Chronic Obstructive Pulmonary Disease
Key Points
- COPD is a progressive inflammatory airflow limitation caused by chronic bronchitis, emphysema, or both.
- COPD burden remains high, with substantial hospitalization and mortality impact in the United States.
- Chronic hypoxemia and hypercapnia drive symptom burden and exacerbation risk.
- Exacerbations are the primary cause of COPD-related hospitalization and mortality acceleration.
- In COPD, low BMI and progressive muscle loss are poor-prognosis cues linked with diaphragmatic weakness, lower lung function, and increased mortality risk.
- Meal-related dyspnea can be reduced with small frequent meals, upright positioning, and energy-conserving pacing.
- In COPD with hypoxemia, antioxidant-dense patterns including lycopene-rich foods may support pulmonary nutrition goals.
- Progressive anorexia and malnutrition can weaken cough effectiveness, increase secretion retention, and raise pneumonia-risk burden.
- Nursing priorities focus on oxygenation monitoring, bronchodilator therapy, and self-management education.
Pathophysiology
COPD results from prolonged exposure to irritants (primarily cigarette smoke) causing airway inflammation, mucus hypersecretion (chronic bronchitis), and alveolar destruction with loss of elastic recoil (emphysema). Airflow limitation is largely irreversible and progressive.
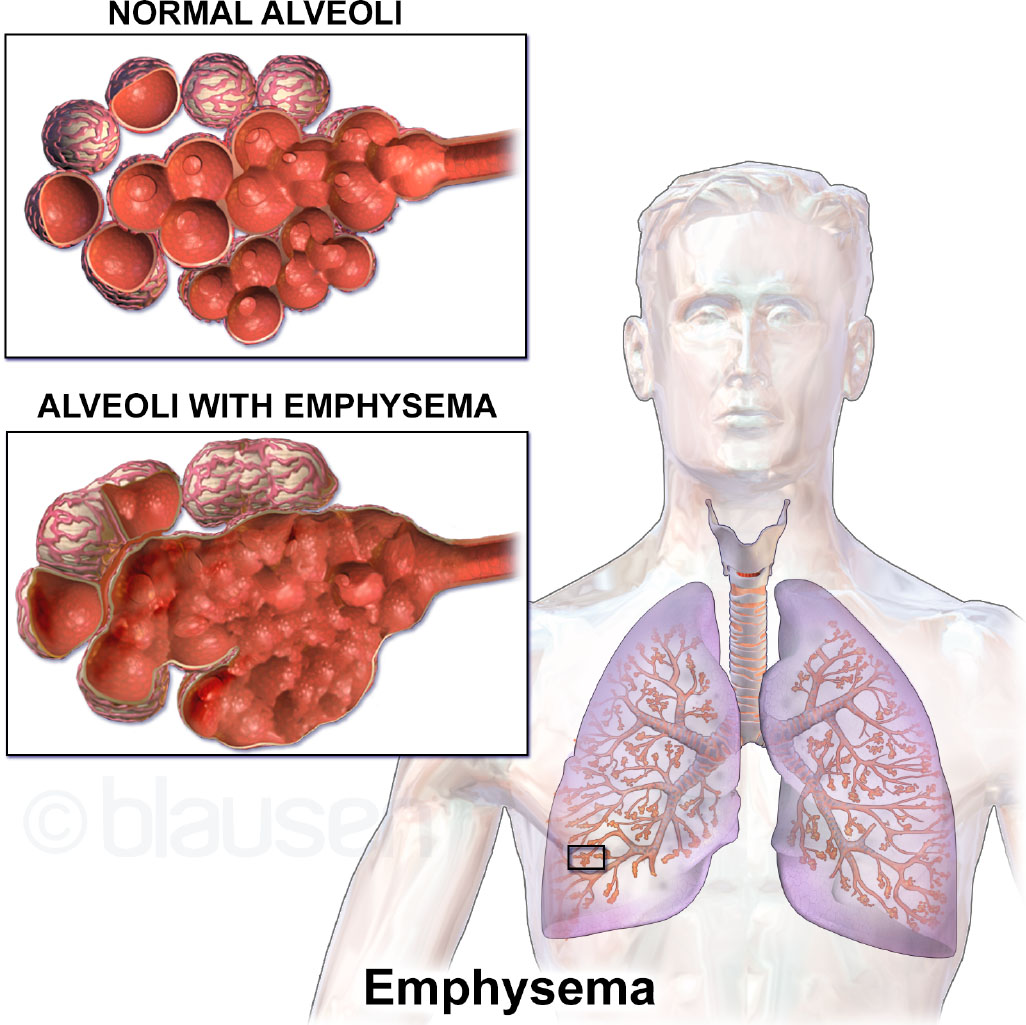 Illustration reference: OpenStax Medical-Surgical Nursing Ch.11.5.
Illustration reference: OpenStax Medical-Surgical Nursing Ch.11.5.
Inflammatory injury can involve airways, pulmonary vasculature, and lung tissue; common remodeling changes include airway narrowing, smooth-muscle hypertrophy, and eventual scar-related fixed narrowing. When alveolar walls lose elasticity, exhalation becomes less effective and carbon-dioxide trapping worsens. COPD is treatable but not curable; symptoms often become apparent only after significant lung injury and usually worsen over time when smoke exposure continues.
In selected patients, alpha-1 antitrypsin deficiency contributes to emphysematous lung injury by reducing protection against protease-mediated tissue damage. Alpha-1 antitrypsin deficiency-related COPD can show lower-lobe-predominant emphysematous involvement compared with smoking-related distribution patterns.
Chronic hypoxemia stimulates erythropoiesis (secondary polycythemia) and pulmonary vasoconstriction (cor pulmonale in severe disease). Hypercapnia develops when respiratory muscle fatigue limits compensation. COPD is also associated with increased risk of comorbid disease burden, including cardiovascular disease and lung cancer. COPD prevalence in U.S. adults in the midlife-to-older range remains high, and annual mortality burden is substantial.
Classification
- Chronic bronchitis: Productive cough ≥3 months/year for 2 consecutive years; “blue bloater” pattern.
- Emphysema: Alveolar destruction and air trapping; “pink puffer” pattern with barrel chest.
- GOLD classification: Severity graded by FEV₁ % predicted (I: >=80, II: 50-79, III: 30-49, IV: <30) with symptom/exacerbation burden integration.
- Risk-factor profile: Modifiable risks include smoking, secondhand smoke, occupational dust/fume exposure, and indoor biomass smoke. Nonmodifiable risks include age over 40 years, childhood respiratory infections, history of asthma, prematurity-associated underdeveloped lungs, and alpha-1 antitrypsin deficiency.
- Smoking-exposure pattern: COPD is frequently associated with smoking exposure above about 10-15 pack-years; environmental/occupational exposures also explain a substantial nonsmoker subgroup.
- Population-burden profile: Prevalence and mortality are influenced by structural factors and exposure burden, with disproportionate impact in some racialized communities.
Nursing Assessment
NCLEX Focus
Monitor O₂ saturation carefully in COPD — high-flow oxygen can suppress hypoxic drive in severe disease.
- Assess dyspnea, cough, sputum production, and activity tolerance at baseline and during exacerbations.
- Recognize adult-onset progression patterns and cold-season symptom worsening in many patients.
- Auscultate for prolonged expiration, wheezing, and diminished breath sounds.
- Monitor oxygen saturation — target 88–92% for hypercapnic COPD to preserve hypoxic drive.
- Trend resting versus exertional oxygen saturation and compare with prior baseline because early exertional desaturation can precede severe functional decline.
- Monitor for frequent respiratory infections, progressive fatigue, barrel chest, cyanosis, and unintentional weight loss in later-stage disease.
- Monitor for cachexia and digital clubbing as advanced chronic-disease cues.
- Treat low BMI plus declining muscle mass as high-risk nutrition findings because they correlate with lower exercise tolerance and worse COPD outcomes.
- Do not rely on BMI alone: some clients with higher body weight still show low fat-free mass and clinically significant pulmonary malnutrition risk.
- Assess for signs of cor pulmonale: peripheral edema, JVD, hepatomegaly.
- Assess exposure history (occupational particulates, indoor coal/wood smoke, secondhand smoke) and nonmodifiable COPD risk cues (childhood infections, prematurity, alpha-1 antitrypsin deficiency history).
- Screen for anxiety and depression, which are common comorbidities affecting adherence.
- Use spirometry criteria for diagnosis/staging: FEV₁/FVC under about 0.70 supports COPD, then stage severity with GOLD FEV₁ ranges.
- In suspected exacerbation, track the core triad (dyspnea increase, sputum volume increase, sputum purulence) plus adjunct signs such as fever, recent URI, and baseline-relative tachycardia/tachypnea.
- Use pulse oximetry and ABG together when oxygenation concern exists; PaCO2 above about 45 mmHg with acidemia supports hypercapnic respiratory-acidosis risk.
Nursing Interventions
- Administer bronchodilators (short-acting beta-agonists, anticholinergics) and corticosteroids as ordered.
- Position in high Fowler’s or tripod position to maximize respiratory effort.
- Teach pursed-lip breathing and diaphragmatic breathing to reduce air trapping.
- Distinguish controller versus rescue inhalers: use inhaled corticosteroids (for example fluticasone) daily with post-dose mouth rinse, and use albuterol for rapid dyspnea relief.
- Educate on smoking cessation — the only intervention proven to slow disease progression.
- Teach household smoke-elimination counseling, including second-hand-smoke harm (increased lung-cancer risk in nonsmokers and higher pediatric risk for lower-respiratory infection, SIDS, ear infections, and asthma worsening).
- Segment ADLs and position for breathing comfort to reduce dyspnea burden during routine activity.
- Reinforce prescribed supplemental-oxygen adherence when ordered.
- Coordinate pulmonary rehabilitation referral for eligible patients.
- Plan energy conservation strategies for activities of daily living.
- For severe dyspnea with malnutrition/cachexia risk, coordinate nutrition support with high-calorie intake planning when clinically appropriate.
- When intake declines, monitor for weak cough/poor secretion clearance and escalate early to reduce pneumonia-risk progression.
- For meal-associated dyspnea or fatigue, teach 4-6 small meals/day, upright posture while eating, slower bites with breathing pauses, and fluid timing between meals when fullness worsens breathing effort.
- In COPD with hypoxemia, reinforce antioxidant-forward choices (for example fruits, vegetables, whole grains, nuts, and lycopene-rich foods) within the patient’s calorie and weight goals.
- If bloating worsens breathing effort, individualize lower-gas meal planning and reassess tolerance.
- During and after exacerbations, trend weight and intake because repeated exacerbations can accelerate weight and muscle loss.
- After oxygen initiation in hypercapnia-risk exacerbations, reassess for CO2-retention deterioration cues within about 30-60 minutes and escalate as needed.
- Coordinate interdisciplinary discharge planning (respiratory therapy, pharmacy, nutrition, social support, and rehabilitation) to reduce readmission risk.
Oxygen Administration
In chronic hypercapnic COPD, excessive oxygen (>92%) can suppress the hypoxic respiratory drive. Titrate O₂ carefully to target 88–92%.
Pharmacology
| Drug Class | Examples | Key Considerations |
|---|---|---|
| Short-acting bronchodilators | Albuterol (SABA), ipratropium (SAMA) | Rescue use; teach inhaler technique |
| Long-acting bronchodilators | Salmeterol (LABA), tiotropium (LAMA) | Maintenance; not for acute relief |
| corticosteroids | Prednisone, budesonide/formoterol | Exacerbations: oral systemic; maintenance: inhaled |
| Phosphodiesterase-4 inhibitors | Roflumilast | Severe COPD with chronic bronchitis |
Clinical Judgment Application
Clinical Scenario
A COPD patient on 2L nasal cannula develops worsening dyspnea. SpO₂ is 86%. The nurse increases O₂ to 6L.
- Recognize Cues: O₂ saturation low, but rapid high-flow increase risks suppressing hypoxic drive.
- Analyze Cues: Patient has chronic hypercapnia; target 88–92%, not normoxia.
- Prioritize Hypotheses: Risk of respiratory depression from excessive O₂ in hypercapnic patient.
- Generate Solutions: Titrate O₂ to 88–92%, reassess frequently, prepare for escalation if needed.
- Take Action: Adjust to lowest O₂ flow achieving target SpO₂; notify provider; monitor ABGs.
- Evaluate Outcomes: SpO₂ maintained in safe range without worsening respiratory depression.
Related Concepts
- asthma-action-plan-and-exacerbation-management - Obstructive lung disease comparison and differentiation.
- respiratory-system - Normal airway physiology and defense mechanisms.
- oxygen-therapy - Oxygen delivery systems and monitoring in respiratory disease.
- evidence-based-respiratory-care - Bronchial hygiene and airway clearance techniques in COPD.
Self-Check
- Why is high-flow oxygen potentially dangerous for hypercapnic COPD patients?
- What distinguishes chronic bronchitis from emphysema clinically?
- Which intervention has the strongest evidence for slowing COPD progression?