Multiple Sclerosis
Key Points
- Multiple Sclerosis is a chronic immune-mediated CNS disorder characterized by progressive demyelination and lesion formation.
- Most common disabling neurologic disease in younger adults, often diagnosed between ages 20 and 40 years.
- Course may be relapsing-remitting or progressive, with variable motor, sensory, visual, cognitive, and psychosocial effects.
- Acute relapse treatment typically uses short-course high-dose IV glucocorticoids; long-term disease control relies on immune-modifying therapies.
- Care priorities focus on relapse control, function preservation, fatigue/heat management, and prevention of falls, skin injury, and social isolation.
- Adjunctive nutrition strategies (including ketogenic-pattern trials) have limited but emerging evidence for cognitive and fatigue-related symptom support.
Pathophysiology
MS is driven by immune-mediated injury to myelin and neural tissue in the brain and spinal cord. Demyelination slows or blocks impulse conduction, producing deficits in mobility, sensation, coordination, and communication.
Early disease may include partial remyelination with temporary symptom improvement, but recurrent lesion formation leads to cumulative neuronal injury, cortical atrophy, and progressive disability.
Classification
- Relapsing-remitting MS: Episodic exacerbations with partial or complete remission; most common presentation.
- Primary progressive MS: Steady neurologic decline without clear remissions.
- Secondary progressive MS: Progressive deterioration after an initial relapsing-remitting course.
- Progressive-relapsing MS: Ongoing deterioration with superimposed relapses and incomplete recovery.
Risk and Etiology Context
- Autoimmune dysfunction is central to disease mechanisms.
- Female sex and early-adult onset (often 20-40 years) are common epidemiologic patterns.
- Infectious associations include prior EBV exposure and other viral triggers.
- Genetic susceptibility increases with affected first-degree relatives.
- Lower vitamin D status and residence farther from the equator are associated risk patterns.
- Smoking increases risk and is linked with faster disability progression.
Nursing Assessment
NCLEX Focus
Track progression pattern over time and distinguish intermittent exacerbations from persistent deterioration.
- Assess symptom timeline (constant vs intermittent, worsening trend, remission pattern) and common aggravators such as fatigue, stress, and temperature extremes.
- Assess visual findings (unilateral vision loss, diplopia, nystagmus, patchy blindness) and communication changes (for example dysarthria).
- Assess for optic-nerve inflammatory patterns (for example painful vision loss) because demyelinating injury can acutely reduce visual function.
- Assess motor/sensory and coordination findings: weakness, spasticity, ataxia, dysmetria, tremor, paresthesia/hypoesthesia, mobility tolerance, and dizziness-related imbalance.
- Assess bowel/bladder and sexual-function changes, cognitive impairment, mood/anxiety/lability, and social-withdrawal risk.
- Assess fatigue burden and its effect on activity pacing, fall risk, and self-care completion.
- Assess diagnostics in context: blood testing is mainly used to exclude mimics (for example Lyme disease) and identify modifiable risk factors such as vitamin D deficiency.
- Assess CSF and neurodiagnostic findings together: lumbar puncture can show MS-pattern antibody abnormalities while MRI lesions and delayed evoked potentials support demyelinating conduction defects.
- During immunomodulator therapy, assess for nutrition-linked adverse-effect cues such as appetite loss, persistent nausea, dark urine, clay-colored stool, or jaundice that may signal hepatotoxicity.
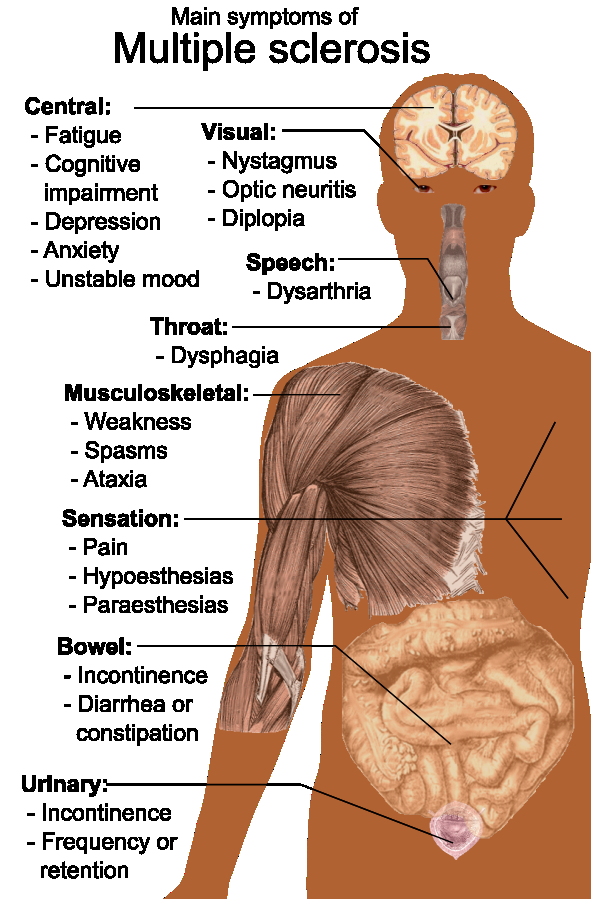 Illustration reference: OpenRN Health Alterations Ch.9.10.
Illustration reference: OpenRN Health Alterations Ch.9.10.
Nursing Interventions
- Encourage safe independent activity and daily stretching/strengthening within tolerance.
- Trend vital signs during exacerbations and during corticosteroid therapy; BP changes and sleep/mood shifts can indicate treatment burden.
- Use energy-conservation plans (activity clustering, planned rest, organization of frequently used items).
- Implement bowel/bladder training with scheduled toileting and hydration support.
- Apply fall precautions and skin-protection measures, including frequent assessment for breakdown with immobility/incontinence.
- Manage heat sensitivity (temperature control strategies and cooling aids when indicated) to reduce temporary symptom worsening.
- Support visual and communication adaptation (for example alternating eye patch use for diplopia, therapy-guided communication strategies).
- Monitor plasmapheresis sessions when ordered, including access-site status, infection prevention, and hemodynamic tolerance.
- Coordinate physical therapy and behavioral-health support for gait/balance deficits and anxiety/depression coping.
- If client pursues therapeutic diet modification, coordinate dietitian-guided planning and monitor tolerance/adherence so nutritional deficits do not worsen fatigue or function.
- Escalate persistent treatment-related nausea or intake decline early to prevent secondary malnutrition and dehydration during relapse-prevention therapy.
- Promote rest, stress-management skills, and participation in support groups to reduce isolation and improve coping.
Medical and Rehabilitation Management
- Disease-modifying management commonly uses interferons, oral immunomodulators, monoclonal antibodies, and S1P-receptor modulators; monitor for infection risk, organ toxicity, and class-specific cardiovascular or neuro-ophthalmic complications.
- Acute exacerbations are commonly treated first with short-course high-dose IV glucocorticoids (for example methylprednisolone); plasmapheresis is a common next-line strategy when steroid response is inadequate.
- IV immune-globulin pathways may be considered in selected refractory or steroid-intolerant contexts.
- Symptom-directed medications may include muscle relaxants for spasticity, anticonvulsant or tricyclic options for paresthesia-related pain, bowel-regimen agents, and bladder-relaxing medications.
- PT/OT/ST are central to mobility, ADL adaptation, visual-scanning strategies, and communication/swallow support.
- In severe refractory tremor, neurosurgical options (including deep-brain stimulation contexts) may be considered.
Functional Decline and Safety Risk
Uncontrolled fatigue, progressive weakness, heat-triggered symptom worsening, and falls can rapidly reduce independence and quality of life.
Pharmacology
| Medication Context | Examples | Key Nursing Considerations |
|---|---|---|
| Acute relapse therapy | IV methylprednisolone high-dose short course; IV gamma globulin context | Monitor glucose, mood, infection risk, and response to relapse treatment. |
| Interferon pathway therapy | interferon beta-1a, interferon beta-1b | Monitor CBC/LFT/thyroid trends, injection-site injury, mood or suicidality, and flu-like symptoms. |
| Oral immunomodulators | teriflunomide, monomethyl fumarate, glatiramer acetate | Screen TB/pregnancy risk, monitor hepatotoxicity and BP, avoid live vaccines, and reinforce barrier contraception when indicated. |
| Monoclonal antibody therapy | ocrelizumab, natalizumab, alemtuzumab | Use infusion-reaction precautions with premedication, monitor for infection/PML and autoimmune toxicity, and follow restricted-program monitoring requirements where applicable. |
| S1P-receptor modulators | siponimod pathway | Baseline ECG/labs and first-dose bradycardia monitoring are required; verify CYP2C9 genotype before siponimod and monitor for macular edema, PRES, and skin malignancy signals. |
| Spasticity and symptom control | baclofen-class and dantrolene contexts | Assess tone, sedation, fall risk, withdrawal safety, and liver toxicity risk with dantrolene. |
| Neuropathic/sensory symptom control | anticonvulsant or tricyclic contexts (for example gabapentin/pregabalin) | Monitor functional response, cognition, edema/weight changes, and abrupt-stop withdrawal risk. |
| Fatigue symptom control | modafinil, methylphenidate contexts | Reassess sleep quality, appetite, HR/BP response, and daytime function with stimulant-class therapy. |
| Bowel/bladder symptom programs | stool-softener/laxative and bladder-relaxant contexts | Monitor elimination pattern, retention risk, and hydration status. |
Health Teaching and Evaluation
- Teach medication purpose, side effects, and when to call the provider for adverse effects or relapse concerns.
- Teach adaptive self-care skills, equipment use, skin care, bowel/bladder routines, and home-safety modifications.
- Reinforce smoking cessation and risk-reduction habits that may slow disability progression.
- Include family/caregivers in education and connect to counseling/community support services.
- Reevaluate outcomes at each reassessment and revise plans as symptom pattern or functional status changes.
Clinical Judgment Application
Clinical Scenario
A client with relapsing-remitting MS reports worsening afternoon fatigue, increased stumbling, urinary urgency, and new social withdrawal during a hot summer period.
- Recognize Cues: Heat-linked symptom worsening with mobility and psychosocial decline.
- Analyze Cues: Exacerbation risk and fatigue burden are reducing safety and participation.
- Prioritize Hypotheses: Immediate priorities are fall prevention, function support, and symptom-trigger reduction.
- Generate Solutions: Initiate energy-conservation plan, heat-mitigation strategies, bladder routine support, and therapy/social-support referrals.
- Take Action: Implement safety interventions, reinforce teaching, and coordinate multidisciplinary follow-up.
- Evaluate Outcomes: Falls decrease, fatigue is better controlled, and participation in daily activities improves.
Related Concepts
- neurological-system - Myelin and impulse-transmission foundations for MS pathology.
- common-neurological-disorders-recognition-and-priority-care - Pattern recognition and escalation context.
- nursing-care-priorities-for-neuromuscular-impairment - Mobility, aspiration, elimination, and skin-risk overlap.
- muscle-relaxants - Spasticity-focused pharmacologic management context.
- fall-prevention - Core safety framework for gait and coordination impairment.
Self-Check
- Which findings suggest progression versus remission in MS symptom patterns?
- How do fatigue and heat sensitivity alter nursing care priorities?
- Which interdisciplinary strategies best preserve mobility and independence?