Acid-Base Regulation Systems
Key Points
- Blood pH homeostasis depends on three coordinated systems: chemical buffer, respiratory, and renal regulation.
- Even small pH shifts can become life-threatening without timely recognition and correction.
- Dangerous deviations are associated with severe outcomes, and pH greater than 7.8 or less than 6.8 is often associated with death.
- The carbonic acid-sodium bicarbonate pair is the major physiologic buffer mechanism and accounts for most chemical buffering.
- Phosphate and protein pathways are key intracellular buffers; protein buffering contributes most intracellular capacity.
- Respiratory compensation occurs quickly, whereas renal bicarbonate compensation is slower.
Pathophysiology
The body maintains blood pH by coupling rapid chemical buffering with physiologic respiratory and renal responses. Buffer systems act first to blunt abrupt hydrogen ion change, while respiratory and renal pathways provide ongoing correction through CO2 and HCO3- control.
Respiratory regulation works through carbonic-acid equilibrium: increasing respiratory rate/depth lowers retained CO2 and carbonic-acid load, while hypoventilation raises retained CO2 and increases carbonic-acid burden. Renal regulation complements this by excreting or retaining bicarbonate to keep blood pH within the usual 7.35 to 7.45 physiologic range.
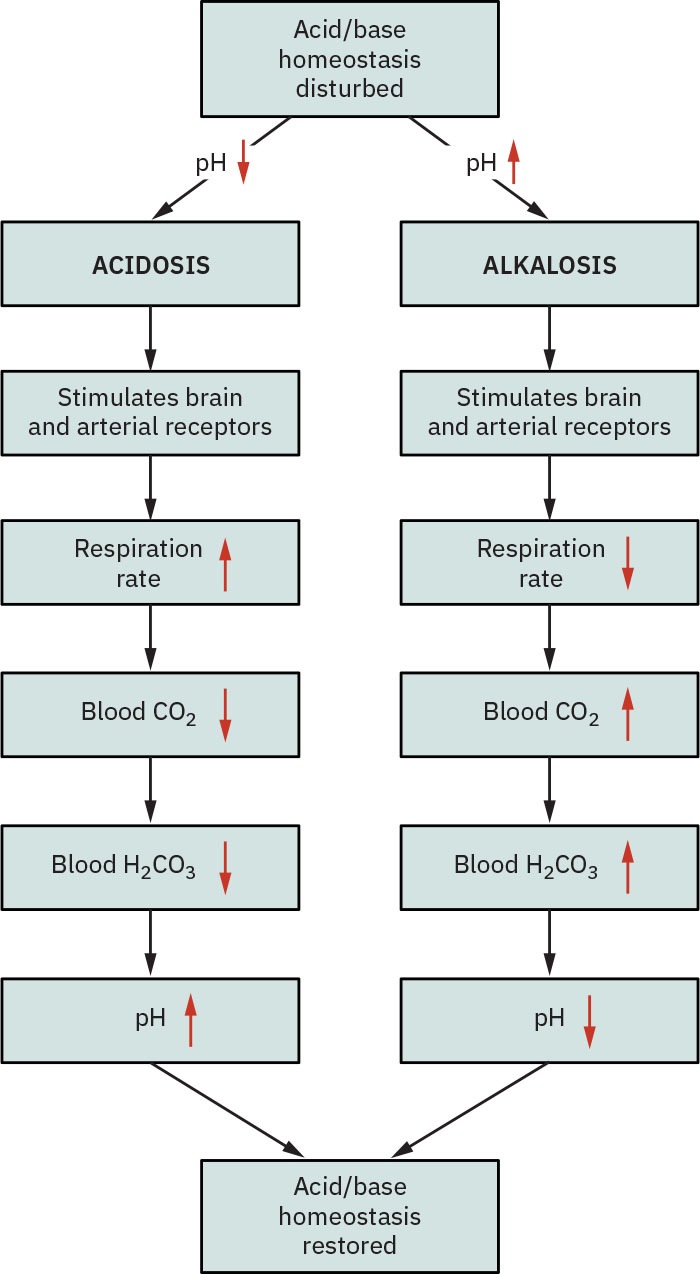 Illustration reference: OpenStax Fundamentals of Nursing Ch.20.2.
Illustration reference: OpenStax Fundamentals of Nursing Ch.20.2.
When one regulatory arm is impaired, compensatory stress shifts to remaining systems. Patients with chronic pulmonary or renal dysfunction therefore have reduced reserve and are more vulnerable to severe acid-base instability.
Classification
- Chemical buffer system: Immediate buffering of hydrogen-ion change, most prominently carbonic acid-bicarbonate.
- Respiratory regulatory system: Ventilation-mediated PaCO2 control.
- Renal regulatory system: Bicarbonate excretion/reabsorption to counter respiratory and metabolic shifts.
- Chemical-buffer pathways: Carbonic acid-bicarbonate (main extracellular pathway), phosphate (main intracellular pathway), and protein buffering (largest intracellular contribution, often reported around 75%).
Nursing Assessment
NCLEX Focus
Identify which regulatory arm is failing first, then determine whether compensation is present or exhausted.
- Trend ABG values (pH, PaCO2, HCO3-) to identify primary disturbance and compensation pattern.
- Distinguish uncompensated, partially compensated, and fully compensated states when serial ABGs are available.
- Assess ventilation effectiveness as a rapid regulator of PaCO2.
- Assess breathing depth and rate trends because shallow breathing raises retained CO2 and deep/rapid breathing lowers it.
- Assess renal status because bicarbonate control depends on kidney function.
- Assess whether respiratory or renal responses are compensating for a primary disturbance in the opposite system.
- Monitor for large pH deviations and associated neurologic/cardiac instability.
- Reassess after interventions to confirm movement toward physiologic range.
Nursing Interventions
- Escalate severe pH derangement urgently, especially when compensation appears inadequate.
- Support ventilation optimization for respiratory regulation deficits.
- Support renal-focused correction pathways for bicarbonate dysregulation.
- Coordinate serial ABG and chemistry monitoring to evaluate trajectory.
- Educate high-risk patients on early symptom reporting for decompensation.
Extreme pH Risk
pH outside survivable physiologic limits can progress rapidly to critical instability.
Pharmacology
This section emphasizes physiologic regulation systems; specific medication protocols are not detailed in this section.
Clinical Judgment Application
Clinical Scenario
A patient with chronic lung disease develops worsening dyspnea and progressive blood gas abnormalities, then shows delayed renal compensation.
- Recognize Cues: pH trend moves abnormal with PaCO2 changes and incomplete HCO3- correction.
- Analyze Cues: Primary respiratory derangement is stressing compensatory renal mechanisms.
- Prioritize Hypotheses: Risk of compensation failure and severe pH shift is the immediate concern.
- Generate Solutions: Intensify respiratory support, monitor serial ABGs, and reassess renal contribution.
- Take Action: Escalate multidisciplinary management and monitor for deterioration.
- Evaluate Outcomes: pH stabilizes as ventilation and compensatory pathways improve.
Related Concepts
- acid-base-balance-principles - Core interpretation framework for all three regulatory systems.
- bicarbonate-regulation - First-line chemical buffering and renal bicarbonate trajectory support.
- respiratory-acidosis - Ventilation-dependent CO2 control pathway can fail rapidly.
- metabolic-acidosis - Kidney-mediated bicarbonate control pathway can become inadequate in severe illness.
- arterial-blood-gas-abg - Primary tool for tracking regulation and compensation.
Self-Check
- Which acid-base regulatory system responds first to sudden hydrogen-ion change?
- Why are renal and respiratory dysfunction patients at higher risk for severe pH instability?
- What ABG trend suggests compensation is becoming inadequate?