Fluid Electrolyte Regulation by Organs
Key Points
- Total body fluid is primarily regulated by the renal system.
- Kidneys circulate roughly 5 to 6 liters of blood repeatedly and generate about 200 liters of daily filtrate before selective reabsorption.
- When body fluid is low (hypovolemia), antidiuretic hormone (ADH) release increases renal water reabsorption.
- When renal tubular fluid volume is high, ADH is suppressed, contributing to more dilute urine.
- Serum osmolality is normally about 275-295 mmol/kg and reflects blood concentration status.
- Osmolality is solute particles per kilogram of solvent, while osmolarity is solute particles per liter of solvent.
- Urine specific gravity is normally about 1.010-1.020; higher values suggest concentration and lower values suggest dilution.
- Thirst, hypothalamic osmoreceptors, ADH, and RAAS coordinate intravascular volume and blood pressure.
- RAAS can be activated by hypotension or low serum sodium and increases vascular tone plus sodium-water retention while promoting potassium excretion.
- Aldosterone is a key regulator of sodium and potassium handling in renal physiology and supports water retention through sodium reabsorption.
- In stable adults, daily fluid output is mostly renal (about 60%) with the remainder as insensible skin/GI/lung losses (about 40%).
- Cardiac natriuretic peptides oppose RAAS during fluid overload by promoting vasodilation and sodium-water excretion.
- Homeostatic control uses receptor → control center → effector signaling loops to correct deviation from set points.
- Negative feedback is the primary homeostatic loop pattern and acts by reversing deviation from normal range.
- Positive feedback amplifies a stimulus for rapid completion of specific biologic events and must terminate promptly to avoid instability.
- Hyperosmolality and hypoosmolality can both cause severe neurologic deterioration when not corrected promptly.
- Failure at sensing, loop activation, response execution, or set-point regulation can progress to cellular injury and organ dysfunction.
- Electrolyte abnormalities are often interdependent, so one severe disturbance can trigger a multi-electrolyte chain reaction.
Pathophysiology
Fluid and electrolyte homeostasis depends on interaction between organ systems and hormones rather than isolated laboratory values. The kidneys regulate total body fluid and respond to tubular pressure and volume conditions through ADH modulation. Kidney filtration is pressure dependent: hydrostatic forces in glomerular capillaries support filtrate formation, and low renal perfusion pressure can reduce waste and fluid clearance.
When fluid is lost and intravascular sodium concentration rises, serum osmolality increases. Hypothalamic osmoreceptors trigger thirst and increase ADH release, which promotes renal water retention. This regulatory pathway depends on functional access to fluids and the ability to drink when thirst cues occur.
RAAS also supports perfusion during low-pressure states and low-sodium states. Reduced blood pressure or reduced serum sodium stimulates renal renin release. Renin converts liver-derived angiotensinogen to angiotensin I, and angiotensin-converting enzyme in the lungs converts angiotensin I to angiotensin II. Angiotensin II causes vasoconstriction and stimulates aldosterone release, increasing renal sodium reabsorption so water follows into the intravascular compartment while potassium excretion increases.
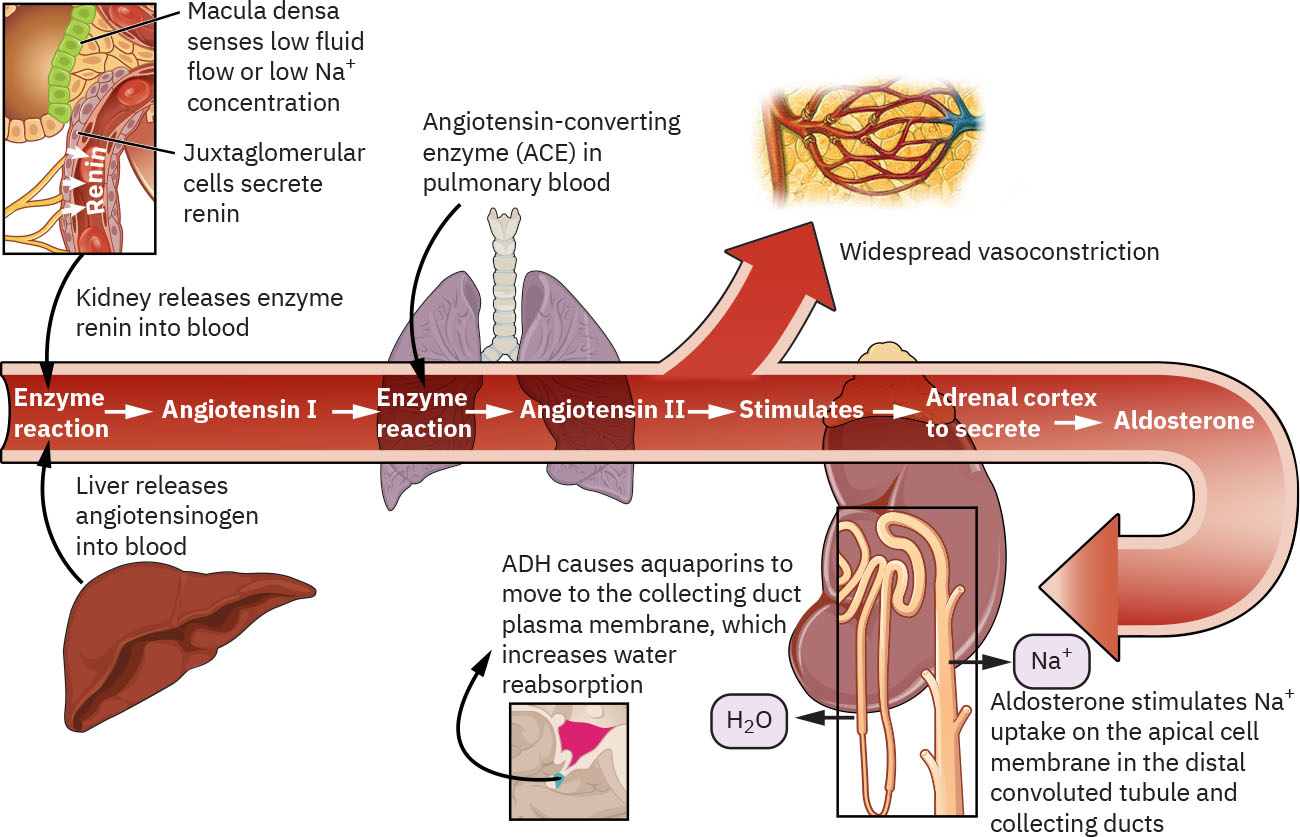 Illustration reference: OpenStax Clinical Nursing Skills Ch.19.1.
Illustration reference: OpenStax Clinical Nursing Skills Ch.19.1.
Natriuretic peptides act in physiologic opposition to RAAS. When cardiac tissue senses fluid overload and elevated pressure, these peptides promote vasodilation and renal sodium excretion to reduce circulating volume and pressure. Along with ADH, RAAS, and natriuretic peptides, endocrine factors such as insulin, epinephrine, glucocorticoids, and parathyroid-axis hormones influence specific electrolyte patterns. Renin release is regulated in part by the juxtaglomerular apparatus adjacent to the glomerulus, linking nephron perfusion sensing directly to RAAS activation.
With adequate intake, kidneys usually generate around 1,500 mL of urine daily. Oliguria is an early warning sign in fluid-deficit and kidney-injury pathways and should be trended with context.
Classification
- Volume-conserving response: Increased ADH in hypovolemia with increased water reabsorption.
- Volume-excreting response: ADH suppression when tubular fluid volume is high, leading to dilute urine.
- Osmoreceptor-thirst response: Increased serum osmolality drives thirst and ADH release.
- Osmolality measurement domain: Osmolality quantifies particles per kg solvent; osmolarity quantifies particles per L solvent.
- Hyperosmolality pattern: High-solute state (for example dehydration or diabetes-linked states) with risk for circulatory stress and neurologic symptoms.
- Hypoosmolality pattern: Low-solute state (for example excess free water or water-balance dysregulation) with risk for cerebral edema and neurologic decline.
- RAAS pressure-restoring response: Renin → angiotensin II → aldosterone increases vascular tone and sodium-water retention.
- RAAS sequence detail: Renin + angiotensinogen (liver) → angiotensin I → ACE (lungs) → angiotensin II.
- Natriuretic-peptide counterregulation: Cardiac peptides respond to fluid overload/high pressure by promoting vasodilation and sodium-water excretion.
- Feedback-loop control architecture: Receptor detection, central integration, and effector response coordinate correction of fluid/electrolyte deviation.
- Negative-feedback loop pattern: Inhibitory response reduces initial deviation and returns variables toward set point.
- Positive-feedback loop pattern: Amplifying response increases initial stimulus until endpoint completion (for example clot formation, lactation let-down, and uterine contraction in childbirth).
- Homeostatic-failure modes: Detection failure, feedback-initiation failure, effector-response failure, and set-point dysregulation.
- Sodium regulation pathway: Renal system with aldosterone influence.
- Potassium regulation pathway: Renal system with aldosterone plus insulin, epinephrine, and glucocorticoids.
- Calcium regulation pathway: Skeletal-endocrine interaction with parathyroid hormone, vitamin D, and calcitonin.
Nursing Assessment
NCLEX Focus
Match symptoms with the likely regulated electrolyte pathway first, then verify with labs and volume trend.
- Track intake-and-output, urine concentration pattern, and net fluid balance for ADH-related volume cues.
- Trend serum osmolality with urine osmolality/specific-gravity patterns when dilution versus concentration status is unclear.
- Assess whether functional barriers (cognitive change, weakness, poor access to fluids) limit response to thirst cues.
- Assess for low-perfusion compensatory patterns (for example tachycardia with hypotension) that can precede worsening renal filtration.
- For sodium imbalance, assess confusion, irritability, thirst, dry mucous membranes, headache, seizure risk, and coma progression.
- For potassium imbalance, assess GI cramping, weakness, pulse quality, and ECG changes including peaked or flattened/inverted T waves and U-wave patterns.
- For calcium imbalance, assess GI symptoms, muscle weakness, tingling, cramps, and tetany.
- Escalate low urine output trends, including persistent output below 30 mL/hour (or below 0.5 mL/kg/hour) over 8 hours.
- Correlate symptom patterns with serial chemistry values and clinical trajectory.
- When fluid retention worsens but urine output drops, consider mixed patterns where serum becomes more dilute while urine remains relatively concentrated.
Nursing Interventions
- Prioritize early intervention when volume or neurologic trends suggest failure of compensatory regulation.
- Coordinate serial labs and focused reassessment to determine if organ-level regulation is restoring balance.
- Implement ordered fluid strategies and monitor urine response for expected ADH-linked effects.
- Monitor daily intake/output patterns against expected adult water-balance ranges (approximately 2,500 mL intake and 2,500 mL output in stable conditions).
- Reinforce sodium and potassium management education in patients with chronic renal and endocrine risk factors.
- Escalate rapidly when ECG or neurologic findings indicate severe electrolyte instability.
Compensation Limits
Organ-hormone compensation is protective in acute states but cannot fully prevent deterioration in chronic disease or severe imbalance.
Pharmacology
| Drug Class | Examples | Key Nursing Considerations |
|---|---|---|
| diuretics | Loop and potassium-sparing categories | Diuretics alter sodium, potassium, and fluid balance and require close trend monitoring. |
| intravenous-fluid-categories-tonicity-and-infusion-regulation (electrolyte-replacement-therapy) | Sodium or potassium replacement protocols | Correction must be guided by serial labs, renal status, and symptom severity. |
Clinical Judgment Application
Clinical Scenario
A patient with chronic kidney disease develops fluid retention, electrolyte abnormalities, and evolving neurologic symptoms.
- Recognize Cues: Abnormal fluid balance, urine pattern change, and concerning sodium/potassium symptom profile.
- Analyze Cues: Renal-endocrine regulation is failing to maintain homeostasis.
- Prioritize Hypotheses: Immediate risks include neurologic decline and cardiac conduction instability.
- Generate Solutions: Intensify monitoring, obtain serial labs/ECG, and apply ordered volume-electrolyte correction.
- Take Action: Escalate deterioration and implement cause-directed management.
- Evaluate Outcomes: Urine, labs, and symptoms trend toward stable baseline.
Related Concepts
- sodium-balance-disorders - Sodium findings map to renal-aldosterone regulation failure or overload states.
- potassium-balance-disorders - Potassium instability reflects renal handling and hormone-modulated shifts.
- fluid-volume-deficit-hypovolemia-and-dehydration - Low volume state drives ADH-mediated water conservation.
- fluid-volume-overload-hypervolemia - High-volume states may accompany suppressed ADH and dilutional patterns.
- kidney-disease - Chronic renal dysfunction reduces capacity to maintain fluid-electrolyte homeostasis.
Self-Check
- How does ADH behavior differ between hypovolemia and high tubular fluid volume states?
- Which symptom clusters suggest sodium versus potassium versus calcium dysregulation?
- Why can compensation fail in patients with chronic renal disease even when hormonal pathways are activated?