Hemostasis Coagulation and Fibrinolysis
Key Points
- The spleen and liver are key accessory organs for blood-cell handling and clotting regulation.
- Hemostasis progresses through vascular spasm, platelet plug formation, and coagulation with fibrin stabilization.
- Fibrinolysis limits clot burden by activating plasmin to break down fibrin and restore vessel flow.
- Warfarin slows clot-factor activation via vitamin K antagonism and requires routine INR monitoring.
- Blood-component therapy may be used for coagulation deficits: FFP for clotting-factor replacement and cryoprecipitate for fibrinogen-related disorders.
Pathophysiology
Hematologic physiology is supported by marrow production, accessory organs, and coordinated clot control. In marrow, erythropoietin-driven RBC production depends on adequate iron, vitamin B12, folate, copper, and cobalt. Thrombopoietin regulates platelet production, with a minority of platelets stored in splenic reserve and most circulating in blood. The spleen stores and filters blood elements, with white pulp supporting white-cell activity and red pulp serving as a reservoir for red cells and platelets. The liver contributes prothrombin and other clotting factors and metabolizes bilirubin from hemoglobin breakdown. Impaired hepatic bilirubin processing contributes to jaundice.
Hemostasis begins after vessel injury and proceeds in linked stages. First, local vascular smooth muscle contraction reduces blood loss (vascular spasm). Next, platelets adhere to exposed collagen, activate, and aggregate to form a temporary platelet plug. The coagulation cascade then activates clotting factors, generates thrombin, and converts fibrinogen to fibrin. Cross-linked fibrin reinforces the platelet plug, and subsequent clot retraction compacts the clot to support wound closure.
Coagulation is often described as extrinsic, intrinsic, and common pathways. The extrinsic (tissue-factor) pathway is typically faster and can complete in seconds after tissue injury, while the intrinsic pathway is more complex and often develops over minutes. Both pathways converge at Factor X activation in the common pathway, followed by conversion of prothrombin (Factor II) to thrombin and fibrinogen (Factor I) to fibrin, with Factor XIII stabilizing the clot.
Fibrinolysis balances clot formation by activating plasmin from plasminogen, often via tissue plasminogen activator (tPA). Plasmin degrades fibrin into smaller products, reducing clot size and helping re-establish blood flow. Dysregulation can shift risk toward thrombosis or hemorrhage.
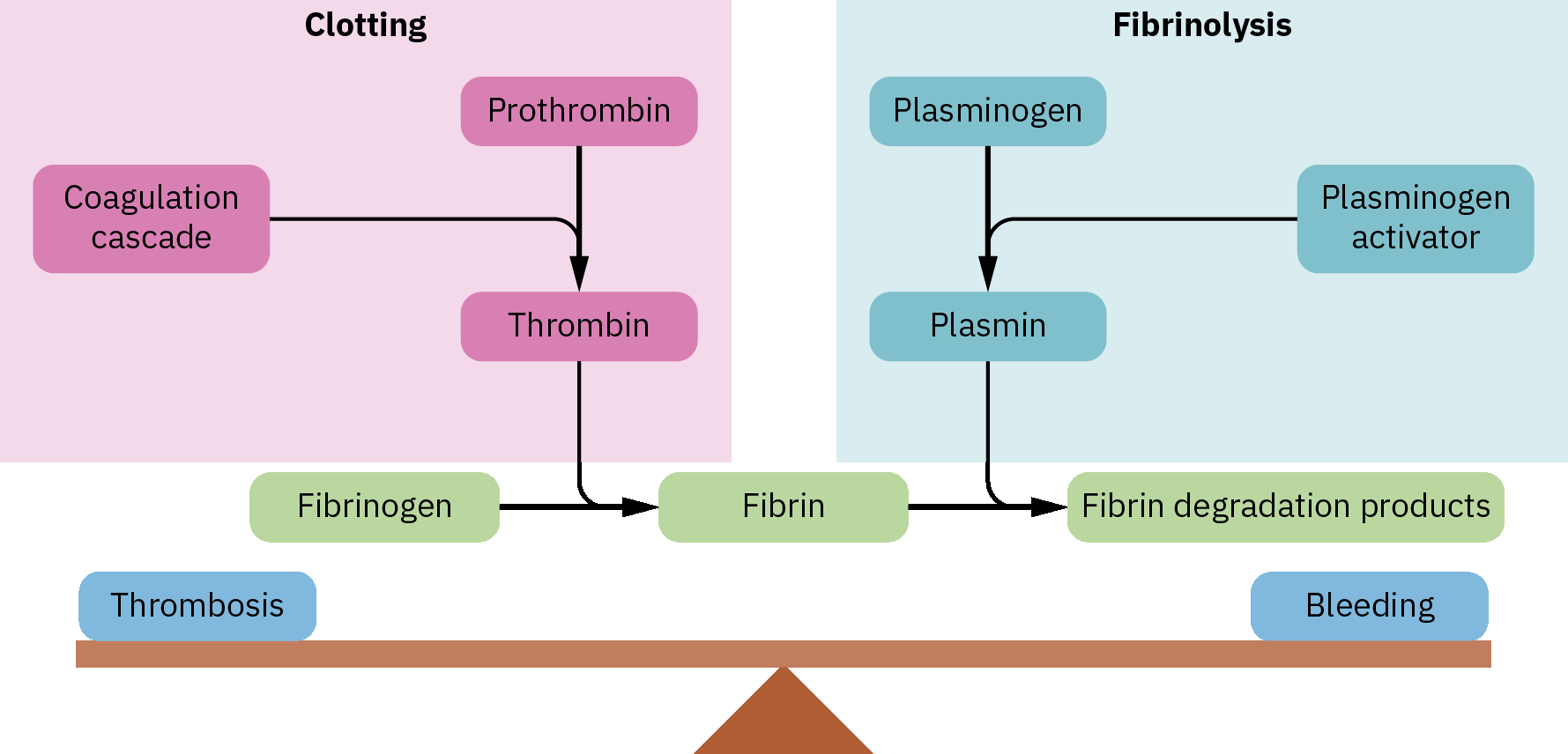 Illustration reference: OpenStax Pharmacology Ch.20.1.
Illustration reference: OpenStax Pharmacology Ch.20.1.
Physiologic anticoagulants limit clot extension beyond the injury site. Antithrombin opposes Factor X activity and thrombin generation, and endogenous heparin (released by basophils and mast-cell pathways) supports this anticoagulant effect. A thrombus that forms within an intact or minimally damaged vessel can partially obstruct flow or become fully occlusive in smaller vessels.
Clinically, thrombotic and bleeding disorders can be grouped as vascular, platelet, and coagulation-pattern disorders. Vascular abnormalities can alter lumen flow and promote stasis-related clotting, platelet quantity/quality defects alter primary hemostasis, and coagulation-pathway failure can occur with vitamin K deficiency or liver dysfunction. Acquired disorders such as disseminated intravascular coagulation create simultaneous clotting-factor consumption, microthrombosis, and bleeding risk.
Classification
- Primary hemostasis: Vascular spasm and platelet adhesion/activation/aggregation.
- Secondary hemostasis: Extrinsic and intrinsic cascade activation with convergence at the common pathway for thrombin and fibrin formation.
- Clot resolution: Fibrinolysis through plasmin-mediated fibrin degradation.
- Disorder-pattern framing: Vascular, platelet, and coagulation/fibrinolytic abnormalities may coexist and rapidly shift between thrombosis and bleeding phenotypes.
Nursing Assessment
NCLEX Focus
Priority items include identifying early bleeding versus thrombosis risk and recognizing unsafe anticoagulant intensity.
- Assess for active bleeding cues (gingival bleeding, epistaxis, melena, hematuria, oozing at puncture sites).
- Assess for possible thrombotic cues (new unilateral swelling, chest pain, acute neurologic deficits).
- Assess for recurrent or unprovoked venous thromboembolism patterns and family history that may indicate inherited thrombophilia.
- Review coagulation-related laboratory trends and INR values for clients on warfarin.
- Use baseline coagulation references for trend interpretation: INR about 0.8-1.2, PT about 11-13 seconds, and aPTT about 25-35 seconds (lab-dependent).
- For anticoagulant monitoring contexts, anticipate therapeutic prolongation targets (for example INR 2-3 with warfarin and PTT/aPTT about 1.5-2 times baseline when ordered).
- Correlate hepatic dysfunction signs (for example jaundice) with possible clotting-factor or bilirubin-processing effects.
- Review nutrition and medication history for vitamin K deficiency risk or vitamin K antagonism when coagulation instability is present.
- In splenic-trauma scenarios, assess urgently for hemorrhage risk and prepare rapid diagnostic escalation; after splenectomy, increase infection-surveillance vigilance.
Nursing Interventions
- Escalate suspected hemorrhage or thrombosis immediately because either can progress rapidly.
- Apply bleeding precautions and avoid unnecessary invasive procedures when coagulation status is unstable.
- Reinforce warfarin safety: consistent dietary vitamin K pattern, adherence, and scheduled INR follow-up.
- Support protocol-based response for acute clot events, including rapid team communication.
- Coordinate factor-replacement transfusion workflows when indicated (for example FFP in coagulation-factor loss/elevated INR contexts and cryoprecipitate for fibrinogen deficiency states such as dysfibrinogenemia or hypofibrinogenemia).
Anticoagulation Safety Risk
Excess anticoagulation increases hemorrhage risk, while insufficient anticoagulation permits disruptive clot formation.
Pharmacology
| Drug Class | Examples | Key Nursing Considerations |
|---|---|---|
| anticoagulants | warfarin | Monitor INR trend and bleeding risk; maintain consistent vitamin K intake pattern; understand VKOR inhibition as the core mechanism. |
| thrombolytics | thrombolytics (tissue-plasminogen-activator) | Used in selected acute occlusive events; monitor closely for bleeding complications. |
Clinical Judgment Application
Clinical Scenario
A client taking warfarin develops spontaneous gum bleeding and an INR above target during routine follow-up.
- Recognize Cues: New bleeding signs plus supratherapeutic anticoagulation indicator.
- Analyze Cues: Coagulation is excessively suppressed, increasing immediate hemorrhage risk.
- Prioritize Hypotheses: Highest priority is anticoagulation-related bleeding complication.
- Generate Solutions: Hold/escalate per protocol, increase monitoring, and review medication/diet factors.
- Take Action: Implement bleeding precautions and notify provider with current findings.
- Evaluate Outcomes: Bleeding resolves and INR returns to therapeutic range.
Related Concepts
- immune-system - Spleen white pulp links immune and hematologic function.
- abo-blood-group-compatibility-and-transfusion-safety - Blood-product safety intersects with coagulation risk states.
- blood-transfusion-verification-initiation-and-reaction-response - Procedure-level response for blood-related instability.
- wound-healing-phases-and-closure-intentions - Hemostasis is the first phase of tissue repair.
- hemophilia - Inherited factor deficiency as a high-yield bleeding-disorder application.
- Marrow failure and cytopenia patterns can change bleeding and infection vulnerability.
Self-Check
- What are the distinct clinical purposes of primary hemostasis, secondary hemostasis, and fibrinolysis?
- Why does warfarin therapy require INR monitoring and stable vitamin K intake patterns?
- Which assessment findings suggest emergent anticoagulation-related deterioration?