Unit-Dose Parenteral Medication Preparation (Vials Ampules and Reconstitution)
Key Points
- Parenteral medications bypass gastrointestinal absorption and first-pass metabolism, supporting faster and more predictable systemic effect when oral use is unsuitable.
- Safe preparation requires strict sterility, correct diluent selection, and accurate dose/volume calculation.
- Single-dose vials are discarded after one use; ampules require filter-needle technique.
- During ampule withdrawal, do not inject air into the ampule and do not expel aspirated air bubbles back into the ampule.
- Reconstitution must follow manufacturer instructions for diluent type, volume, concentration, and storage.
- Reconstitution instructions must always be verified from authoritative labeling/eMAR resources and never assumed.
- Reconstitution math should be based on labeled post-reconstitution concentration for administration dose calculations; do not substitute dry-vial amount, diluent-only volume, or estimated displacement volume unless labeling explicitly directs that method.
- Mixing medications in one syringe requires confirmed compatibility and route-specific volume limits.
- Multi-dose vial handling requires open-date and beyond-use-date labeling (commonly up to 28 days unless manufacturer labeling requires earlier discard).
- Needle selection must match route/site/body composition and ordered volume; higher gauge numbers indicate smaller needle diameters.
- Syringe choice should match both target volume and graduation precision (for example smaller-volume syringes provide finer calibration marks).
- Route-specific common selections include finer gauges for ID/SQ and relatively lower gauges for many IM preparations, with individualized adjustment for very thin/cachectic patients.
- Prefilled cartridges/injector devices can improve speed, dose accuracy, and contamination control when product integrity and route instructions are verified.
- Prefilled syringe air-management follows product-specific rules; some products (for example enoxaparin prefilled syringes) require retaining the manufacturer air bubble.
- Hazardous/cytotoxic medication reconstitution requires containment controls, PPE, and pharmacy-trained handling workflows.
- Total mixed volume must stay within route/site and age-specific tolerance limits to avoid pain, poor absorption, and local adverse events.
Equipment
- MAR and active provider order access
- Ordered medication containers (single-dose vial, multidose vial, ampule, or prefilled cartridge)
- Sterile syringe/needle options (including Luer-lock or Luer-slip connectors, and fixed-needle formats such as insulin/tuberculin syringes) plus filter needle for ampule withdrawal
- Needleless/blunt vial-access devices when available for sharps-risk reduction
- Approved diluent and manufacturer/package instructions
- Alcohol swabs, gloves, sharps container, and labeling/documentation tools
Procedure Steps
- Verify patient, order, route appropriateness (for example oral intolerance or need for non-oral onset profile), medication rights, concentration, expiration, and route-specific volume limits.
- Perform hand hygiene and prepare a clean, low-interruption workspace.
- Maintain injection-device sterility throughout setup: keep needles capped when not in use and avoid contaminating syringe plunger-shaft/Luer connection surfaces.
 Illustration reference: OpenStax Clinical Nursing Skills Ch.12.
Illustration reference: OpenStax Clinical Nursing Skills Ch.12. - Select syringe and needle size/connection style appropriate for solution characteristics and target volume (common syringe sizes span about 0.5-60 mL; many adult IV push doses are prepared in 3, 5, or 10 mL syringes). Needle gauge/length selection should reflect injection site, patient size/body composition, and dose volume; use Luer-lock when a secure twist-lock connection is needed and use Luer-slip/slip-tip formats when rapid attach/remove workflow is appropriate for the device context. After opening sterile packaging, handle the syringe by the barrel and keep tip/plunger sterile. Read syringe volume at the leading edge of plunger contact with solution, and match syringe size to required measurement precision (for example 1 mL syringes commonly marked by hundredths, 3 mL by tenths, many 5-12 mL by fifths/two-tenths, and large 60 mL syringes by whole-number marks).
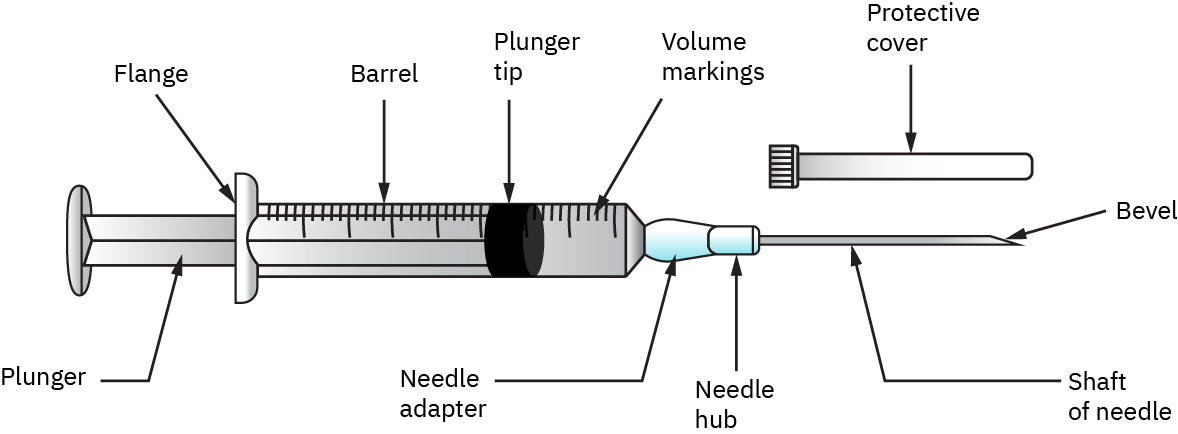 Illustration reference: OpenStax Clinical Nursing Skills Ch.12.
Illustration reference: OpenStax Clinical Nursing Skills Ch.12.
 Illustration reference: OpenStax Pharmacology Ch.2.3.
Illustration reference: OpenStax Pharmacology Ch.2.3. - For vials, remove dust cover (not sterile), scrub diaphragm with 70% isopropyl alcohol, inject air as indicated (commonly matching planned withdrawal volume), keep needle tip within medication during draw-up, and withdraw exact volume with sterile technique.
- If using multiple vials, use separate alcohol wipes per vial and maintain separate aseptic access events.
- For multidose vials, label open date and beyond-use date per policy/manufacturer guidance (commonly up to 28 days unless manufacturer labeling specifies earlier disposal).
- Keep multidose vials out of immediate patient-treatment areas unless policy requires point-of-care use with controlled handling.
- For ampules, tap fluid down, cleanse neck, snap away from body with gauze or ampule breaker, and withdraw using a filter needle or filter blunt device; do not inject air into the ampule, and if air bubbles are aspirated, clear them in the syringe rather than expelling them back into the ampule.
- Use medication withdrawn from ampules promptly and discard unused ampule medication per policy after the immediate preparation window.
- Replace filter needle with administration-appropriate needle/device before giving medication.
- If ampule break is irregular or glass shards are present in medication field, discard the ampule contents, clean glass safely, and restart with a new ampule.
- Before calculation and draw-up, confirm the ordered dose and target final concentration from vial directions.
- Verify reconstitution instructions from vial/insert/eMAR references (diluent type and volume, final concentration, storage conditions, and use-by timing) and never assume missing details.
- For powdered drugs, reconstitute using the exact diluent and volume specified by manufacturer/pharmacy.
- Mix as directed (gently rolling/swirl technique when indicated), confirm complete dissolution, verify final concentration, and confirm beyond-use/storage guidance.
- When dose calculation is required after reconstitution, use the labeled post-reconstitution concentration (mg/mL) as the dose-conversion basis rather than dry-powder amount per vial alone.
- For dual-compartment vial systems, activate the built-in diluent release per product design, gently swirl (do not shake), and verify no residual particles before withdrawal.
- If combining medications in one syringe, confirm compatibility first (electronic database, institutional compatibility chart, or pharmacist consult) and keep total volume within route/site limits (for example ID ⇐ 0.5 mL, SQ usually ⇐ 1.5 mL, and IM limits adjusted by site/age).
- When mixing medication from both vial and ampule, withdraw vial medication first, then use ampule-filter workflow, and finish with route-appropriate administration needle replacement.
- For insulin mixing, follow ordered insulin-specific sequencing (clear before cloudy): commonly inject air into cloudy NPH first without withdrawing, then inject air into regular and withdraw regular first, then return to NPH to withdraw remaining dose to the ordered total; select an insulin syringe that can accurately measure the combined dose and never mix incompatible insulin types (for example glargine/detemir with other insulins).
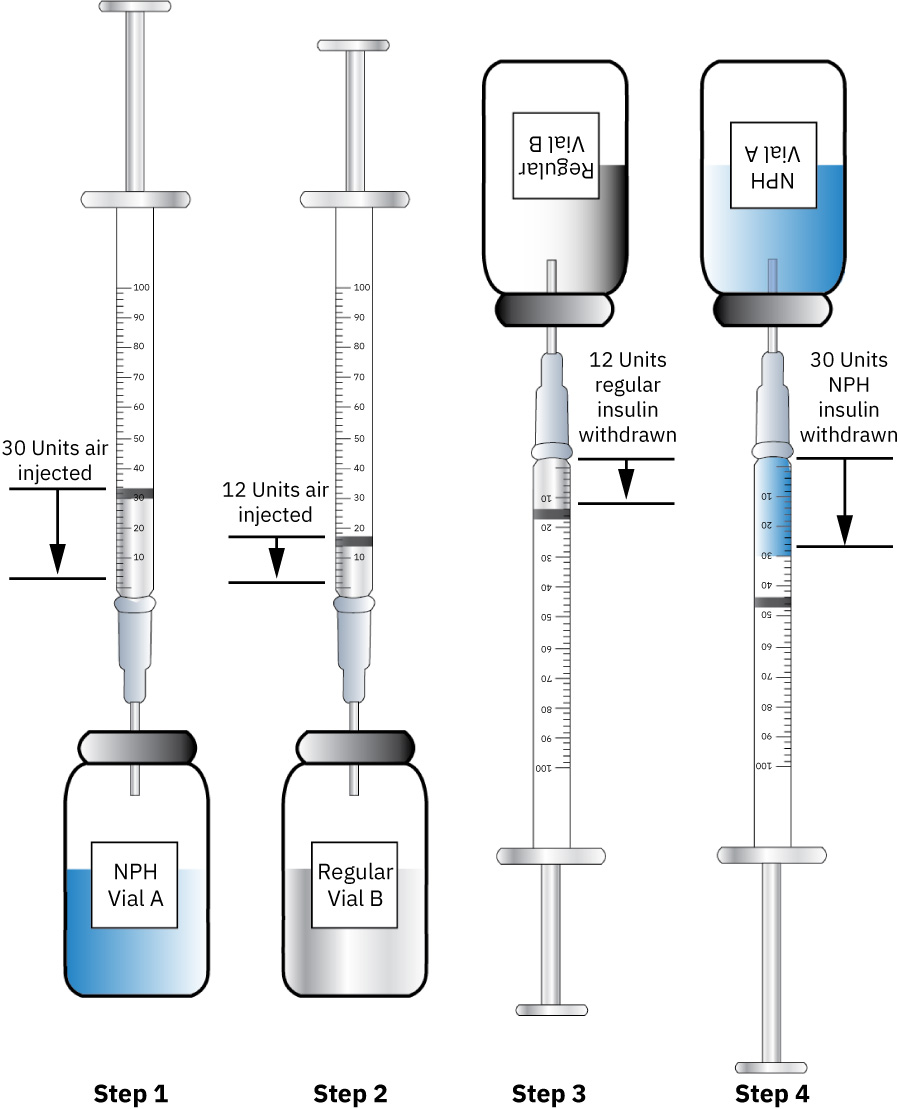 Illustration reference: OpenStax Clinical Nursing Skills Ch.12.
Illustration reference: OpenStax Clinical Nursing Skills Ch.12. - For prefilled syringes/cartridges, verify cartridge integrity, expiration, and volume against ordered dose; attach required plunger/needle component and manage air per product instructions (including retaining required air bubbles such as enoxaparin products when specified).
- For reusable single-dose cartridge holders, disinfect holder surfaces between uses; if withdrawing medication from cartridge with needle/syringe, do not inject air into the cartridge system.
- For auto-injector devices, confirm route/site suitability and follow product-specific activation sequence to deliver the preloaded single dose safely.
- For hazardous/cytotoxic reconstitution, use required containment controls and PPE and defer compounding to trained pharmacy professionals per policy.
- Prepare one medication syringe/device at a time; do not pre-label empty syringes.
- Label prepared medication per policy unless prepared at bedside for immediate administration; when preparer and administrator differ, include diluent type/volume, diluent expiration, and medication use-by time.
- Inspect final preparation for color/clarity/integrity changes and do not administer compromised products.
- Complete medication-right verification checkpoints according to policy during preparation workflow (for example selection, preparation, and preadministration checks).
- Dispose sharps immediately (including opened glass ampules in sharps container) and never reuse syringe/needle components.
- Document preparation details, dose, route, and response plan.
Common Errors
- Reusing single-dose vials → contamination and infection risk.
- Withdrawing from ampule without filter needle → particulate contamination risk.
- Wrong diluent or wrong reconstitution volume → unsafe concentration and dose errors.
- Mixing incompatible medications or excess volume → precipitation/microparticle formation, altered potency, toxicity, or poor absorption risk.
- Using small/high-gauge needles for highly viscous medication withdrawal → bend/break and needlestick risk.
- Needle size mismatch for route/body habitus/volume → pain, bruising, incomplete delivery, or local tissue injury risk.
- Omitting multidose vial date/BUD labeling or treating vial dust caps as sterile → preventable contamination risk.
- Contaminating syringe plunger-shaft or Luer connection surfaces → syringe-content contamination and infection risk.
- Using medication from a chipped/shard-contaminated ampule → embolic, phlebitic, granulomatous, or inflammatory injury risk.
- Injecting air into ampules or pushing aspirated bubbles back into ampules → dosing contamination and glass-particulate exposure risk.
- Removing required prefilled-syringe air bubbles (for example enoxaparin products) → incomplete dose delivery risk.
- Injecting air into cartridge systems not designed for air injection → stopper expulsion and medication loss/injury risk.
- Reconstituting hazardous medications outside required containment/PPE workflow → occupational exposure risk.
- Exceeding route/site volume limits when mixing medications → pain, leakage, and unreliable absorption risk.
- Insulin sequencing errors or mixing incompatible long-acting insulin formulations → unstable glycemic response risk.
- Prelabeling empty syringes or leaving prepared syringes unattended → wrong-product/wrong-patient risk.
- Using dry-vial amount, diluent volume, or estimated final-volume displacement in place of labeled post-reconstitution concentration → wrong-dose calculation risk.
Related
- intradermal-medication-administration - ID-specific limits and technique constraints after preparation.
- subcutaneous-medication-administration - SQ route volume/angle requirements.
- intramuscular-medication-administration - IM site and volume decisions for prepared doses.
- intravenous-medication-administration-safety - IV compatibility and monitoring requirements.
- sharps-disposal-and-needlestick-response - Required sharps safety during preparation and administration.