Comprehensive Pain Assessment and Documentation
Key Points
- A 0-10 pain rating is a useful screening question but is not a complete pain assessment.
- Pain is commonly treated as a routine vital-sign domain, requiring repeated assessment across care transitions and interventions.
- Effective pain care starts with multidimensional baseline assessment, not score-only screening.
- Subjective data collection is structured with PQRSTU and patient-centered follow-up questions.
- Objective data (vitals, behavior, exam findings) are essential when self-report is limited.
- Documentation and timed reassessment determine whether interventions meet comfort-function goals.
- The patient should remain an active participant in selecting realistic comfort-function goals and evaluating response.
Pathophysiology
Pain is subjective and variably expressed across developmental stages, cognition, culture, and illness context. Because expression may be muted, exaggerated, or nonverbal, nursing assessment must integrate patient report with observable physiologic and behavioral cues.
Inadequate assessment leads to misclassification and ineffective treatment, while thorough baseline data supports targeted intervention selection and safer escalation. Reassessment closes the clinical loop by proving response or treatment failure.
Classification
- Subjective stream: PQRSTU, OLDCARTES, or COLDSPA with descriptors, pain history, and comfort-function goal.
- Objective stream: Physical findings, vital trends, behavior/posture/facial cues.
- Scale selection stream: Numeric scale, Wong-Baker FACES, McCaffrey Initial Pain Assessment Tool, FLACC, COMFORT Behavioral Scale, PAINAD, Behavioral Pain Scale (BPS), or CPOT based on age and communication profile.
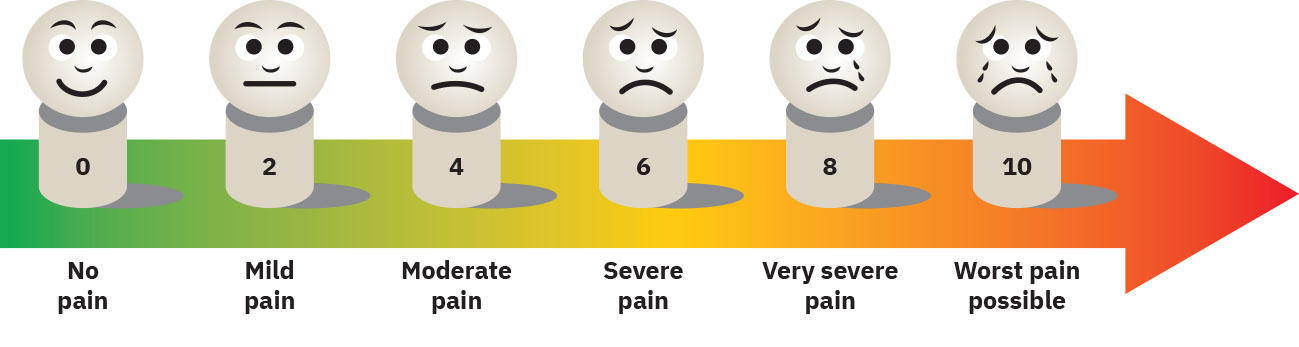 Illustration reference: OpenStax Clinical Nursing Skills Ch.16.2.
Illustration reference: OpenStax Clinical Nursing Skills Ch.16.2. - Documentation stream: Baseline, intervention, follow-up, and outcome comparison to goal.
Nursing Assessment
NCLEX Focus
Select the assessment tool the patient can understand; tool mismatch can produce misleading severity ratings.
 Illustration reference: OpenRN Nursing Skills 2e Ch.2.5.
Illustration reference: OpenRN Nursing Skills 2e Ch.2.5.
- Use PQRSTU for consistent subjective baseline: provocation/palliation, quality, region, severity, timing/treatment, understanding.
- In postoperative pathways, begin pain assessment immediately on PACU arrival and anchor early trends to this transfer-time baseline.
- Perform routine pain checks during baseline care, before and after procedures, and whenever the patient reports pain.
- Use alternative structured frameworks (OLDCARTES or COLDSPA) when they better fit setting workflow while maintaining equivalent data depth.
- Use location mapping/body charts when available and clarify whether pain is localized, radiating, or referred.
- Determine duration pattern early (acute, chronic/persistent, or breakthrough flare) because this changes urgency, expected physiologic response, and treatment planning.
- Assess developmental level separately from chronological age because communication ability and pain expression may not align with age alone.
- Assess psychosocial modifiers: mood symptoms, trauma/PTSD history, interpersonal support quality, and perceived social rejection.
- Assess culture, socioeconomic constraints, and access barriers (for example treatment cost, insurance, job-security concerns) that can suppress reporting or delay care.
- Address culture with individualized inquiry rather than assumptions; stereotype-based pain expectations can lead to undertreatment.
- Assess sex-assigned-at-birth and gender context without stereotyping, and document barriers to equitable treatment when present.
- Select pain scale by age, cognition, language, and communication ability.
- Treat patient pain self-report as valid subjective data; avoid nurse-side downrating based on appearance alone.
- Confirm the patient understands the selected scale before scoring; if numeric scoring is not understood (for example language, literacy, cognition, sedation), switch to a visual or alternate structured tool.
- For language barriers during acute postoperative recovery, use visual scales (for example FACES) with professional medical-interpreter support to confirm score meaning and reduce misclassification risk.
- For Wong-Baker FACES, use age-appropriate explanation (commonly for age 3 and older, and adults who prefer visual scales) and instruct the patient to choose the face that matches their pain; do not select a face based on appearance alone.
- Consider a more comprehensive initial tool (for example McCaffrey) when severity alone is insufficient and broader impact/cause/effect mapping is needed.
- Use FLACC (Face, Legs, Activity, Cry, Consolability) for infants/young children or other patients unable to provide reliable verbal self-report.
- In neonatal assessment, use NIPS for preterm and full-term infants; use FLACC for infants from about 2 months onward and for other nonverbal children.
- Use COMFORT Behavioral Scale in mechanically ventilated pediatric patients when pain/sedation must be assessed behaviorally.
- Use PAINAD (0-10) in advanced dementia when self-report is unreliable, tracking breathing, vocalization, facial expression, body language, and consolability.
- Use CPOT in nonverbal/intubated critical-care adults when self-report is not possible, scoring facial expression, body movement, muscle tension, and ventilator compliance/vocalization pattern.
- In acute pain, document vocalization profile (for example crying, screaming, moaning, gasping, or grunting) with timing and intervention response.
- In infants and younger children, treat crying and related vocal cues as primary pain-expression data when verbal descriptors are limited.
- In older children, pair age-appropriate self-report tools (for example Wong-Baker FACES) with observed vocal cues such as crying or yelling.
- For toddlers and many preschoolers, use dolls/body maps to support pain-location communication when verbal precision is limited.
- For preschoolers who frame pain as punishment, use corrective reassurance and developmentally simple explanations to reduce distress and improve reporting accuracy.
- For adolescents, include private assessment time away from peers/caregivers when social pressure may suppress pain disclosure.
- In children with disabilities, trend mood, activity, eye-contact/attention, and body-part-directed behavior changes as potential pain indicators.
- In infants, do not assume all crying reflects pain; reassess after comfort measures (for example swaddling, feeding, rocking) and escalate pain evaluation when crying persists.
- For NIPS, use score bands as rapid severity support (
0-1no pain,2mild,3-4moderate,5-7severe) while still integrating full clinical context. - Treat atypical referred patterns (for example jaw/arm/shoulder pain with autonomic symptoms) as high-priority escalation cues.
- Assess objective cues including vital signs, guarding, grimacing, mobility limits, and exam abnormalities.
- Include skin/muscle/joint review for swelling, laceration, discoloration, tenderness, temperature change, gait change, range-of-motion limits, and strength decline as location/aggravation clues.
- In patients with limited self-report, use autonomic trend shifts (sympathetic stress versus parasympathetic settling) as one indicator of worsening pain or treatment response.
- In nonverbal or sedated patients, include respiratory distress cues (for example nasal flaring, loud breathing, accessory-muscle use) when evaluating pain-related physiologic stress.
- In older adults with cognitive impairment, include caregiver-reported pain observations and communication patterns as assessment data.
- Structure behavioral review across five domains when possible: facial expression, vocalization, movement, emotional response (including flat affect), and interaction pattern changes.
- Recognize that behavioral presentation can vary widely (quiet/withdrawn versus angry/agitated) and may not match numeric self-report, especially in chronic pain.
- Assess functional impact (sleep, ADLs, activity, work, social engagement) and define comfort-function goal aligned to patient priorities (for example return-to-work function or family-mealtime comfort in serious illness).
- Clarify the comfort-function goal as the patient’s acceptable pain tolerance while maintaining daily function; goal thresholds vary by patient and should drive plan evaluation.
- Before giving analgesic or musculoskeletal medications, verify baseline allergy history and prior adverse medication responses.
- Assess expectation/interpretation patterns (for example anticipated severe pain, placebo-responsive optimism, or catastrophic interpretation) because these can change perceived severity and response.
- Assess pain-experience history and learned pain behaviors (for example family-modeled catastrophizing, avoidance, or delayed reporting) because these patterns can persist and distort current assessment.
- Assess avoidance behaviors (pain non-disclosure, activity withdrawal, delayed evaluation) that may mask progression or increase risk.
- Perform targeted exam of painful areas for tenderness, swelling, discoloration, temperature change, guarding, and mobility limits; integrate associated cues such as diaphoresis, nausea, vomiting, or appetite loss.
- In surgical patients, include incision/surgical-site inflammation or swelling checks and identify movement/position triggers that worsen or relieve pain.
- Compare posture/gait/ROM findings with patient baseline and age-context expectations, because abnormal physical findings can exist with or without active pain.
- Avoid single-cue conclusions; corroborate posture, facial expression, vitals, exam findings, and report history before attributing abnormalities to pain.
- Use open-ended questions and validate inconsistent responses with follow-up probing (for example, high numeric score but pain described as currently tolerable at rest).
- Assess expectation framing (for example “nothing will help” versus confidence in plan) because expectancy can amplify or reduce perceived pain burden.
- Screen for catastrophizing, distress, and withdrawal behaviors that can worsen disability and adherence.
- In chronic debilitating pain with depression or severe hopelessness, complete suicidality safety screening and escalate per protocol.
- Account for social context when interpreting cues because patients may hide facial or behavioral pain signs around clinicians, coworkers, or specific family members.
- In children who withdraw from touch at painful sites, avoid forceful palpation; use visual cues, patient/caregiver descriptors, and adjunct diagnostics while planning analgesia before provocative assessment when needed.
Nursing Interventions
- Document patient-reported pain verbatim in quotation marks when appropriate, identify who provided the report (patient versus caregiver), and clearly identify scale used and context.
- Document the patient-defined comfort-function goal because it serves as the baseline for treatment-plan evaluation and revision.
- Document objective findings that corroborate or contextualize report.
- Document relevant team communication/handoffs tied to pain findings and plan updates.
- Document every pharmacologic and nonpharmacologic intervention with time and rationale.
- Perform and document timely reassessment to determine if comfort-function goal is reached.
- For analgesics, use route-timed reassessment benchmarks per policy (commonly about 15-30 minutes after IV dosing and within about 60 minutes after oral dosing), then document response and next-step plan.
- If current pain exceeds the documented comfort-function goal, intervene and complete follow-up reassessment within 1 hour, then document response and next-step plan.
Documentation Safety Risk
Delayed or incomplete pain documentation can cause duplicate dosing, undertreatment, and unsafe handoffs.
Pharmacology
Reassessment timing must reflect route and expected onset (for example IV versus oral). Medication-effect documentation should include efficacy and adverse-effect surveillance, not pain score alone.
Clinical Judgment Application
Clinical Scenario
A patient reports 8/10 pain after intervention but appears calmer and has improved mobility.
- Recognize Cues: Persistent high score with partial functional improvement.
- Analyze Cues: Pain burden remains above goal despite some response.
- Prioritize Hypotheses: Current regimen is insufficient for target function.
- Generate Solutions: Reassess cause/type, optimize multimodal plan, and escalate if needed.
- Take Action: Update care plan and document all data streams clearly.
- Evaluate Outcomes: Trend toward patient-defined comfort-function goal.
Related Concepts
- pain-pathway-gate-control-and-classification - Mechanistic and classification foundation for assessment logic.
- physiologic-behavioral-and-affective-pain-responses - Objective and affective cue interpretation support.
- pain-management - Converts assessment results into treatment strategy.
- nursing-assessment-and-care-plans - General process alignment for reassessment-driven care.
- health-literacy-assessment-and-plain-language-education - Improves scale comprehension and shared goals.
Self-Check
- Why can a numeric scale alone be unsafe for complex pain presentations?
- What documentation elements are required after a pain intervention?
- How does the comfort-function goal improve individualized pain management?