Feminizing Hormonal Therapy
Key Points
- Feminizing regimens usually combine an antiandrogen with estrogen to suppress testosterone and promote female secondary sex characteristics.
- Common antiandrogen pathways include spironolactone, gonadotropin-releasing-hormone-gnrh-analogs, or finasteride (when spironolactone is unsuitable).
- Practical follow-up hormone targets are testosterone about 30-100 ng/dL and estradiol less than 200 pg/mL.
- High-priority risks include thromboembolic/cardiovascular events with estrogen and hyperkalemia with spironolactone.
- Nursing priorities include baseline risk screening, scheduled laboratory surveillance, fertility counseling, and culturally sensitive education.
Regimen Overview
Feminizing hormone therapy is individualized based on baseline health, current medications, organ function, risk profile, and treatment goals. Regimens are commonly staged or combined, and dose plans may be adjusted over time to balance efficacy and safety.
Initial antiandrogen use may precede estrogen, or antiandrogen and estrogen may be initiated together depending on protocol and patient factors.
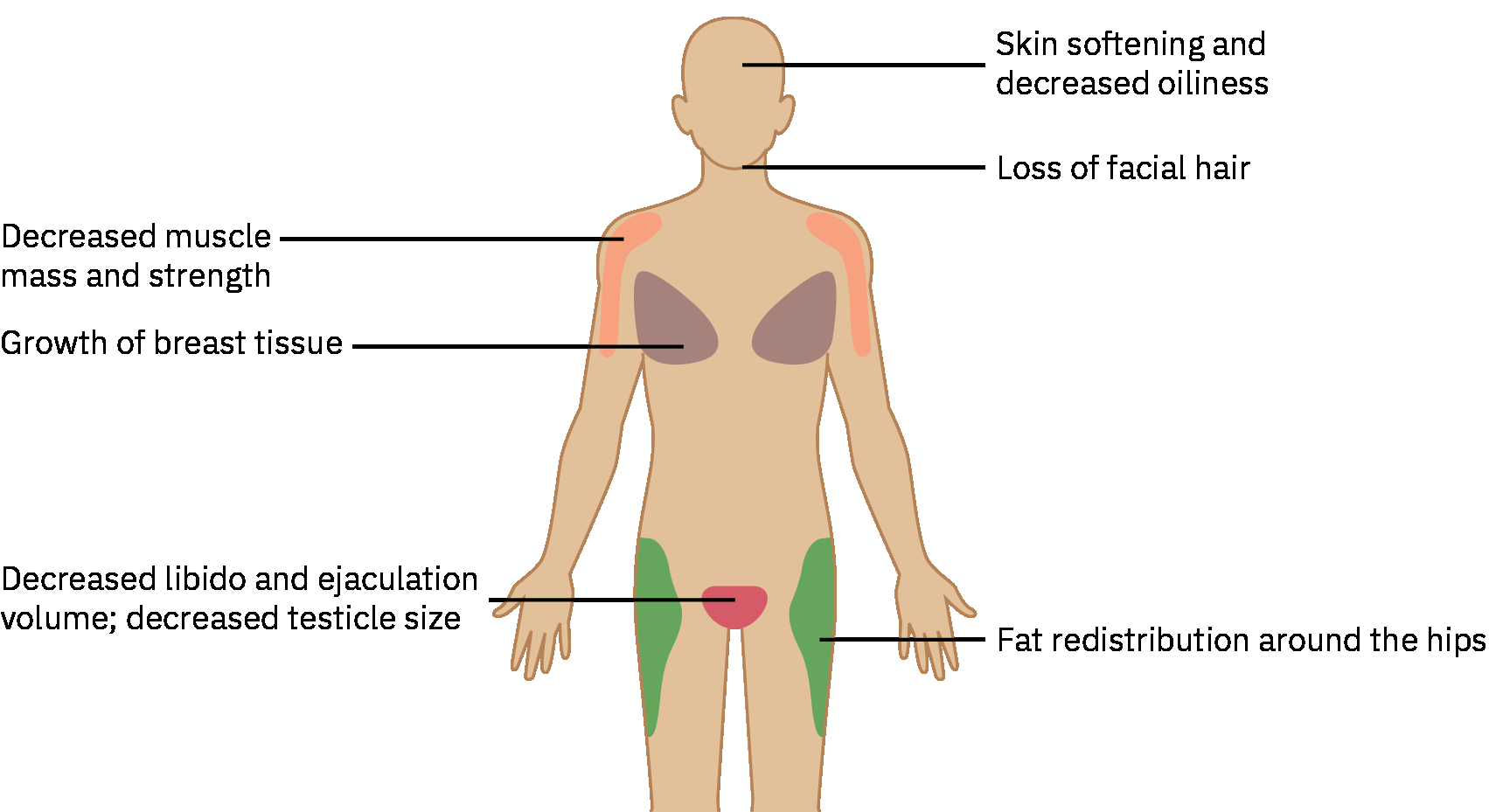 Illustration reference: OpenStax Pharmacology Ch.37.2.
Illustration reference: OpenStax Pharmacology Ch.37.2.
Common Components and Reference Dosing
| Component | Typical Adult Dosing Reference | High-Yield RN Notes |
|---|---|---|
| Spironolactone | 100-200 mg/day PO; may be titrated up to 400 mg/day | Monitor potassium, renal function, and cardiac rhythm risk; review ACE inhibitor/ARB/NSAID interactions and potassium-rich salt substitutes. |
| Estradiol oral (17-beta-estradiol) | 2-4 mg/day PO | Monitor thromboembolic/cardiovascular risk and hormone levels; route alternatives may be selected by risk profile. |
| Estradiol IM (valerate/cypionate) | Valerate 5-30 mg IM every 2 weeks; cypionate 2-5 mg IM weekly | Confirm injection-technique teaching and adherence to follow-up labs. |
| Estradiol transdermal patch | 0.1-0.4 mg transdermally twice weekly | Useful non-oral pathway; reinforce patch-site rotation and schedule adherence. |
| Progesterone/progestin | Variable dosing in available protocols | Routine use for additional breast development is not consistently supported; risk-benefit should be individualized. |
| GnRH analog (for example leuprolide) | About 3.75-7.5 mg IM monthly (agent-specific) | May reduce estrogen or spironolactone requirements; cost/access and dosing burden can limit feasibility. |
| Finasteride | 1-5 mg PO daily | Lower antiandrogen potency than spironolactone; useful when spironolactone is contraindicated or not feasible. |
Monitoring Targets and Follow-Up
- Monitor feminizing response and adverse effects every 3 months during the first year, then about every 6 to 12 months.
- Track serum testosterone and estradiol at follow-up visits.
- Common practical targets:
- Testosterone about 30-100 ng/dL
- Estradiol less than 200 pg/mL
- Monitor prolactin and triglycerides at baseline and on follow-up.
- Monitor potassium in spironolactone-containing regimens.
- Screen bone density before treatment in clients at osteoporosis risk; otherwise initiate screening by age 60 or earlier if sex-hormone levels remain low.
- Continue anatomy-appropriate cancer screening (for example breast and prostate pathways when clinically indicated).
Adverse Effects and Contraindication Priorities
- Estrogen-related high-risk events: DVT/PE, stroke, myocardial infarction, hypertension, and lipid/prolactin abnormalities.
- Spironolactone-related high-risk event: Hyperkalemia with possible life-threatening dysrhythmia.
- Finasteride safety: Pregnancy-exposure precautions for tablet handling and fetal-risk counseling.
- GnRH analog considerations: Bone-density effects, potential QT/cardiac concerns, and evidence gaps in long-term outcomes.
- Use added caution in clients with smoking history, obesity, hypertension, renal disease, or thrombotic history.
Immediate Escalation Cues
Escalate rapidly for chest pain, dyspnea, unilateral leg swelling, severe mood changes, irregular rhythm, seizure-like activity, lip/mouth swelling, or severe muscle cramps.
Nursing Interventions and Client Education
- Complete baseline assessment: comorbidities, medication reconciliation, and relevant labs/diagnostics before starting therapy.
- Provide culturally sensitive and identity-affirming communication throughout assessment, teaching, and follow-up.
- Coordinate fertility counseling before treatment escalation (for example sperm cryopreservation pathways when desired).
- Teach medication names, purpose, dosing schedule, administration route, and major interaction precautions.
- Reinforce wellness follow-up and preventive screening adherence during hormone therapy.
- Connect clients with supportive community and specialty resources when needed.
Related Concepts
- potassium-sparing-diuretics - Spironolactone antiandrogen pathway and hyperkalemia monitoring.
- gonadotropin-releasing-hormone-gnrh-analogs - Alternative testosterone-suppression pathway.
- 5-alpha-reductase-inhibitors - Finasteride adjunct/alternative antiandrogen option.
- hormonal-therapy - Estrogen/progestin adverse-effect and black-box context.
- masculinizing-hormonal-therapy - Parallel regimen-level framework for testosterone-centered pathways.
- sexual-identity-orientation-and-inclusive-nursing-care - Communication and bias-aware care framework.
- transgender-inclusive-breast-and-cervical-cancer-screening - Anatomy-based preventive screening planning.
Self-Check
- Which labs and follow-up intervals are core safety requirements in the first year of feminizing therapy?
- Why is potassium monitoring mandatory when spironolactone is used in a feminizing regimen?
- Which urgent symptoms require immediate escalation during therapy?