Metabolic Alkalosis
Key Points
- Metabolic alkalosis is present when pH is greater than 7.45 due to net bicarbonate increase.
- Three major cause groups are gastrointestinal hydrogen loss, renal hydrogen loss, and bicarbonate retention or addition.
- Common examples include vomiting, gastric suction, diuretic-related secondary hyperaldosteronism, and antacid overuse.
- Hypokalemia can sustain metabolic alkalosis through intracellular hydrogen-ion shifts.
- Uncorrected alkalosis can cause arrhythmias, confusion, reduced cerebral blood flow, and death.
Pathophysiology
Metabolic alkalosis occurs when hydrogen ions are lost or bicarbonate accumulates beyond physiologic buffering needs. This shifts plasma toward alkalemia and alters neuromuscular and cardiovascular stability, particularly in hospitalized patients with ongoing losses or medication effects.
GI and renal mechanisms are dominant drivers. Gastric hydrochloric acid loss creates relative bicarbonate excess, while aldosterone-mediated renal hydrogen excretion and bicarbonate retention can maintain or worsen alkalosis. Fluid losses above the stomach (for example prolonged vomiting) tend to increase alkalosis risk, whereas fluid losses below the stomach (for example prolonged diarrhea) are more often associated with acidosis patterns.
Classification
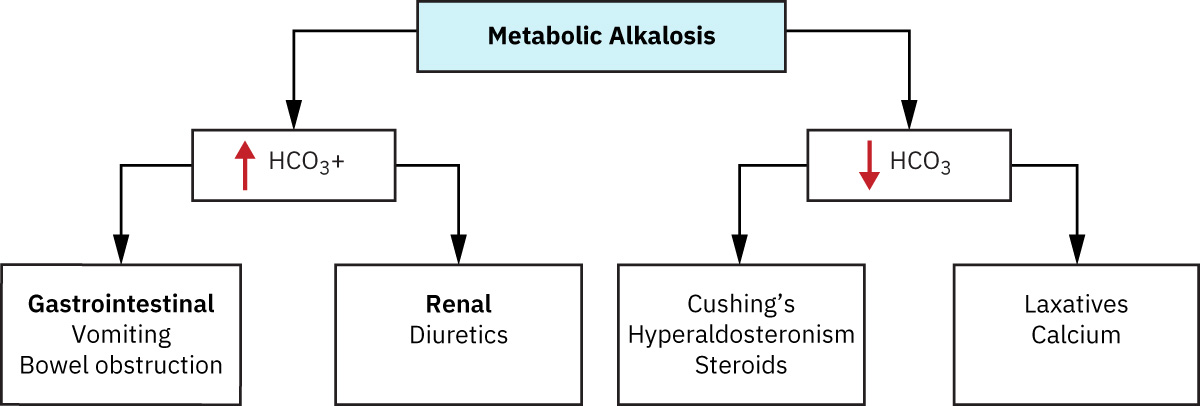 Illustration reference: OpenStax Fundamentals of Nursing Ch.20.2.
Illustration reference: OpenStax Fundamentals of Nursing Ch.20.2.
- GI hydrogen loss alkalosis: Vomiting or gastric suction removes acid-rich gastric contents.
- Renal hydrogen loss alkalosis: Diuretics and hyperaldosteronism-associated states increase urinary hydrogen loss.
- Bicarbonate retention/addition alkalosis: Antacid excess or compensation to chronic respiratory acidosis contributes.
Nursing Assessment
NCLEX Focus
Distinguish ongoing loss mechanism (GI vs renal vs bicarbonate gain) because correction requires stopping the driver, not only monitoring pH.
- Confirm alkalemia pattern (pH > 7.45) and trend bicarbonate with serial chemistry/ABG data.
- Assess for ongoing vomiting, gastric suction output, and dehydration indicators.
- Review medications for loop/thiazide diuretics and high alkali intake.
- Assess for concurrent hypokalemia because potassium depletion can maintain or worsen alkalosis.
- Monitor ECG rhythm and mental-status changes for worsening physiologic impact.
- Assess perfusion and neurologic status when alkalosis persists despite treatment.
Nursing Interventions
- Escalate persistent alkalemia early, especially with neurologic or cardiac symptoms.
- Reduce or correct active hydrogen-ion losses (for example, address excessive GI losses) per treatment plan.
- Support medication review and diuretic-related monitoring to reduce preventable progression.
- Correct contributing hypokalemia and discontinue exogenous bicarbonate inputs when present per treatment plan.
- Prepare dialysis escalation for clients with kidney disease when alkalosis cannot be corrected conservatively.
- Reassess blood gas and chemistry trends after each intervention cycle.
- Educate patients on safe antacid and diuretic use where relevant.
Hospital Complication Risk
Metabolic alkalosis is common in acute care and can progress to life-threatening complications if not corrected.
Pharmacology
This section frames medication contributors (notably diuretics and antacid exposure) as causes rather than providing detailed drug-treatment protocols.
Clinical Judgment Application
Clinical Scenario
A hospitalized patient with prolonged vomiting and ongoing gastric suction develops pH above 7.45 and rising serum bicarbonate.
- Recognize Cues: Persistent acid loss history with alkalemia trend.
- Analyze Cues: GI hydrogen loss is likely the primary alkalosis driver.
- Prioritize Hypotheses: Immediate priorities are stopping active losses and preventing cardiac/neurologic complications.
- Generate Solutions: Optimize fluid/electrolyte replacement, reassess suction indications, and monitor rhythm.
- Take Action: Escalate provider communication and implement correction plan promptly.
- Evaluate Outcomes: pH and bicarbonate trend toward normal with symptom stabilization.
Related Concepts
- acid-base-balance-principles - Baseline interpretation framework for alkalemia and bicarbonate trends.
- antiemetics - Common cause of gastric hydrogen loss causing alkalosis.
- nasogastric-tube-indications-and-safety - Procedure-related acid loss risk.
- fluid-electrolyte-regulation-by-organs - Hormonal driver of renal hydrogen excretion.
- diuretics - Common medication contributor to renal hydrogen loss alkalosis.
Self-Check
- Which clinical data support GI-loss metabolic alkalosis over renal-loss alkalosis?
- Why can chronic diuretic use worsen alkalosis through aldosterone pathways?
- What are the priority danger signs that require urgent escalation?