Neurological Physical Assessment and Red Flag Screening
Key Points
- Neurological assessment integrates subjective history with objective cognitive, cranial-nerve, motor, sensory, and coordination testing.
- Bilateral comparison and trend-based reassessment are essential for detecting acute neurologic deterioration.
- BEFAST findings, new aphasia, abrupt mental-status change, and focal weakness require immediate escalation.
- Early change in level of consciousness is a high-priority cue and should trigger structured reassessment (for example, Glasgow Coma Scale) and rapid escalation when worsening.
Pathophysiology
Neurological deficits can arise from structural injury, ischemia, infection, toxic-metabolic imbalance, neurodegeneration, or peripheral nerve dysfunction. Assessment findings vary by lesion location and progression speed.
Early bedside detection of subtle changes in language, movement, sensation, and coordination helps prevent delays in time-sensitive treatment.
Neurologic outcomes are also shaped by social and structural factors. Access barriers, bias, and socioeconomic exposures can delay diagnosis and specialist follow-up, increasing risk of advanced presentation and poorer functional outcomes.
Classification
- Subjective domains: Onset/timing of dizziness, vertigo, numbness, weakness, dysphagia, dysphasia, seizures, confusion, and recent injury.
- Objective domains: Mental status/LOC, pupils, cranial nerve exam, motor strength, sensory function, cerebellar tests, gait, balance, and deep tendon reflex patterns.
- Mental-status component domains: LOC/orientation, appearance, behavior, affect/mood, speech, and cognitive performance.
- Escalation domains: Suspected stroke, seizure emergencies, worsening consciousness, and progressive focal deficits.
- Equity/risk domains: Access barriers, health-literacy limits, and social determinants that can affect neurologic follow-up and treatment adherence.
- Screening-tool domains: GCS for LOC trend, NIHSS/BEFAST for acute stroke screening, and MMSE for cognitive impairment stratification.
- Abnormal-pattern domains: Altered mental status, altered language (aphasia), cranial-nerve deficits, sensory alteration (including peripheral neuropathy), and motor-coordination alteration.
- Exam-type domains:
- Routine bedside exam: RN-focused mental status/LOC, pupils, motor strength, sensation, and gait checks with GCS trend use when indicated.
- Comprehensive neurologic exam: Expanded cranial-nerve, cerebellar, deep tendon reflex, and MMSE-focused assessment, often in specialty evaluation contexts.
- Periodic reevaluation exam: Time-interval reassessment and documentation for acute neurologic injury or illness according to agency policy.
Nursing Assessment
NCLEX Focus
Prioritize airway, breathing, and acute stroke or seizure risk over lower-acuity chronic findings.
- Assess orientation, attention, behavior, and level of consciousness using standardized tools when indicated.
- Establish LOC/orientation baseline at admission and trend at least each shift or per acuity protocol; compare changes against that baseline.
- Use direct orientation prompts (name, place, day/date; if needed day-of-week/month/season) and document preserved versus impaired domains (for example oriented to self only).
- Select exam depth (routine vs comprehensive vs periodic reevaluation) based on care setting, current diagnosis, and active neurologic symptom burden.
- Obtain history for current neurologic symptoms, prior neurologic conditions, recent falls/head injury, and swallowing/speech concerns.
- For acute neurologic complaints, use PQRSTU-focused follow-up while preparing immediate escalation if active stroke-like findings are present.
- Ask explicitly about recent medication changes that may worsen dizziness or orthostatic symptoms, especially in older adults at fall risk.
- Review sedatives, analgesics, neuromuscular-blocking agents, alcohol, and substance use because these may confound neurologic exam interpretation.
- Trend level of consciousness using consistent tools (for example, Glasgow Coma Scale in acute care): 15 = best response, less than 8 = comatose range, 3 = unresponsive, and a drop of 2 or more points is clinically significant.
 Illustration reference: OpenStax Clinical Nursing Skills Ch.26.2.
Illustration reference: OpenStax Clinical Nursing Skills Ch.26.2.
- Use MMSE when cognitive screening is indicated and interpret by score bands (about 24-30 no major impairment, 18-23 mild impairment, less than 18 severe impairment) with clinical context.
- Differentiate altered mental-status patterns using history and trend: progressive dementia pattern, acute fluctuating delirium pattern, depressive cognitive slowing, and coma-level unresponsiveness.
- Assess cranial-nerve function with side-to-side comparison and pupil reactivity checks.
- Classify cranial-nerve findings by sensory, motor, or mixed modality when localizing deficits.
 Illustration reference: OpenStax Clinical Nursing Skills Ch.26.1.
Illustration reference: OpenStax Clinical Nursing Skills Ch.26.1.
- Include focused cranial-nerve screening tasks when indicated: CN I odor identification (eyes closed), CN II distance-vision check (for example Snellen with corrected-vision documentation), and combined CN III/IV/VI ocular tracking with PERRLA checks.
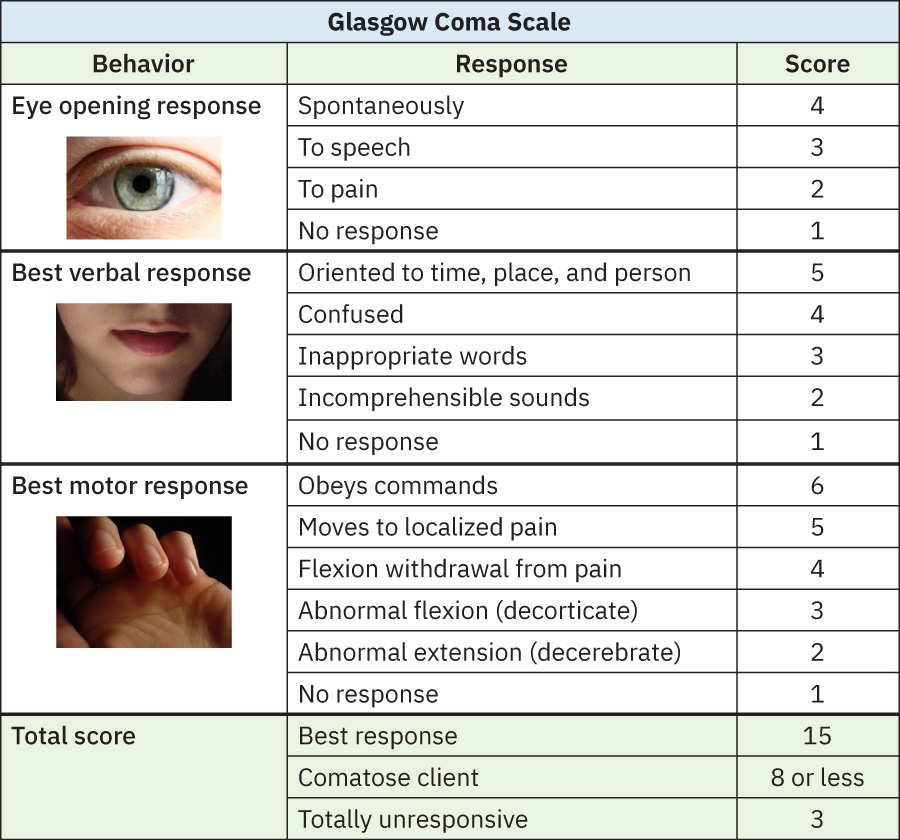 Illustration reference: OpenStax Clinical Nursing Skills Ch.26.2.
Illustration reference: OpenStax Clinical Nursing Skills Ch.26.2.
- During CN III/IV/VI screening, verify both direct and consensual pupillary light constriction and document asymmetric or sluggish responses as abnormal cues.
- For CN V assessment, test light-touch sensation across forehead/cheek/chin (eyes closed) and test motor strength by palpating temporalis/masseter contraction during teeth clenching and jaw opening/closing symmetry.
- In comatose-patient assessment contexts, include supervised corneal-reflex check (cotton-wisp blink response) when protocol requires advanced CN V screening.
- For CN VII screening, assess facial symmetry and muscle strength during smile, teeth-showing, bilateral eye closure, cheek puff, frown, and eyebrow raise tasks.
- Include taste screening when needed (for example separate salt/sweet/sour stimuli with eyes closed) and document correct identification and side-to-side differences.
- For CN VIII bedside hearing screening, test each ear separately with contralateral occlusion using low-volume letter-number whisper sequences; document repeat performance and pass/fail trend.
- For whispered-voice screening consistency, treat correct repetition of at least 3 out of 6 total letter-number items as a practical pass threshold.
- For CN IX-focused screening, assess palate and uvula midline symmetry while the patient phonates (“Ah”) and document asymmetrical rise or deviation.
- For CN X-focused screening, assess gag response (posterior pharynx stimulation) and swallowing coordination while monitoring aspiration-risk cues.
- For CN XI screening, test sternocleidomastoid and trapezius strength against resistance during head turn, forward head push, shoulder shrug, and shoulder retraction tasks; assess asymmetry or shoulder droop.
- For CN XII screening, assess tongue protrusion for midline deviation/unilateral weakness and use cheek-resisted tongue push for focal motor deficit detection.
- Use CN expected-vs-unexpected mapping during interpretation: CN I anosmia; CN II acuity/field loss; CN IV/VI gaze-limitation with diplopia/ptosis pattern; CN VII facial asymmetry or taste loss; CN IX/X absent gag or dysphagia/speech change; CN XI shoulder-turn weakness; CN XII tongue deviation from midline.
- During pupillary testing, compare both pupils for symmetry and reactivity (commonly about 2-5 mm range); assess for anisocoria, sluggish response, or nystagmus as abnormal cues needing escalation context.
- Check accommodation/convergence by shifting visual focus from near to far target and confirm expected pupillary constriction (near) with dilation (far).
- Assess memory and mood during interview (for example, agitation, anger, depression, euphoria) and whether affect is congruent with context.
- Assess cranial-nerve function with side-to-side comparison of face/neck symmetry; combine related testing when appropriate (for example, CN III/IV/VI eye-movement assessment).
- Assess motor strength and symmetry in upper and lower extremities with resistance testing.
- Use bilateral bedside strength maneuvers (simultaneous handgrip, forearm pull against resistance, seated lower-leg pull, hip flexion lift against resistance, ankle dorsiflexion/plantar-flexion resistance) and document side-to-side asymmetry.
- Interpret slight dominant-side strength advantage cautiously and escalate when asymmetry is clearly disproportionate or progressively worsening.
- Assess sensory function with eyes closed and cue-neutral prompts; test light touch and sharp/dull discrimination across major extremity regions and torso when indicated.
- For dermatomal screening, test representative distal skin regions first rather than every surface area during routine exams; expand to focused dermatome mapping when numbness, weakness, or focal deficits are present.
- If light touch is not detected and advanced assessment is required, escalate to supervised higher-intensity sensory checks (for example cold or pin-prick methods) per protocol.
- Grade motor strength on a 1-5 scale at shoulders, elbows, wrists, hips, knees, and ankles; assess tone via passive range of motion and identify hypotonia versus hypertonia.
- Observe for involuntary motor findings (tics, tremors, myoclonus) and document movement symmetry/coordination.
- Assess cerebellar function with gait/posture screening and focused coordination tests (finger-to-nose, heel-to-shin, rapid alternating hand movements).
- For cerebellar gait screening, assess 10-foot walk with pivot/return, heel-to-toe walk, tiptoe walk, and heel walk; abnormal cues include shuffling, jerky steps, balance loss, or arm-swing incoordination.
- Assess pronator drift for subtle upper-limb weakness: eyes closed, arms extended at shoulder level with palms up for 20-30 seconds; pronation or downward drift is abnormal.
- For gait-coordination screening, include short-distance ambulation with pivot/return, heel-to-toe walk, toe walk, and heel walk; document shuffling, jerky movement, arm-swing asymmetry, or balance loss.
- For finger-to-nose testing, have the patient extend both arms, close eyes as directed, and alternately touch nose tip right/left with return to extension; note overshoot, tremor, or side-dominant dysmetria.
- For heel-to-shin testing, place the patient supine and have each heel track from opposite knee toward ankle in a straight smooth line; heel drop-off or irregular tracking is abnormal.
- Use Romberg testing when appropriate (feet together, eyes closed) with close guarding for fall prevention; positive sway or loss of balance is an abnormal finding.
- In routine RN neurologic exams, auscultation is usually limited; when advanced assessments identify carotid bruits, treat them as potential cerebral blood-flow warning cues.
- Correlate reflex findings (for example, deep tendon and Babinski patterns) with overall neurologic context and provider-reported exam findings.
- Use reflex exam context appropriately: routine adult RN neuro checks may not include full tendon-reflex panels, but specialty or advanced assessments often do.
- When deep tendon reflexes are assessed, use standard 0-4+ grading (
2+expected):0absent,1+hypoactive,3+hyperactive without clonus,4+hyperactive with clonus; repeat after relaxation before confirming weak/absent responses. - When localization detail is needed, map reflexes by segment: brachioradialis (C5-C6), triceps (C6-C7), patellar (L2-L4), and plantar/Babinski pathway (L5-S1 context).
- Interpret Babinski by age context: extensor response can be expected in children under about 2 years, whereas persistent extensor response in older children/adults is abnormal.
- Assess cortical sensory processing with stereognosis by placing a familiar object in each hand (eyes closed) and asking for object identification.
- Add graphesthesia when cortical-sensory detail is needed by tracing a number or letter on the palm and asking for identification with eyes closed.
- Assess psychosocial response to neurologic illness and coping capacity because anxiety, role disruption, and function loss alter adherence and safety.
- Before language/communication testing, confirm baseline sensory supports (for example hearing aids or glasses) to avoid false-positive aphasia findings.
- Apply lifespan context during interpretation: pediatric fall/head-injury vulnerability and older-adult risks such as baseline atrophy, vascular change, and neurocognitive decline.
- For infants and children, account for developmental anatomy and safety (for example, anterior fontanel closure around 12-18 months, posterior fontanel closure around 2-3 months, immature thermoregulation, higher head-injury susceptibility, and motor-development-related fall risk).
- Include age-expected primitive-reflex checks in newborns and confirm expected disappearance windows during infancy.
- For older adults, interpret findings with expected aging changes (atrophy, reduced vascularization, neurotransmitter decline, and increased ischemic white-matter burden) while still escalating acute deviations from baseline.
- In older adults, account for expected sensory changes (reduced near-vision focus, smaller pupils/lens changes, hearing decline, and taste-smell-touch attenuation) while still treating abrupt changes as abnormal until proven otherwise.
- In older adults, also account for motor-aging patterns (reduced muscle mass/velocity, proprioceptive decline, osteoarthritis burden, and medication-related dizziness) when differentiating expected slowing from acute neurologic decline.
- Include contributory screening labs when clinically indicated (for example CBC, nutrition-related markers, BMP/electrolytes/glucose, ABG, toxicology, and infection cultures).
Nursing Interventions
- Activate urgent response for BEFAST-positive findings or sudden focal neurologic changes.
- Use NIHSS/BEFAST-style stroke screening prompts consistently and treat positive findings as time-critical.
- Perform repeat focused neurologic checks at ordered intervals and communicate trend changes promptly.
- Reduce injury risk with fall precautions, aspiration precautions, and communication support strategies.
- Implement environment-based injury prevention for neurologic deficits (for example clutter control, call-light access, supervised ambulation, and alarm/assistance strategies based on risk level).
- Document assessment tools used, baseline findings, and subsequent trend changes clearly so progression or recovery can be reliably compared over time.
Time-Sensitive Neurologic Decline
New unilateral weakness, speech changes, or declining consciousness can indicate rapidly evolving stroke or intracranial pathology.
Pharmacology
| Drug Class | Examples | Key Nursing Considerations |
|---|---|---|
| anticonvulsants | Levetiracetam-class contexts | Monitor breakthrough events and postictal recovery trends. |
| thrombolytics | Acute ischemic stroke contexts | Strict time-window and neurologic-monitoring protocols apply. |
Clinical Judgment Application
Clinical Scenario
A patient reports sudden dizziness and has facial asymmetry with new slurred speech during routine assessment.
- Recognize Cues: Acute focal deficits and communication change.
- Analyze Cues: Findings are highly concerning for cerebrovascular event.
- Prioritize Hypotheses: Immediate stroke-pathway activation is highest priority.
- Generate Solutions: Initiate emergency escalation and obtain focused neurologic baseline documentation.
- Take Action: Activate rapid response according to protocol and continue frequent reassessment.
- Evaluate Outcomes: Time-to-treatment is minimized and neurologic decline risk is reduced.
Related Concepts
- neurological-system - Core anatomy and function context for exam interpretation.
- neuromuscular-diagnostic-testing - Objective testing pathways after bedside screening.
- neurological-diagnostic-testing-and-nursing-considerations - Neuroimaging, CSF, electrophysiology, and procedural nursing-prep framework.
- assisting-with-sensory-deficits - Safety adaptation for persistent sensory deficits.
- documenting-and-reporting-data - Trend-based neurological documentation supports safe escalation.
Self-Check
- Which neurologic findings should trigger immediate BEFAST-based escalation?
- Why is bilateral comparison essential in cranial-nerve and motor testing?
- How does serial reassessment improve safety in suspected neurologic decline?