Masculinizing Hormonal Therapy
Key Points
- Masculinizing regimens are used to develop male secondary sex characteristics in individuals assigned female at birth.
- Expected effects include increased facial/body hair, voice deepening, fat redistribution, and increased muscle mass.
- Common risks include acne, dyslipidemia, hypertension, polycythemia, diabetes risk, infertility, and thromboembolic/cardiovascular events.
- Testosterone and related androgen pathways are teratogenic in pregnancy exposure contexts.
- Baseline pregnancy status should be confirmed before therapy initiation in clients with pregnancy potential.
- Typical testosterone goals are in the male physiologic range (about 320-1000 ng/dL), with many follow-up plans targeting about 400-700 ng/dL.
Regimen Overview
Masculinizing hormone therapy is individualized and may involve androgen-centered regimens to achieve virilization goals while limiting adverse effects. Clinical plans should integrate comorbidity risk, medication interactions, fertility goals, and patient priorities.
Common Components and Reference Dosing
| Component | Typical Adult Dosing Reference | High-Yield RN Notes |
|---|---|---|
| Testosterone cypionate/enanthate (IM/SC) | 50-100 mg weekly or 100-200 mg every 2 weeks | Check injection timing for peak/trough interpretation; monitor hematocrit, lipids, and BP trends. |
| Testosterone transdermal gel | 50-100 mg daily | Prevent secondary exposure to children/household contacts at application sites. |
| Testosterone transdermal patch | 2.5-7.5 mg daily | Watch for application-site reactions and skin-tolerance issues. |
| Testosterone undecanoate IM | 750 mg every 10 weeks or 1000 mg every 12 weeks | Observe for acute serious reactions after injection per product safety protocol. |
| Testosterone pellet implant | 75 mg pellet, often 6 pellets every 3-4 months | Reinforce insertion follow-up and adverse-effect surveillance. |
| Danazol | Agent-specific dosing/protocol | Synthetic androgen option with major pregnancy and thromboembolic cautions. |
| Fluoxymesterone / methyltestosterone | Agent-specific oral dosing | Anabolic-androgenic pathways with hepatic/cardiovascular and mood-risk monitoring. |
| Progesterone (selected cases) | Variable by protocol | Consider only when clinically indicated; evidence for virilization augmentation is limited. |
Expected Effects and High-Risk Concerns
Expected virilizing effects:
- Facial and body hair growth
- Voice deepening
- Increased muscle mass
- Body-fat redistribution
- Increased libido and cessation of menses
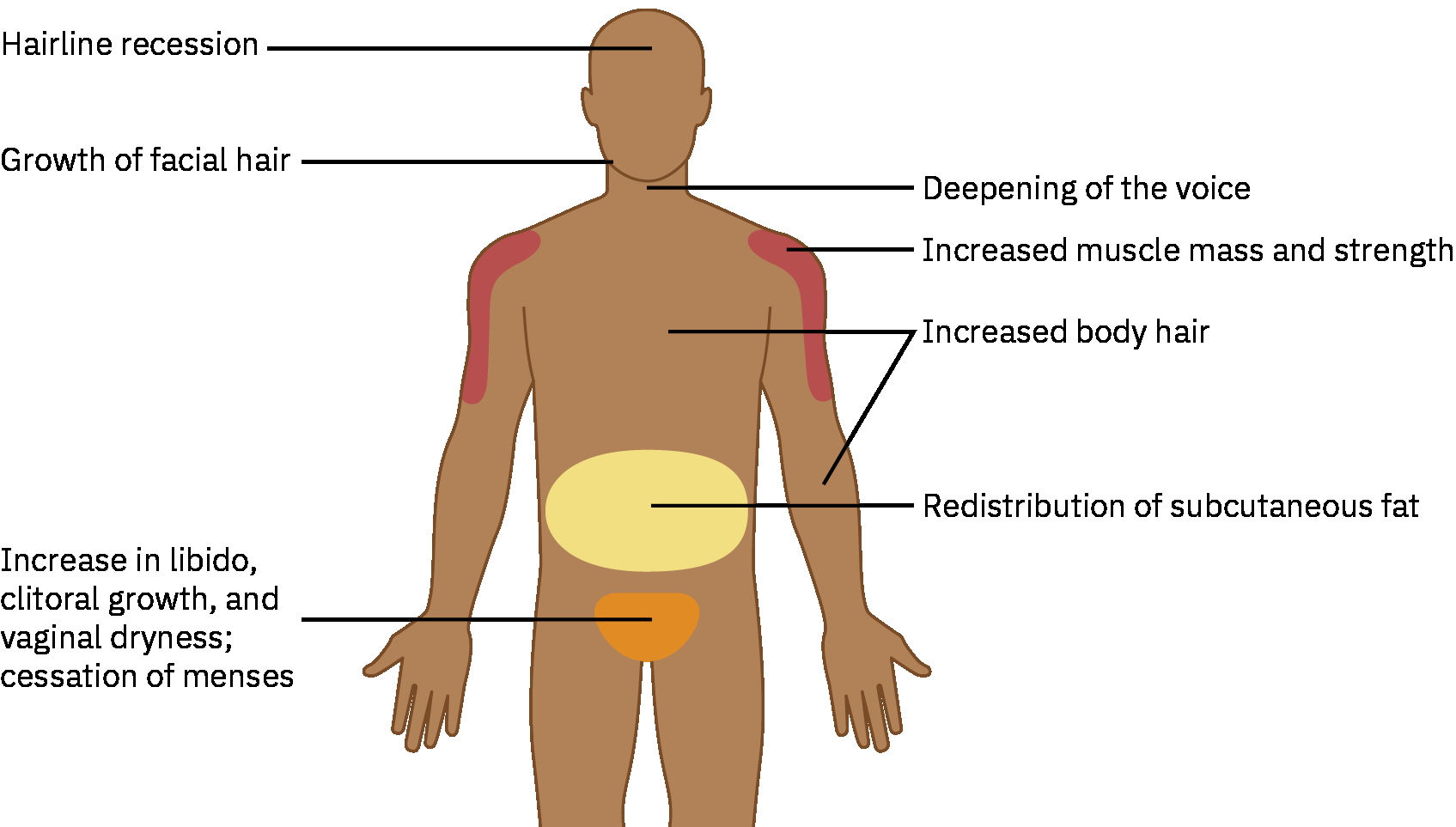 Illustration reference: OpenStax Pharmacology Ch.37.3.
Illustration reference: OpenStax Pharmacology Ch.37.3.
Potential complications requiring active monitoring:
- Acne, weight gain, androgenic alopecia
- Elevated cholesterol, hypertension, polycythemia
- Type 2 diabetes risk and cardiovascular/thromboembolic complications
- Vaginal dryness, pelvic discomfort, and infertility concerns
Monitoring Targets and Follow-Up
- Monitor virilizing and adverse effects every 3 months during the first year, then about every 6 to 12 months.
- Follow serum testosterone levels at follow-up visits.
- Practical testosterone goals commonly used:
- Male physiologic range about 320-1000 ng/dL
- Follow-up management range often about 400-700 ng/dL
- For parenteral testosterone, peak levels are commonly checked about 24-48 hours after injection and trough levels immediately before injection.
- Obtain hematocrit and lipid profile at baseline and follow-up.
- Perform bone-density screening before initiation in clients at osteoporosis risk; otherwise begin by age 60 or earlier if sex-hormone levels remain low.
- Continue anatomy-based cancer screening (for example cervix or breast tissue when present).
Nursing Precautions
- Verify baseline pregnancy status when clinically relevant because testosterone-pathway therapy can harm fetal development.
- Review baseline comorbid conditions and medications before starting treatment.
- In clients with diabetes, monitor for glucose-lowering effects of androgens and coordinate antihyperglycemic dose adjustments when needed.
- Review interaction risk with oral anticoagulants, corticosteroids, and diabetes medications in testosterone-based regimens.
- Use culturally sensitive, affirming communication throughout counseling and follow-up.
- Provide fertility counseling (for example oocyte cryopreservation options) before long-term therapy escalation.
Danazol Pregnancy Risk
Danazol can cause androgenic fetal effects. Confirm negative pregnancy status before therapy and use nonhormonal contraception during treatment when pregnancy is possible.
Testosterone Safety Alerts
Escalate for anaphylaxis-like symptoms after testosterone undecanoate injection, monitor for blood-pressure increases with enanthate/undecanoate products, and prevent secondary exposure to topical testosterone in children.
Client Education
- Teach route-specific administration (injection, patch, gel, implant) and adherence schedule.
- Reinforce urgent reporting of mouth/lip/tongue swelling, breathing difficulty, rash, severe acne or pelvic pain, and symptoms of hormone imbalance or loss of virilization effect.
- Reinforce continued wellness visits and routine preventive screening.
- Connect clients with affirming community, specialty-clinic, and support resources.
Related Concepts
- androgens-and-anabolic-steroids - Testosterone and anabolic-agent pharmacology details.
- feminizing-hormonal-therapy - Parallel regimen-level framework for estrogen-centered pathways.
- sexual-identity-orientation-and-inclusive-nursing-care - Inclusive communication and bias-aware care framework.