Multimodal Pain Management and PCA Safety
Key Points
- Multimodal pain management combines medications and nonpharmacologic interventions for better outcomes.
- Opioid therapy requires tight monitoring for sedation, respiratory depression, dependence, and overdose risk.
- Nonopioid and adjuvant therapies expand control while reducing opioid burden.
- PCA is effective when patient selection, pump programming, dual verification, and reassessment protocols are followed.
Pathophysiology
Pain is biologically, psychologically, and socially mediated; single-modality treatment often leaves residual burden. Multimodal strategy targets multiple mechanisms simultaneously, improving relief while limiting toxicity from dose escalation in any one class.
Opioids reduce pain signaling but can suppress respiratory drive and cognition. NSAIDs and acetaminophen target inflammatory and nociceptive pathways, while adjuvants support neuropathic, spastic, or localized pain states.
Nonpharmacologic interventions (movement therapy, guided imagery, massage, environmental modulation, cognitive strategies) reduce central amplification and improve function, coping, and quality of life.
When postoperative pain is poorly controlled, stress-hormone signaling (for example cortisol excess), hyperglycemia tendency, inflammatory persistence, and reduced mobility can slow tissue healing and rehabilitation progress while increasing complication risk.
Classification
- Pharmacologic core: Opioid, nonopioid, and adjuvant analgesic layers.
- Nonpharmacologic core: Physical, psychological, emotional, and environmental interventions.
- Severity ladder context: Mild/moderate/severe pain categories can guide stepwise medication escalation.
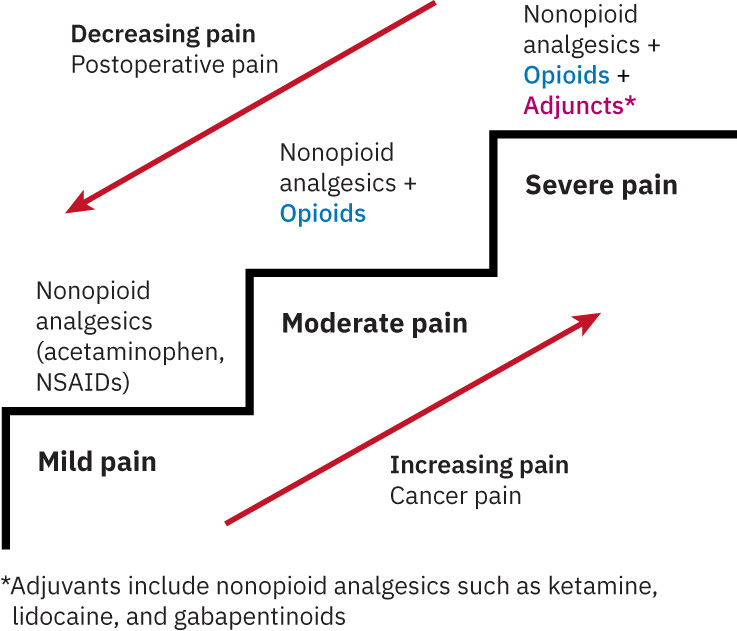 Illustration reference: OpenStax Medical-Surgical Nursing Ch.7.3.
Illustration reference: OpenStax Medical-Surgical Nursing Ch.7.3.
- PCA structure: Loading dose, demand (bolus) dose, basal rate (if used), lockout interval, and one-hour maximum limit.
- Safety domains: Candidate selection, monitoring, adverse-effect prevention, and documentation integrity.
- Standards-of-care domains: Admission/ED pain screening, age/condition-appropriate reassessment, functional-goal tracking, and opioid-risk discharge education.
Nursing Assessment
NCLEX Focus
Evaluate treatment success by function and safety trends, not analgesic intensity alone.
- Assess pain mechanism and severity pattern to select medication class mix.
- In hospital settings, ensure pain is screened at admission (and ED entry when applicable) and reassessed with tools matched to age, cognition, and communication ability.
- Escalate promptly for new severe pain or pain unresponsive to current plan; sudden severe pain or chest pain/pressure requires immediate emergency-level response per protocol.
- Treat persistent severe breakthrough pain despite active chronic-pain therapy as a higher-acuity escalation cue that may require advanced inpatient management.
- Characterize breakthrough-pain trigger pattern (for example activity, stress/illness flare, end-of-dose failure, or unprovoked episode) to decide whether rescue-only treatment is sufficient or baseline regimen revision is required.
- Assess opioid risk factors: sedation vulnerability, respiratory compromise risk, concurrent sedative use.
- Assess nonopioid risk profile before selection (for example older adult status, pregnancy, and liver/cardiovascular/kidney/GI comorbidity context).
- Assess PCA candidacy: cognition, ability to follow directions, and contraindication profile.
- Assess PCA contraindication profile explicitly (for example opioid allergy, active infection concern, elevated intracranial pressure, severe renal dysfunction, or bleeding-risk context) according to facility policy.
- Treat confusion or oversedation as PCA exclusion cues because safe PCA use requires alert, self-directed button activation.
- Assess opioid misconceptions (for example fear of addiction during short-term postsurgical use) that reduce adherence to analgesia plans.
- In postoperative pathways, reinforce that short-term opioid use for acute surgical pain is distinct from substance use disorder, while still applying standard safety monitoring.
- In patients with known substance-use disorder history, anticipate possible higher postoperative opioid requirements from tolerance and escalate collaboratively for individualized dosing.
- For adult primary-care opioid use, assess plan alignment with CDC prescribing domains: initiation need, opioid selection/dose, initial duration with follow-up, and risk-harm mitigation.
- Before outpatient opioid prescribing or continuation, verify controlled-substance history in PDMP; when chronic opioid therapy continues, repeat PDMP review at least every 3 months per policy/guideline alignment.
- Assess response to nonpharmacologic therapies and patient adherence barriers.
- Assess whether selected nonpharmacologic modalities are being used as complementary therapy (with medications) or alternative therapy (in place of medications), and verify safety/goal fit for each pathway.
- Assess all current natural-product use (herbs, vitamins, supplements, oils) and possible interaction risk; remind patients that many supplements are not FDA-regulated for safety/effectiveness in the same way as prescription medications.
- Assess eligibility for electrical modalities (for example TENS) by screening implantable-device status and major contraindication contexts (for example pregnancy-specific restrictions, active bleeding/hemorrhage, seizure disorder, cancer, or DVT risk).
- Assess undertreatment risk in nonverbal, cognitively impaired, culturally pain-suppressing, and language-barrier populations.
- Assess patient beliefs about pain meaning, acceptable treatments, and traditional remedies to prevent culturally driven underreporting or refusal.
- Assess fear-amplification patterns (for example algophobia, catastrophizing, or avoidance behavior) that can increase perceived pain and reduce function.
- Assess trauma and PTSD context when pain is linked to past injury or abuse, because untreated trauma can sustain pain distress loops.
- Assess maladaptive coping (for example alcohol/substance self-medication, severe activity withdrawal) and protective habits (movement, sleep routine, stress regulation).
- Assess learned pain behaviors (for example prolonged inactivity because it gives short-term relief) that can worsen long-term pain and function.
- Assess whether preserving awareness/consciousness is a priority in serious illness or end-of-life care, because this can affect acceptance of sedating analgesics.
- Assess practical access barriers (for example interpreter availability, medication cost/coverage, and follow-up access) that can reduce pain-plan effectiveness.
Nursing Interventions
- Implement medication safety steps: right patient/medication/dose/route/time plus post-dose surveillance.
- Document administered analgesics immediately in the MAR/eMAR and update nonpharmacologic intervention records promptly to reduce duplicate-dose and handoff error risk.
- For planned rehabilitation sessions, coordinate interdisciplinary premedication timing (commonly 30-60 minutes before PT/OT) to improve participation and safety.
- Complete pain reassessment before and after analgesic administration to confirm indication and response.
- If nausea/vomiting is present, coordinate with the prescriber to use the most effective route and avoid oral dosing when oral absorption/tolerance is unreliable.
- Minimize distractions during opioid preparation/administration and follow independent double-check processes for high-alert handling per policy.
- Follow controlled-substance waste policy with required witness documentation when opioid waste occurs.
- Pair pharmacologic treatment with at least one nonpharmacologic modality when feasible.
- For acute soft-tissue injury, start an early RICE pattern (rest, ice, compression, elevation) and reassess progression after the first 24-48 hours as goals shift from inflammation control to tissue healing and mobility restoration.
- Use thermotherapy deliberately: heat commonly about 5-20 minutes for stiffness/chronic discomfort and cold up to 20 minutes for acute inflammation; keep a barrier between skin and heat/cold source and inspect skin routinely for injury.
- Avoid stereotype-based assumptions (for example “stoic culture means no pain”); combine self-report, behavior, and clinical findings for individualized plans.
- Select nonpharmacologic modalities aligned to patient preference and goals (for example distraction, relaxation, positioning, cutaneous stimulation, or mind-body approaches), and document effectiveness against comfort goals.
- In inpatient settings, confirm provider-order requirements before applying nonpharmacologic modalities such as heat/cold, massage, acupuncture/acupressure, and TENS-based pathways.
- Frame nonpharmacologic trials as iterative and individualized: if a modality is safe but ineffective, switch methods rather than abandoning nonpharmacologic care entirely.
- Pair chronic-pain plans with CBT/mindfulness-based coping strategies and graded-exposure/graded-activity approaches when fear avoidance is present.
- Use practical movement-based options (physical therapy, low-intensity exercise, or yoga-style movement/breathing) to improve mobility, stress regulation, and function.
- Offer selected natural and mind-body adjuncts when appropriate (for example aromatherapy, guided imagery, meditation, mindfulness, CBT, or yoga/tai-chi style movement), and document patient-specific benefit versus burden.
- Use supportive creative/animal-assisted distraction options when available (for example art, music, or pet-therapy pathways) to improve relaxation and reduce pain-signal salience.
- For chronic pain with inflammatory pattern (for example fibromyalgia contexts), reinforce dietary coaching that emphasizes vegetables, whole grains, and lean proteins while reducing refined sugars and excess red meat when aligned with nutrition goals.
- Provide medication teaching in culturally sensitive, linguistically appropriate language, using visuals/demonstration when needed for health-literacy fit.
- Reinforce that pain management is a patient right and that early treatment is safer than waiting for severe escalation.
- In coexisting SUD and pain, frame planning around two parallel goals: treat pain adequately while addressing SUD risk with structured safeguards.
- Coach patients that pain avoidance and non-disclosure can delay diagnosis/treatment of serious causes; reinforce early reporting and timely reassessment.
- Build pain plans with the patient/family using realistic measurable goals that include function (for example breathing, turning, ambulation), not score-only targets.
- Use small, achievable function goals to counter hopelessness and reinforce treatment efficacy signals over time.
- Match analgesic intensity and route to reported severity using a stepwise approach (nonopioid first when appropriate, then escalate), favoring the lowest effective dose with the least invasive route.
- In acute severe pain trajectories, teach expected transition steps (for example IV opioid to oral opioid to nonopioid as condition improves) so patients can anticipate step-down changes.
- In postoperative discharge planning, transition analgesia from IV to oral route as recovery stabilizes; avoid last-minute IV opioid dosing immediately before discharge because respiratory monitoring is still required.
- Apply WHO-ladder logic flexibly: severe acute pain may require starting at a higher potency rung with later step-down, while chronic pain is often escalated stepwise from lower rungs.
- In chronic non-cancer pain, avoid routine opioid-first strategies when effective alternatives exist; use opioids only after risk-benefit review and monitoring plan.
- For OUD-associated pain, maximize nonpharmacologic and nonopioid options first (for example exercise/physical therapy/CBT, NSAIDs, topical agents, and selected adjuvants such as SNRIs/TCAs); if opioids are clinically necessary, do not withhold solely because OUD history is present.
- In patients younger than 18 years, avoid opioid-first pain strategies when effective alternatives are available; if opioids are required, use the lowest effective dose with intensified monitoring.
- For PRN analgesics, verify doses administered in the prior 24 hours and cumulative limits before giving an additional dose.
- For outpatient PRN ranges (for example one-to-two tablets), coach “start low then reassess” to avoid unnecessary oversedation or overdose.
- When opioid therapy is required in patients with SUD risk, co-establish shared goals/known risks, use a treatment agreement, start low and titrate slowly, and schedule close follow-up reassessment.
- For PCA, verify bolus/basal/lockout settings with dual nurse sign-off per policy.
- In pediatrics, use PCA only when the child is developmentally/cognitively able (commonly age >6 years with adequate understanding) and reinforce patient-only button activation.
- Educate patient/family that only the patient should activate PCA unless explicit protocol says otherwise.
- Intensify PCA monitoring for high-risk profiles (for example sleep apnea, obesity, renal/hepatic impairment, or recent sedation/anesthesia exposure).
- Keep naloxone access immediate when opioid therapy is active and treat oversedation as an early warning for impending respiratory depression.
- Intensify sedation/respiratory checks for opioid-naive patients, recent opioid dose increases, or concurrent benzodiazepine/sedative exposure.
- Monitor common opioid/PCA adverse effects (for example constipation, nausea/vomiting, pruritus, urinary retention) and treat early to sustain adherence.
- Anticipate opioid adverse-effect management orders early (for example stool softener/laxative and antiemetic) to reduce treatment interruption.
- Reassess analgesic effect by route and onset (commonly about 15-30 minutes after IV and within about 60 minutes after oral dosing, per policy); if pain remains above acceptable level, escalate pharmacologic and nonpharmacologic options and notify the provider.
- If breakthrough episodes recur despite rescue dosing, escalate for plan revision (for example baseline regimen adjustment plus trigger-targeted nonpharmacologic support) rather than repeating rescue-only cycles.
- For neuraxial/implanted opioid delivery (for example epidural or intrathecal pumps), monitor closely for dosing/device complications and escalate immediately for withdrawal signs, neurologic change, or cardiorespiratory deterioration.
- At handoff, reconcile demand-versus-delivered counts and remaining controlled-substance volume with dual-nurse documentation.
- Monitor bowel function and start prevention measures (fluids, early ambulation, stool softener/laxative plan) when opioid-induced constipation risk is present.
- For complex pain or opioid-risk cases, escalate referral pathways (for example pain specialist or opioid-treatment support) per policy.
- For trauma-linked pain burden, coordinate trauma-informed mental-health referral pathways alongside analgesic care.
- Proactively start opioid bowel-prevention strategies (hydration, fiber/diet as appropriate, stool-softener/laxative plan, mobility) unless contraindicated.
- Use environmental nonpharmacologic supports (light/noise/temperature optimization) with relaxation, breathing, CBT, and movement-based approaches.
- Cluster care tasks when feasible to reduce sleep interruption and preserve restorative rest during painful recovery periods.
- Use distraction strategies (music, conversation, games, guided activities) around procedures and daily flares; pediatric patients often respond strongly to age-appropriate distraction.
- Screen contraindications before therapeutic-touch options (for example active rash/infection, selected cardiovascular risk states, pregnancy-specific precautions) and individualize pressure/intensity.
- Use massage/manipulation only when risk screen is acceptable; avoid or escalate review in anticoagulated patients and in contexts such as fracture, clot risk, open wound/infection, or fragile bone status.
- For acupuncture pathways, confirm trained-provider delivery and contraindication screen (for example pacemaker context, anticoagulation risk, and pregnancy-related restrictions) before referral.
- Explain that evidence strength differs across complementary modalities (for example stronger for selected movement/behavioral approaches, more limited for magnet-therapy claims) and align expectations to documented outcomes.
- Combine compatible nonpharmacologic methods when useful (for example environmental adjustment plus movement plus relaxation) and reassess which component mix produces the best patient-specific response.
- For TENS use, place electrodes near target nerve pathways per protocol and stop/escalate if symptoms worsen or contraindication concerns emerge.
- Consider biofeedback for chronic pain when available, emphasizing repeated training sessions and self-regulation skill carryover between visits.
- Use professional interpreter pathways for non-English-speaking patients and adapt medication plans to affordability/access constraints when barriers are identified.
- Support discharge safety teaching for opioid use, storage, and disposal, plus home factors that may worsen pain or reduce plan effectiveness.
- Include secure medication storage teaching (keep out of reach/locked away from children and visitors) at first-dose counseling and discharge.
- In end-of-life comfort-focused care, dosing decisions prioritize symptom relief aligned with goals of care rather than routine numeric withholding thresholds alone.
- In culturally or spiritually sensitive end-of-life contexts, co-design plans that balance symptom relief with awareness goals (for example titration strategy plus nonpharmacologic support).
- Apply the rule of double effect in end-of-life care: when the intended action is relief of suffering, proportional opioid use is ethically supported even if unintended sedation may occur.
- For chronic pain follow-up, encourage a pain journal tracking triggers, interventions, and function response to guide plan revision with the care team.
- In pediatric analgesia, keep dosing weight-based and verify safe limits before administration:
acetaminophen 10-15 mg/kg/dose q4-6h PRN, maximum4 doses/dayand80 mg/kg/day.ibuprofen 4-10 mg/kg/dose q6-8h PRN, maximum400 mg/doseand40 mg/kg/dayup to1200 mg/day.
- Avoid aspirin in children younger than 12 years and in children/teens with varicella or flu-like illness because of Reye syndrome risk.
- For severe pediatric pain, prefer morphine or hydromorphone over codeine/tramadol (and use fentanyl with heightened caution) due variable metabolism and respiratory-risk concerns.
Opioid and PCA Harm Risk
Oversedation typically precedes respiratory depression; early recognition and naloxone readiness are critical.
Pharmacology
Use opioid-sparing strategies when possible, especially in chronic pain and high-risk populations. Monitor for class-specific adverse effects: GI bleeding/kidney/cardiovascular risk with NSAIDs (with short-duration caution for agents such as ketorolac), hepatic risk with excess acetaminophen or concurrent heavy alcohol use, and CNS depression with opioid or sedative combinations. Differentiate tolerance (reduced response requiring higher dose), physical dependence (withdrawal with abrupt stop), and addiction (compulsive use despite harm) while still applying strict safety monitoring. For adjuvant options (for example tricyclics, gabapentinoids, corticosteroids, muscle relaxants, topical anesthetics), include agent-specific psychiatric/sedation/metabolic monitoring and taper plans when discontinuation risk exists.
Clinical Judgment Application
Clinical Scenario
A postsurgical patient on PCA reports persistent breakthrough pain and increasing drowsiness.
- Recognize Cues: Inadequate control plus sedation warning signs.
- Analyze Cues: Current PCA or adjunct plan may be imbalanced for efficacy versus safety.
- Prioritize Hypotheses: Immediate priority is respiratory safety while preserving analgesia.
- Generate Solutions: Reassess PCA settings, review concurrent sedatives, add nonopioid/nonpharmacologic support.
- Take Action: Escalate to provider, intensify monitoring, and implement revised multimodal plan.
- Evaluate Outcomes: Safer alertness profile with improved pain-function balance.
Related Concepts
- comprehensive-pain-assessment-and-documentation - Supplies baseline and reassessment data required for safe titration.
- pain-pathway-gate-control-and-classification - Mechanism-based rationale for multimodal approach.
- physiologic-behavioral-and-affective-pain-responses - Interprets treatment response signals and distress cues.
- medication-administration-safety-measures - Medication process safeguards and error prevention.
- pain-in-older-adults - Population context for altered risk-benefit balance.
Self-Check
- Which findings suggest opioid toxicity rather than uncontrolled pain alone?
- What three programmed PCA elements must nurses verify every handoff?
- Why does multimodal therapy generally outperform single-agent escalation?