Prenatal Testing During the Second Trimester
Key Points
- Second-trimester testing refines fetal-risk assessment and expands diagnostic options.
- Anatomy ultrasound and quad/integrated screening are common midpregnancy tools.
- Diagnostic tests such as amniocentesis and PUBS are more definitive but invasive.
- Nursing care focuses on informed choice, preparation, postprocedure safety, and follow-up.
Pathophysiology
Second-trimester testing evaluates structural development, chromosomal-risk signals, placental function, and selected fetal blood/genetic conditions. Screening tests estimate probability; positive screens require confirmatory diagnostics. Comprehensive obstetric ultrasound is commonly performed around 18 to 22 weeks to evaluate fetal number and viability, gestational age, anatomic survey, placental location, amniotic fluid volume, and maternal pelvic structures.
The quad screen is a common maternal-serum screen performed at about 15 to 20 weeks (with some pathways extending to about 22 weeks) and includes AFP, estriol, hCG, and inhibin A. Abnormal screening results increase concern for selected aneuploidy or neural-tube-defect risk and prompt discussion of targeted ultrasound and/or invasive diagnostic confirmation (for example amniocentesis). It is often emphasized when risk factors are present, such as maternal age 35 years or older or prior pregnancy affected by neural tube defect. AFP screening between about 15 and 22 weeks can be interpreted with other markers: low AFP with low estriol and abnormal hCG patterns can increase concern for trisomy risk, while elevated AFP increases concern for neural tube defects. Inhibin A addition improves trisomy 21 risk-screen performance in quad-screen pathways. Integrated/sequential screening frameworks commonly pair first-stage screening at about 10 to 13 weeks (blood plus ultrasound) with second-stage serum testing at about 15 to 22 weeks to refine risk estimates. Routine second-trimester laboratory surveillance also includes CBC or hemoglobin/hematocrit trend review, gestational-diabetes screening (commonly 24 to 28 weeks), and repeat blood type/Rh/antibody screening in Rh-negative pregnancies near 28 weeks.
Diagnostic procedures can identify specific chromosomal and blood disorders and guide pregnancy management, delivery planning, and newborn specialty preparation. Amniocentesis can provide diagnostic fetal-cell analysis in midpregnancy but carries procedural risk (generally under 1 percent miscarriage risk, with lower rates reported in some datasets), including fluid leakage, miscarriage, needle injury, Rh sensitization, uterine infection, and possible infectious transmission. Depending on indication, amniotic-fluid analysis can also support infection assessment and selected fetal-maturity evaluation. PUBS can be performed from about 18 weeks and is used for targeted diagnosis of fetal blood and chromosomal conditions (for example fetal anemia/thrombocytopenia, infection, isoimmunization), with potential therapeutic use for intrauterine transfusion or medication delivery. PUBS has high diagnostic accuracy but is used selectively because procedural risk is higher than many other prenatal tests. Counseling should include risk of miscarriage, fetal/maternal bleeding, cord hematoma, transient fetal bradycardia, infection, placental separation, or emergency delivery. Umbilical artery Doppler assessment may be used from about 22 weeks when fetal growth concerns emerge; abnormal flow can indicate impaired oxygen delivery and need for additional surveillance. If Doppler findings are abnormal, follow-up sonographic evaluation may include fetal middle cerebral artery, ductus venosus, and umbilical venous flow assessment to characterize placental insufficiency severity. Fetoscopy (after about 12 weeks, indication-dependent) can support direct fetal intervention in selected severe conditions such as twin-to-twin transfusion syndrome or amniotic band syndrome but carries invasive-risk tradeoffs; the fetoscope can also provide direct visualization and access for laser vessel ablation, band release, selected fetal procedures, or tissue sampling. In selected complex fetal-anomaly pathways, MRI may be used as adjunct imaging when ultrasound detail is limited.
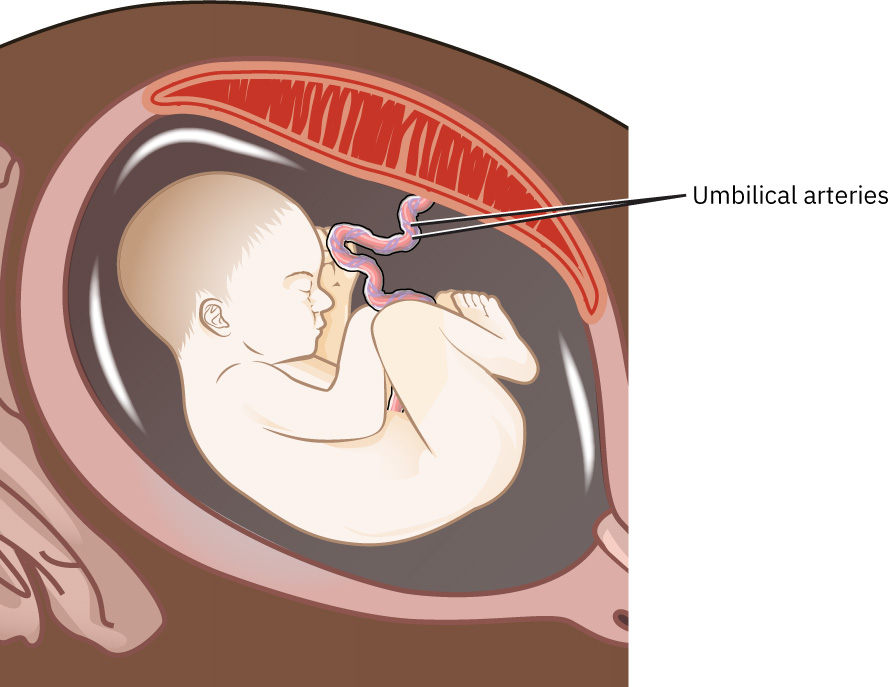 Illustration reference: OpenStax Maternal-Newborn Nursing Ch.13.2.
Illustration reference: OpenStax Maternal-Newborn Nursing Ch.13.2.
Classification
- Routine screening domain: Detailed anatomy ultrasound (often 18 to 22 weeks) and marker screening (quad/integrated part 2).
- Metabolic/hematologic surveillance domain: CBC-H&H trends, platelet surveillance, and gestational-diabetes screening workflow.
- Risk-focused surveillance domain: Umbilical Doppler and targeted imaging.
- Diagnostic domain: Amniocentesis (often 15 to 20 weeks), PUBS (from about 18 weeks), fetoscopy (after about 12 weeks when indicated), and selected invasive evaluations.
- Immunohematologic domain: Rh antibody titers and Rh prophylaxis planning.
- Adjunct imaging domain: Fetal MRI for selected structural or central nervous system concern pathways.
Nursing Assessment
NCLEX Focus
Always verify patient understanding of “screening risk” versus “diagnostic confirmation” before consent.
- Confirm gestational age and test timing eligibility.
- Review prior first-trimester results and current indications.
- Before ordering targeted second-trimester ultrasound pathways, reassess indication clusters such as vaginal bleeding, abdominal or pelvic pain, suspected cervical insufficiency or multifetal pregnancy, uterine-size and date discordance, amniotic-fluid abnormalities, possible placental complications, or abnormal biochemical markers.
- Assess procedural contraindications and Rh/infection status.
- For amniocentesis pathways, verify common indication triggers such as maternal age 35 or older, prior affected pregnancy, unexplained persistent AFP elevation, recurrent pregnancy loss, or Rh-sensitization concerns.
- Correlate ultrasound findings with dating history when fundal-height trend and gestational age appear discordant.
- For gestational-diabetes screening, verify prep and interpretation sequence: 1-hour 50 g glucose challenge (non-fasting in most protocols) followed by diagnostic 3-hour oral GTT when screening is elevated.
- Use local screen-positive thresholds for the 1-hour GCT (commonly over 130 mg/dL in serum pathways or over 140 mg/dL in fingerstick/whole-blood pathways).
- For the 3-hour 100 g GTT, treat two or more elevated values among the four time points as diagnostic of gestational diabetes in common protocols.
- After abnormal AFP/marker results, assess readiness for staged follow-up options (repeat marker testing, high-definition ultrasound, and/or invasive confirmation).
- When AFP is abnormal, reassess dating accuracy and multifetal status early because these are common non-anomaly causes of unexpected AFP levels.
- For PUBS planning after about 23 weeks, verify preprocedure instructions (including NPO window when ordered for possible procedural escalation).
- For Doppler and fetoscopy pathways, assess whether patient understands indication-specific goals (surveillance versus intervention) and procedure-related risk profile.
- In fetoscopy pathways, assess understanding of procedure risks including membrane rupture, procedure-triggered labor, and infection.
- Evaluate anxiety, values, and decision-support needs.
- Assess whether personal or religious values affect screening uptake and ensure the patient understands that risk screening is optional.
- Check posttest symptom vigilance capacity and follow-up access.
Nursing Interventions
- Provide test-specific education (purpose, limits, and possible next steps).
- For anatomy ultrasound preparation, verify last-menstrual-period dating, teach arrival with a full bladder when requested, and explain semi-recumbent positioning on a padded table during scanning.
- Teach practical glucose-testing workflow and thresholds used locally (screen-positive then diagnostic confirmation), and clarify fasting requirements for the diagnostic test.
- Clarify that positive marker screens indicate increased risk, not diagnosis, and that many abnormal AFP results do not result in confirmed fetal anomaly.
- Prepare comfort/privacy and procedural readiness.
- For amniocentesis, obtain baseline fetal heart rate, review blood type/Rh/antibody status, and explain expected sensations (brief stinging/pressure/cramping) before needle insertion under ultrasound guidance.
- Monitor maternal-fetal status after invasive testing and reinforce warning signs (for example persistent cramping, bleeding, fluid leakage, fever, or decreased fetal movement).
- After amniocentesis, teach temporary activity restriction (for example avoid strenuous exercise for about 24 hours) and distinguish expected mild spotting/discomfort from urgent warning symptoms.
- After PUBS, monitor fetal heart rate in the immediate recovery period, reinforce activity restriction for the remainder of the day, and teach urgent return precautions (chills, persistent cramps, fever, fluid leakage, bleeding, or decreased fetal movement).
- For fetoscopy workflows, reinforce consent quality, explain expected peri-procedure monitoring, and teach urgent symptom escalation for rupture-of-membranes signs, contractions, or infection symptoms.
- Coordinate genetics referral and multidisciplinary planning for abnormal findings.
- In Rh-negative pathways, verify repeat blood type/Rh/antibody screen near 26 to 28 weeks and coordinate antepartum rh-immune-globulin when antibody screen remains negative.
- If sensitizing events occur (for example placental abruption, abdominal trauma, or unexplained vaginal bleeding), coordinate Rh immune-globulin administration within about 72 hours and ongoing prophylaxis scheduling per protocol until delivery.
- Teach that postpartum Rh immune-globulin is indicated only if the newborn is Rh positive.
- Clarify that once true alloimmunization is established (persistent positive anti-Rh antibody), prophylactic Rh immune globulin will not reverse sensitization.
- Support patient autonomy, including informed refusal.
- Use noncoercive counseling when patients decline screening and document informed decision quality.
Positive-Screen Panic
Framing a positive screen as a diagnosis can trigger avoidable distress and rushed decisions.
Pharmacology
| Drug Class | Examples | Key Nursing Considerations |
|---|---|---|
| rh-immune-globulin | Rh-negative prophylaxis contexts | Review blood type/antibody status and administer per protocol timing. |
| analgesics | Postprocedure discomfort contexts | Mild cramping may occur; persistent pain or bleeding needs urgent reassessment. |
Clinical Judgment Application
Clinical Scenario
A patient has a positive quad screen and asks if the fetus definitely has a chromosomal condition.
- Recognize Cues: Misinterpretation of screening result.
- Analyze Cues: Anxiety may impair informed decision-making.
- Prioritize Hypotheses: Priority is clarification and confirmatory pathway planning.
- Generate Solutions: Explain risk-based nature of screening and discuss diagnostic options.
- Take Action: Arrange counseling and follow-up testing per preference.
- Evaluate Outcomes: Patient demonstrates accurate understanding and informed next-step choice.
Related Concepts
- prenatal-testing-during-the-first-trimester - Midpregnancy testing often follows early screening results.
- prenatal-testing-during-the-third-trimester - Abnormal midpregnancy findings may require late surveillance.
- fetal-growth-and-development - Anatomy and growth milestones drive second-trimester testing focus.
- genetics-in-reproductive-care - Carrier and chromosomal risk pathways shape counseling.
- person-and-family-centered-care - Testing decisions should reflect patient values and goals.
Self-Check
- Which second-trimester tests are screening versus diagnostic?
- What findings after amniocentesis or PUBS require urgent contact?
- How should nurses explain next steps after a positive quad screen?