Physiologic Changes Due to Pregnancy
Key Points
- Pregnancy produces coordinated changes across nearly all body systems to support fetal growth, labor, and postpartum transition.
- Placental hormones drive many maternal adaptations, including cardiovascular, respiratory, endocrine, and metabolic shifts.
- Many changes are expected and benign, but some can mimic pathology and require careful assessment.
- Nursing education helps patients distinguish expected discomforts from warning signs.
Pathophysiology
Maternal physiology adapts from early gestation onward to increase oxygen/nutrient delivery, protect uteroplacental perfusion, and prepare for birth and lactation. Key reproductive changes include uterine enlargement, cervical softening, increased vaginal vascularity/discharge, and breast preparation for feeding.
Uterine growth is substantial: approximately 60 g pre-pregnancy to about 1,000 to 1,200 g near term. The fundus is typically at the symphysis pubis by about 12 weeks and near the xiphoid around 36 weeks, then contributes to organ displacement, rib-cage shape change, and center-of-gravity shift.
Early endocrine support is also dynamic: hCG maintains the corpus luteum so progesterone production continues until placental progesterone production is established by mid-pregnancy. Rising estrogen/progesterone also suppresses FSH/LH secretion, which helps prevent ovulation during pregnancy. Cervical glands produce thick mucus that forms a protective mucus plug against ascending infection until labor-related dilation begins. Cervical connective tissue is relatively firm early in pregnancy to support retention, then becomes more elastic in the final 3 to 4 weeks as labor approaches.
Vaginal tissue undergoes vascular and structural expansion, and secretions typically become thicker, white, and more acidic. This helps limit bacterial ascent but can increase candidiasis susceptibility. Vaginal and perineal tissues also relax and elongate in preparation for birth. Breast glandular tissue enlarges, nipples/areola become more prominent and darker, Montgomery glands increase lubrication, and colostrum production typically begins around 12 to 16 weeks even though full milk secretion remains hormonally suppressed until postpartum.
Cardiovascular adaptations include increased blood volume (about +1,500 mL by 30-32 weeks, or roughly 40-50% above nonpregnant baseline), increased cardiac output (about 30-50% by around 28-30 weeks), mild heart-rate rise (about +15 to +20 bpm), lower peripheral vascular resistance, and relative hemodilution (physiologic anemia). Stroke volume also increases (about 25-30%). A soft systolic flow murmur may be present in many normal pregnancies, and BP commonly reaches its lowest point around 28 weeks before trending back toward prepregnancy values by about 36 weeks; expected maternal pulse is commonly interpreted in a broader range (about 60-120 bpm). As the uterus enlarges, the heart is displaced upward and to the left.
Hematologic adaptation includes plasma expansion (about 50%) that outpaces RBC rise, producing lower hematocrit trends despite increased oxygen-carrying demand. Hypercoagulability intensifies during pregnancy (including plasma fibrin increase around 40% and fibrinogen increase around 50%), with slight platelet-count decline from dilution, and can increase venous-thrombus risk when combined with venous stasis.
Respiratory adaptations include diaphragm elevation (commonly about 4 cm), a shift from abdominal toward thoracic breathing, rib-cage diameter increase (about 6 cm), increased tidal volume (about 35-50%), and mild respiratory alkalosis. Maternal metabolic rate can rise by about 15% with oxygen consumption increase around 20%, so increased ventilatory demand is commonly experienced as mild dyspnea. Estrogen-related nasal mucosal vascular congestion can cause stuffiness, congestion, or epistaxis.
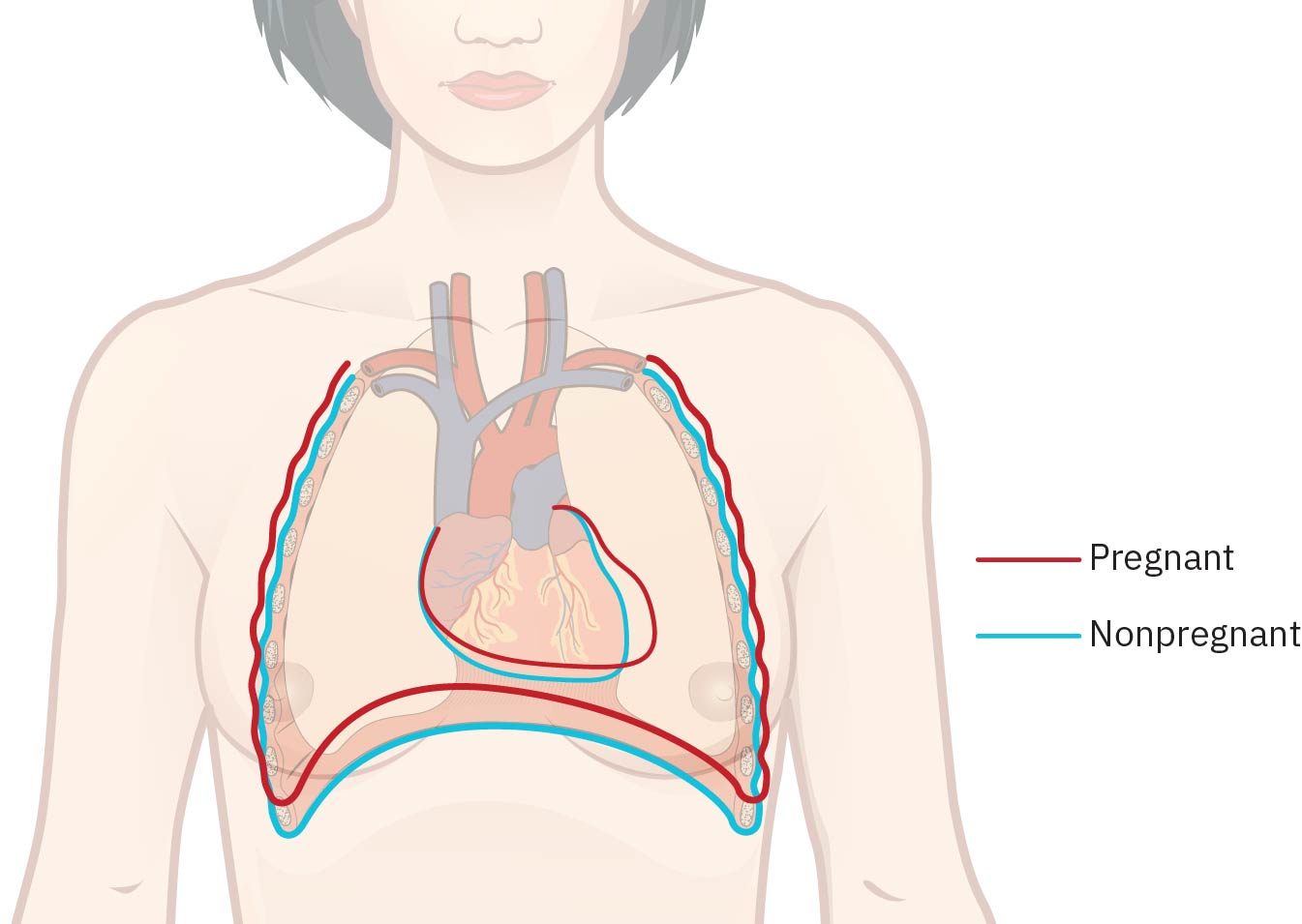 Illustration reference: OpenStax Maternal-Newborn Nursing Ch.10.1.
Illustration reference: OpenStax Maternal-Newborn Nursing Ch.10.1.
Pregnancy-associated rhinitis with nasal hyperemia/edema can increase snoring and sleep-disordered-breathing burden, so worsening nocturnal breathing symptoms should be reassessed with hypertensive-risk context (including preeclampsia cues).
Gastrointestinal and oral adaptations are largely progesterone-mediated motility slowing, which contributes to nausea/vomiting, reflux/GERD, delayed gastric emptying, and constipation. Oral changes may include taste/smell shifts, increased salivary acidity, ptyalism, and increased gingival vascularity with bleeding/periodontal vulnerability. Slower gallbladder emptying can increase bile stasis and raise cholelithiasis/cholecystitis risk, especially in higher-risk age groups.
Renal and urinary changes include increased kidney blood flow (about 50%), increased filtration, reduced bladder tone, urinary stasis risk, and faster renal excretion of some drugs, contributing to higher UTI vulnerability and medication-adjustment needs. Mild urine glucose/protein increases can be physiologic from filtration load, but clinicians must still distinguish these from pathology (for example gestational diabetes or significant renal disease). In some treatment contexts, altered renal clearance requires regimen changes (for example longer antibiotic courses or closer anticonvulsant-level monitoring).
Musculoskeletal adaptation is driven in part by relaxin and progesterone effects on pelvic joints and connective tissue, which support vaginal birth but can increase gait changes, round-ligament discomfort, lumbar lordosis-related back pain, and fall risk from center-of-gravity shift. In some pregnancies, diastasis recti develops and may influence core support in later pregnancy and postpartum recovery. Excess relaxin effect can rarely contribute to symptomatic symphysis-pubis separation with severe pelvic pain and impaired ambulation.
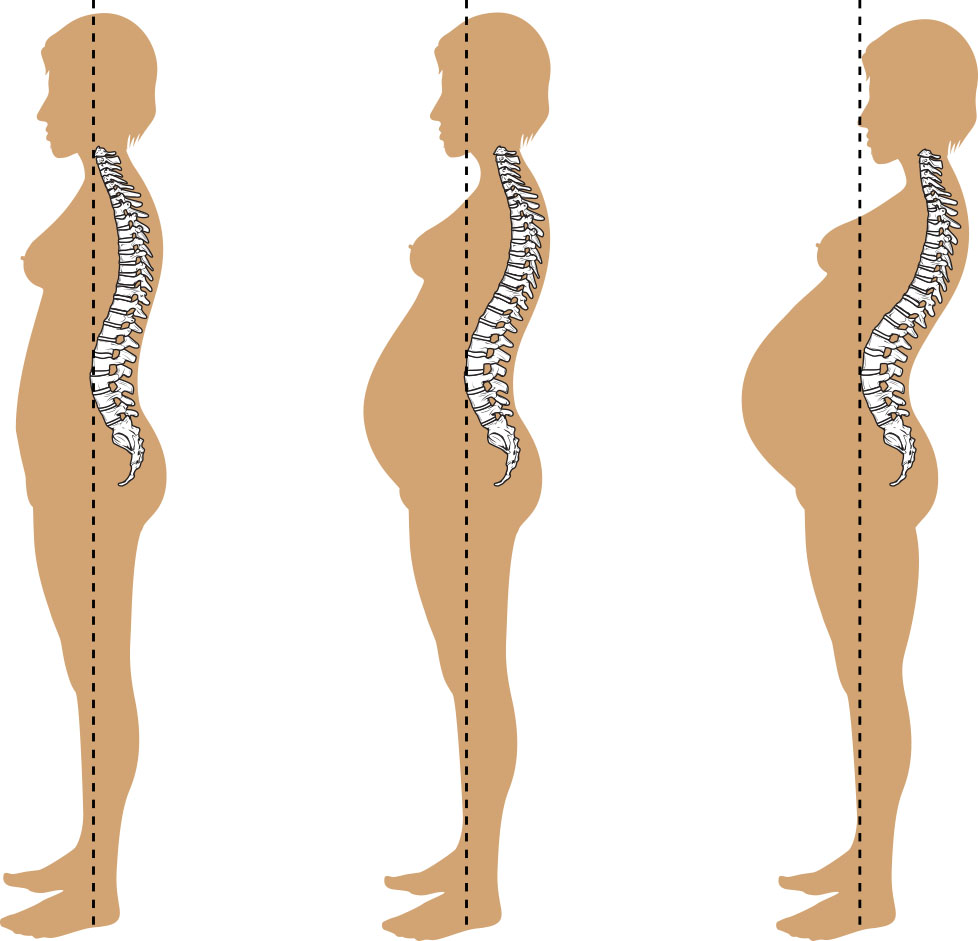 Illustration reference: OpenStax Maternal-Newborn Nursing Ch.10.1.
Illustration reference: OpenStax Maternal-Newborn Nursing Ch.10.1.
Immune adaptation supports fetal tolerance but also increases susceptibility to selected infections, and some autoimmune conditions may worsen in symptom burden during pregnancy.
Endocrine and metabolic adaptations are strongly influenced by placental hormones (hCG, progesterone, estrogen, hPL, relaxin), supporting implantation, uterine growth, fetal metabolism, and maternal tissue changes. Pituitary prolactin and oxytocin output increase across pregnancy in preparation for lactation and labor. Adrenal-endocrine balance also shifts: serum cortisol rises with delayed excretion and aldosterone supports fluid/electrolyte balance and BP regulation.
Pregnancy-specific thyroid-parathyroid adaptations are also clinically important. Thyroid output rises to support fetal neurodevelopment (including T3/T4 pathway demand), and parathyroid-hormone activity shifts across trimesters to maintain calcium-phosphate balance for fetal skeletal growth. Prolactin can increase markedly as lactotroph activity expands, while relaxin-mediated vasodilation and connective-tissue effects support uteroplacental adaptation and birth preparation.
As placental and maternal endocrine demand rises, insulin resistance increases and total insulin need often increases across gestation. Maternal basal metabolic rate also rises (often about 10-20%) with higher oxygen demand, and fluid-retention burden increases alongside plasma-volume expansion and fetal-placental fluid needs.
Early pregnancy physiology includes rapidly rising hCG produced by trophoblastic tissue; hCG can be detected in blood about 7 to 12 days after conception and in urine at about 2 weeks after conception, then rises quickly during early first trimester and commonly peaks around 10 weeks before declining. Placental hPL rises progressively and typically peaks in the late third trimester (around 34 weeks), amplifying fetal glucose availability and insulin-resistance burden.
Clinical obstetric dating is usually based on the first day of the last menstrual period (LMP), with an estimated due date around 40 weeks from LMP even though fetal age from conception is shorter. Pregnancy course is typically organized into first, second, and third trimesters for surveillance and counseling.
Classification
- Reproductive adaptations: Uterus, cervix, vagina, ovaries, and breast changes. Braxton-Hicks contractions can occur as mild, irregular uterine activity and usually do not change the cervix until late pregnancy.
- Trimester framework: First trimester (through 13 weeks 6 days), second trimester (14 weeks through 27 weeks 6 days), and third trimester (28 weeks through term).
- Cardiorespiratory adaptations: Volume expansion, vascular resistance changes, mild BP decrease with relative recovery later in pregnancy, heart displacement, and ventilation shifts.
- Renal/metabolic adaptations: Filtration increase, urinary stasis, insulin-resistance progression.
- Integumentary/musculoskeletal adaptations: Hyperpigmentation, striae, posture/ligament changes, and altered center of gravity.
- Neuro-immune-endocrine adaptations: Sleep/cognitive shifts, immune modulation to support fetal tolerance, and hormone-driven metabolic regulation. Examples include sleep disruption from nocturia/fetal movement, short-term concentration-memory decline, immune tolerance with retained pathogen defense, and thyroid/pituitary activity increases that support pregnancy and lactation.
Nursing Assessment
NCLEX Focus
Distinguish expected physiologic adaptations from signs of pregnancy complications requiring urgent escalation.
- Trend vital signs, edema pattern, cardiopulmonary symptoms, and activity tolerance.
- Assess for worsening snoring, witnessed apnea, or daytime somnolence in clients with pregnancy rhinitis because sleep-disordered breathing can coexist with hypertensive-risk patterns.
- Interpret hematologic trends with trimester context because physiologic hemodilution lowers expected hemoglobin/hematocrit ranges during pregnancy.
- Assess for supine hypotension patterns in late pregnancy and reinforce lateral positioning to improve venous return and placental perfusion.
- Assess urinary symptoms, hydration status, and UTI risk cues.
- Monitor common GI and integumentary changes while screening for severe or atypical presentations.
- Screen for common neuro-musculoskeletal symptoms (sleep disturbance, carpal-tunnel/sciatica exacerbation, muscle cramping) and functional impact.
- Evaluate glucose-risk factors and reinforce gestational diabetes screening windows.
- Document presumptive, probable, and positive pregnancy signs with differential awareness.
Nursing Interventions
- Provide anticipatory education on normal trimester-based body changes.
- Teach practical comfort measures for reflux, constipation, edema, dyspnea, and back pain.
- Reinforce nutrition, hydration, and micronutrient guidance aligned with pregnancy needs.
- Educate on warning signs (severe headache, visual changes, fever, bleeding, severe pain, reduced fetal movement when applicable).
- Coordinate timely follow-up testing for anemia, glucose tolerance, UTI, and other prenatal surveillance markers.
Normalization Error
Labeling all pregnancy discomfort as “normal” can delay recognition of serious maternal or fetal complications.
Pharmacology
| Drug Class | Examples | Key Nursing Considerations |
|---|---|---|
| prenatal-vitamins | Folate, iron, and calcium support contexts | Supports increased maternal-fetal nutritional demand and helps reduce deficiency-related risk. |
| antiemetics | Nausea/vomiting symptom contexts | Use pregnancy-safe options and monitor hydration and weight trends. |
Clinical Judgment Application
Clinical Scenario
A 32-week pregnant patient reports worsening dyspnea, marked lower-extremity edema, persistent headache, and visual changes.
- Recognize Cues: Symptoms exceed routine physiologic adaptation expectations.
- Analyze Cues: Findings may indicate hypertensive or cardiopulmonary complication risk.
- Prioritize Hypotheses: Immediate maternal-fetal safety evaluation is the priority.
- Generate Solutions: Escalate assessment, obtain urgent vitals/labs, and coordinate obstetric review.
- Take Action: Initiate rapid triage pathway rather than routine reassurance.
- Evaluate Outcomes: Complication risk is ruled out or treated early, preventing progression.
Related Concepts
- preconceptual-care - Baseline health optimization improves adaptation to pregnancy physiology.
- health-promotion-across-the-reproductive-lifespan - Pregnancy adaptation is part of lifespan reproductive care.
- urinary-tract-infections - Renal/urinary adaptations increase infection susceptibility.
- functional-reproductive-disorders - Some pre-existing disorders may worsen or shift during pregnancy.
- person-and-family-centered-care - Education should align with patient goals, literacy, and culture.
Self-Check
- Which cardiovascular changes are expected in normal pregnancy, and which are red flags?
- How do placental hormones drive maternal metabolic and systemic adaptations?
- What education best helps pregnant patients differentiate expected changes from danger signs?