information-processing dysfunction.
- Major brain functional regions: Frontal lobe (movement and executive planning), parietal lobe (somatic sensation), temporal lobe (auditory and language comprehension), and occipital lobe (visual processing).
- Cortical lobe detail:
- Frontal lobe: Concentration, executive function, motor control, affect/personality/inhibition/judgment, expressive language (Broca area), and primary motor-strip coordination of contralateral voluntary movement.
- Parietal lobe: Sensory integration (touch, pressure, vibration), proprioceptive body-awareness mapping, and spatial/right-left orientation.
- Temporal lobe: Auditory processing, sound-language memory, and receptive language comprehension (Wernicke area).
- Occipital lobe: Visual interpretation/visual-memory processing plus visual-reflex and smooth-eye-movement coordination support.
- Additional core regions: Cerebellum (fine motor control), hypothalamus (autonomic and homeostatic regulation), and thalamus (sensory/motor relay).
- Diencephalon integration: Hypothalamus and thalamus coordinate autonomic relay, endocrine regulation, and sensory prioritization; hypothalamus supports thirst, sleep, hunger, temperature, stress, reproduction, and pituitary-driven hormone control.
- Basal-ganglia role: Deep cerebral nuclei that support fine motor control and movement coordination.
- Protective/support structures: Ventricles (CSF production), meninges (pia, arachnoid, dura), and clinically relevant meningeal spaces (epidural, subdural, subarachnoid).
- Brain stem organization: Midbrain (visual/auditory reflex and pathway relay), pons (bridge and respiration modulation), and medulla (vital reflex centers plus reticular-formation arousal/sleep-wake contribution).
- Spinal-cord organization: Extension of medulla to approximately L2 level with cervical/thoracic/lumbar/sacral regional outflow patterns.
- Cauda equina context: Distal lumbosacral nerve-root bundle forms as vertebral-column growth exceeds spinal-cord length in early development.
- Spinal gray-horn functions:
- Posterior horn (dorsal): Sensory information processing.
- Anterior horn (ventral): Somatic motor output to skeletal muscle.
- Lateral horn: Autonomic motor cell-body region in thoracic/upper-lumbar and sacral levels.
- Autonomic regulation domains: Hypothalamus-brain stem-spinal coordination of involuntary cardiopulmonary, vascular, gastrointestinal, glandular, and thermoregulatory functions.
- Autonomic divisions:
- Sympathetic division (“fight or flight”): Supports energy expenditure and stress response (for example mydriasis, bronchodilation, vasoconstriction in selected beds, increased heart rate/contractility, reduced GI motility/secretions).
- Parasympathetic division (“rest and digest”): Dominates nonstress conditions with opposing effects that support digestion, cardiac relaxation, and restorative homeostasis.
- Neural pathway organization:
- Ascending pathways carry sensory input from peripheral receptors through spinal relays to thalamic/cerebellar and cortical processing centers.
- Descending pathways deliver motor output to lower motor neurons for voluntary and involuntary movement control.
- Major descending tracts include corticospinal/corticobulbar systems for voluntary movement and vestibulo/reticulo/rubrospinal systems for posture, tone, and automatic motor modulation.
- Spinal injury localization patterns: Higher cervical injury can cause tetraplegia (quadriplegia), lower lesions more often cause paraplegia, and motor/sensory deficits may be dissociated.
- SCI sensory safety pattern: Paralysis in an extremity does not prove absent sensation; preserve pain-safety precautions until sensory status is explicitly assessed.
- Limbic contribution: Emotional behavior and memory integration that influence survival responses.
- Motor-response governance: Somatic pathways mediate intentional voluntary movement, whereas autonomic pathways mediate involuntary organ responses and reflex-associated visceral effects.
- Neuro-nutrition vulnerability pattern: Dysphagia, depression, movement disorder, gastroparesis, and cognitive impairment can reduce intake while neurologic illness increases metabolic demand.
- Age-related decline domains: Slower reaction time, sensory loss, and higher safety risk.
- Neurodevelopmental context: Chronic neurologic conditions include neurodevelopmental disorders (for example ASD and ADHD) that often coexist with anxiety or other mental-health concerns.
- Diagnostic and treatment complexity: Presentation can vary substantially across individuals, so persistent changing symptoms often require prolonged medication and longitudinal reassessment.
- Chronic condition contexts: Parkinson disease, ALS, dementia, seizure/stroke vulnerability.
Nursing Assessment
NCLEX Focus
Priority questions focus on rapid recognition of neurologic change and immediate escalation for safety-critical findings.
- Assess baseline cognition, orientation, communication, and behavior patterns.
- Observe for gait changes, tremor progression, coordination deficits, and mobility decline.
- Identify swallowing/speaking difficulty and signs of aspiration or respiratory compromise in progressive disease.
- Assess nutrition-risk cues alongside neurologic findings: intake decline, unintentional weight loss (for example >5% in 6 months or >10% in 1 year), and BMI/metabolic-syndrome burden.
- Assess dietary pattern factors that worsen cognitive/vascular risk (for example high saturated-fat/high-sugar processed intake, heavy alcohol use, and unsupervised supplement use).
- Interpret albumin/prealbumin trends with clinical context because inflammation, stress, or infection can lower values independent of intake.
- Report sudden confusion, unilateral weakness, seizure activity, or acute neurologic deterioration immediately.
Nursing Interventions
- Maintain safety environment and adaptive aids (glasses, hearing devices, mobility supports).
- Encourage cognitively stimulating activities and social interaction (for example board/card/word games, reminiscence activities) to support function.
- Encourage nutrition choices that support neurologic health, including adequate protein and omega-3/antioxidant-rich foods when appropriate.
- Prioritize whole-food neurologic-protective patterns (for example Mediterranean-style intake) and reduce refined sugars, highly processed foods, and excess saturated fat linked to cognitive decline risk.
- Trend glucose and sodium balance in high-risk neurologic clients because dysglycemia and dysnatremia can worsen cognition, seizure risk, and cerebral-function stability.
- Use structured, incremental nutrition counseling (for example assess-advise-agree-assist-arrange workflows) and coordinate early dietitian involvement when risk cues cluster.
- After acute neurologic events with unsafe swallow, prioritize early swallow evaluation and timely alternate nutrition planning (including enteral support when indicated) to avoid prolonged intake gaps.
- Allow extra time for ADLs and communication in slowed-movement or cognitive-limited clients.
- Provide emotional support to clients and families coping with progressive neurologic conditions.
Rapid Neurologic Deterioration Risk
Acute neurologic changes can signal stroke, seizure, or metabolic crisis and require immediate nurse notification.
Pharmacology
| Drug Class | Examples | Key Nursing Considerations |
|---|---|---|
| dopaminergic-therapy | Parkinson-management context | Monitor mobility/function response and report worsening gait or fall trends. |
| anticonvulsants | Seizure-management context | Report breakthrough events or postictal safety concerns promptly. |
Clinical Judgment Application
Clinical Scenario
A resident with Parkinson disease develops increased shuffling gait, delayed transfers, and near-falls over one week.
- Recognize Cues: Progressive movement slowing and rising injury risk.
- Analyze Cues: Neurologic decline is reducing safe mobility and ADL performance.
- Prioritize Hypotheses: Immediate priority is fall prevention and prompt reassessment of care plan.
- Generate Solutions: Increase supervision during transfers, optimize assistive-device access, and report trend.
- Take Action: Implement safety interventions and communicate objective changes.
- Evaluate Outcomes: Falls are prevented and mobility plan is adjusted.
Related Concepts
- caring-for-clients-with-dementia - Shared cognitive-support and behavior-safety strategies.
- fall-prevention - Neurologic decline significantly elevates fall risk.
- assisting-with-sensory-deficits - Adaptive-aid use improves safety and communication.
- endocrine-system - Glucose instability can acutely impair cognition and neurologic function.
- stroke - Time-sensitive emergency requiring rapid cue recognition.
Self-Check
- Which neurologic findings require immediate escalation versus routine reporting?
- How does sensory decline interact with neurologic fall risk?
- Which daily interventions best preserve function in progressive neurologic disease?