Alzheimer’s Disease
Key Points
- Alzheimer disease is the most common dementia subtype and causes progressive decline in memory, language, judgment, and daily function.
- AD is a neurodegenerative disorder with cortical atrophy and progressive neuronal loss.
- Although most cases occur after age 65, early-onset presentations can occur in the 40s and 50s.
- Current AD pharmacotherapy is not curative and usually provides modest, symptom-focused slowing of decline.
- Prevalence rises sharply with age (about 5% ages 65-74, 13.2% ages 75-84, and 33.4% ages 85+).
- Family caregivers often provide intensive unpaid support and are at high risk for physical and emotional strain.
- Diet patterns with anti-inflammatory and antioxidant emphasis (for example Mediterranean-style intake) may support slower cognitive decline as adjunctive care.
Pathophysiology
Alzheimer disease (AD) is a progressive neurodegenerative disease and a leading cause of major cognitive decline in older adults. Compared with expected aging changes, AD involves accelerated structural and functional brain deterioration.
Early described changes include progressive cortical atrophy and neuronal degeneration, with worsening impairment in cognition and independence over time.
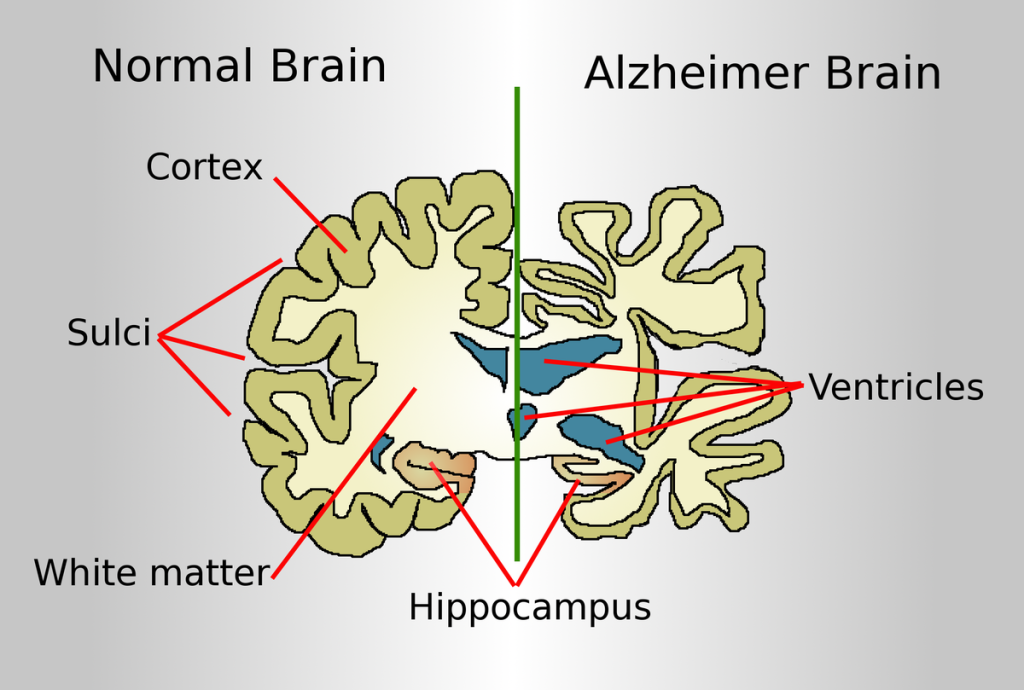 Illustration reference: OpenRN Health Alterations Ch.9.5.
Illustration reference: OpenRN Health Alterations Ch.9.5.
Hallmark microscopic findings include neurofibrillary tangles and neuritic plaques. Abnormal beta-amyloid and tau protein accumulation disrupts neuron-to-neuron signal transmission and accelerates neurodegeneration.
Initial damage often affects regions for memory, language, and thinking, so early manifestations commonly include short-term memory loss and language/executive decline. Pathologic brain changes may begin many years before clear clinical symptoms.
Neurotransmitter imbalance (including reduced acetylcholine, norepinephrine, dopamine, and serotonin) is also associated with worsening cognition and new-memory retention problems.
Classification
- Disease context: Most common dementia subtype in adults older than 65.
- Functional impact: Progressive deficits in memory, language, comprehension, attention, reasoning, and judgment.
- Core pathology: Neurofibrillary tangles and neuritic plaques causing impaired neuronal communication.
- Nonmodifiable risk factors: Increasing age, female sex in older age groups, family history/first-degree relative, and selected genetic/developmental vulnerability contexts.
- Modifiable and contextual risk factors: Tobacco exposure, inactivity, poor cardiometabolic control, low cognitive/social stimulation, and selected medication/toxin-related contributors.
- Clinical stages:
- Preclinical AD: Biomarker/brain changes without clear clinical symptoms.
- MCI due to AD: Mild symptoms that may not yet interfere with daily function.
- Mild dementia due to AD: Early symptom cluster with growing IADL impact (often finances and complex planning first), increasing vulnerability to financial exploitation/scams.
- Moderate dementia due to AD: Greater confusion, multistep ADL difficulty, wandering/incontinence, hallucinations-delusions, and behavioral symptoms including sundowning.
- Severe dementia due to AD: Profound communication and mobility decline with dysphagia, anorexia/weight loss, and complete ADL dependence, with high complication risk (for example aspiration pneumonia, thrombosis, infection/sepsis).
- Care context: High caregiver demand for ADL and home-safety support.
Nursing Assessment
NCLEX Focus
Assess both patient cognitive decline and caregiver health impact from the start of care.
- Assess baseline cognitive and functional status and trend progression over time.
- Assess onset, duration, and progression pattern and differentiate chronic decline from acute delirium superimposition.
- Assess for early-warning pattern changes (memory disruption of daily life, planning/problem-solving decline, familiar-task difficulty, time-place confusion, visuospatial trouble, word-finding changes, misplacing items without retracing, poor judgment, social withdrawal, and mood-personality change).
- Assess caregiver burden, fatigue, coping strain, and ability to sustain home support.
- Assess safety risks related to impaired judgment and declining self-management.
- Use family or significant-other collateral history when patient insight is limited.
- Use cognition tools matched to literacy and ability (for example, MMSE, Mini-Cog, set test, clock-drawing screening) to trend decline over time.
- Integrate diagnostic context: AD remains definitively confirmed by tissue pathology, while clinical workup may include CSF beta-amyloid/tau and PET imaging support.
- Use diagnostic patterns to exclude mimics: blood testing for reversible contributors (for example vitamin or thyroid disorders), MRI for regional atrophy/rule-out, PET for hypometabolism, and CT primarily for structural differential diagnosis.
- Check reversible contributors to cognitive impairment (for example vitamin B12, thyroid, and liver-related etiologies) during differential assessment.
- Screen for depression and other mood disorders because they can overlap with memory and concentration complaints.
Nursing Interventions
- Use structured cognitive and functional reassessment to guide stage-appropriate support planning.
- Reinforce food-first neuroprotective patterns (fruits/vegetables, whole grains, fish, nuts, olive-oil dominant fats) and limit ultraprocessed carbohydrate-heavy intake that can worsen metabolic-inflammatory burden.
- Integrate caregiver-support strategies early, including referral to respite and community support services.
- Reinforce home-safety planning as cognitive decline progresses.
- Use redirection/distraction, low-stimulation environments, and consistent routines to reduce agitation.
- Prefer validation approaches in severe-stage distress; avoid confrontational reality orientation that escalates agitation.
- Build injury-prevention plans: decluttered spaces, adequate lighting, wandering precautions/ID supports, and stage-appropriate supervision.
- Schedule toileting/incontinence care, nutrition support (finger foods, texture/liquid modification as needed), sleep-promoting daytime activity, and simplified communication cues.
- In acute-care settings, maximize observation, minimize room transfers, monitor wandering risk, and use nonpharmacologic calming strategies before restraint pathways.
- Use structured pain assessment (including tools suited for advanced dementia communication limits) and treat discomfort early to reduce agitation escalation.
- Coordinate case-management referral early after diagnosis for longitudinal support-service planning and caregiver support.
- Reinforce caregiver stress-reduction planning: realistic expectations, daily respite breaks, self-care routines, support groups/hotline use, and early advance-care/legal planning.
- For AD medications, teach no abrupt discontinuation, no doubling after missed doses, and no crushing/chewing of ER capsule formulations.
Caregiver Health Risk
Long-duration unpaid caregiving can cause physical and emotional decline that reduces care quality and safety.
Pharmacology
Pharmacologic management is stage- and symptom-dependent and should be integrated with nonpharmacologic safety, communication, and caregiver-support interventions. Current agents do not reverse AD and typically provide limited-duration symptomatic benefit.
| Medication Class | Examples | Key Nursing Considerations |
|---|---|---|
| Amyloid-targeting agents | lecanemab (aducanumab discontinued by manufacturer in 2024) | IV therapy used in selected mild-stage pathways; monitor MRI-linked amyloid-related imaging abnormalities and infusion/allergic reactions (for example fever, palpitations, dyspnea, angioedema). |
| Cholinesterase inhibitors | donepezil, rivastigmine, galantamine | GI effects and bradycardia risk; monitor heart rate, dizziness, and tolerance. |
| Glutamate regulators | memantine | For moderate-severe AD; monitor dizziness/confusion/constipation (constipation), renal function, and urine pH-related interaction risk (for example sodium bicarbonate, carbonic anhydrase inhibitors, and thiazides). |
| Combination cognitive agents | donepezil + memantine ER (Namzaric) | Used in moderate-severe AD when donepezil is already established; monitor mental status and ADL trajectory during weekly memantine titration. |
| Orexin receptor antagonists | suvorexant | Monitor next-day alertness, mood/suicidality concerns, and respiratory status. |
| Psychotropic agents for severe aggression/psychosis | risperidone, olanzapine, carbamazepine, valproic acid | Considered chemical-restraint contexts; follow policy-level safeguards and reassess necessity frequently. |
Clinical Judgment Application
Clinical Scenario
An older adult with progressive short-term memory loss is living at home with a spouse who reports exhaustion from increasing daily supervision needs.
- Recognize Cues: Progressive cognitive decline with caregiver strain.
- Analyze Cues: AD progression is affecting both patient safety and caregiver health.
- Prioritize Hypotheses: Immediate priorities are safety stabilization and caregiver support.
- Generate Solutions: Update care plan with stage-based supports and community support linkage.
- Take Action: Implement safety routines and initiate caregiver-support referral.
- Evaluate Outcomes: Home safety and caregiver coping improve while function is monitored.
Evaluation should be continuous: reassess cognitive/functional trends, review new diagnostic data, and revise the care plan when outcomes are partially met or unmet.
Related Concepts
- dementia - Broader major-neurocognitive framework and differential context.
- acetylcholinesterase-inhibitors - Donepezil-centered cholinergic pharmacotherapy and administration safety.
- caring-for-clients-with-dementia - Daily communication and behavior-support strategies.
- caregiver-role-strain - Psychosocial and functional burden in long-term family caregiving.
- neurological-system - Neuroanatomic and neurotransmission foundation for disease understanding.
Self-Check
- Why must caregiver assessment begin early in Alzheimer care planning?
- Which findings best indicate progression from mild functional impact to higher-risk dependence?
- How should nursing priorities shift as cognitive decline worsens?