Thorax and Lung Assessment Breath Sounds and Respiratory Patterns
Key Points
- Thorax assessment combines symptom interview, inspection, palpation, and auscultation.
- Chest symmetry, tracheal alignment, breathing effort, and respiratory pattern trends guide acuity decisions.
- Adventitious sounds and escalating work of breathing can signal rapid decompensation.
- Respiratory-rate and capnography changes can appear before oxygen-saturation decline during early deterioration.
Pathophysiology
Effective respiration depends on coordinated thoracic mechanics, airway patency, alveolar ventilation, and gas exchange. Structural or functional disruption alters airflow patterns and the breath sounds transmitted through the chest wall. The thorax extends from the base of the neck to the upper abdomen and is organized into the thoracic cage and thoracic cavity. The thoracic cage is formed by the sternum, costal cartilages, and 12 rib pairs anchored posteriorly to T1-T12; this framework protects the heart and lungs while enabling ventilatory motion. Rib groups are commonly described as true ribs (1-7), false ribs (8-12), and floating ribs (11-12). The sternum includes the manubrium, body, and xiphoid process, with clinically useful landmarks at the suprasternal notch, sternal angle, and second-rib attachment level for rib/intercostal counting.
Abnormal respiratory patterns often reflect systemic acid-base stress, neurologic dysregulation, fatigue, or worsening cardiopulmonary disease.
Classification
- Assessment domains: Subjective respiratory history, thoracic inspection and palpation, breath-sound auscultation, and respiratory-pattern analysis.
- Thoracic-structure domain: Thoracic cage (sternum, ribs, costal cartilages, thoracic vertebrae) and thoracic cavity organization containing heart and lungs.
- Landmark domain: Midclavicular, anterior axillary, and midaxillary reference lines for localization during exam and interventions.
- Breath-sound groups: Normal bronchial/bronchovesicular/vesicular sounds versus adventitious sounds.
- Adventitious categories: Fine/coarse crackles, rhonchi, wheeze, stridor, and pleural friction rub.
- Pattern abnormalities: Apnea, bradypnea, tachypnea, Cheyne-Stokes, Kussmaul, orthopnea, and agonal breathing.
- Special-voice maneuvers: Whisper pectoriloquy, egophony, and bronchophony can support consolidation/pleural-process suspicion when voice transmission is abnormally clear or altered.
- Normal breath-sound map: Bronchial (trachea/larynx, loud/high pitch, expiration longer), bronchovesicular (major bronchi near 1st-2nd interspaces, medium pitch with inspiratory-expiratory pause), vesicular (peripheral lung fields, soft/lower pitch).
- Adventitious profile: Fine crackles (high-pitched inspiratory popping), coarse crackles (lower-pitched inspiratory wet sounds), rhonchi (low-pitched continuous sounds from larger-airway narrowing/secretions), wheeze (high-pitched continuous sounds from smaller-airway narrowing), stridor (inspiratory upper-airway obstruction), pleural rub (grating pleural-inflammation sound).
Nursing Assessment
NCLEX Focus
Prioritize recognition of respiratory distress and airway threat before completing full lower-acuity documentation.
- Assess dyspnea timing, cough/sputum changes, tobacco or vaping exposure, and functional tolerance.
- Quantify smoking burden with pack-years (packs/day multiplied by years smoked) and include secondhand-smoke exposure context for pediatric and household risk analysis.
- During bedside reassessment, include current oxygen-delivery setup and active respiratory treatments when interpreting effort and breath-sound changes.
- Include current/past respiratory diagnoses, medication history, and family respiratory history in focused subjective assessment.
- Include nutrition-focused history when respiratory symptoms persist: 24-hour food/fluid recall, restrictive-diet pattern, and food-insecurity barriers that can worsen pulmonary-risk burden.
- Ask whether the patient uses home respiratory support or monitoring equipment (for example oxygen, CPAP/BiPAP, nebulizer, or peak-flow meter) and how it is being used.
- Integrate age, sex, cultural/environmental context, and health-practice factors when interpreting reported symptoms.
- During dyspnea interview, ask onset, trigger exposure (activity/dust/animals/food), exertion threshold, duration, relievers, and positional/nocturnal symptoms (orthopnea, pillow count, nighttime breathlessness).
- If shortness of breath is severe or is associated with chest pain, stop routine interview and escalate for emergency support.
- During cough interview, ask sputum amount/color, hemoptysis, associated fever/chills/night sweats, symptom duration, triggers, and treatment response.
- Quantify dyspnea with a 0-10 patient-reported scale and trend serial scores before and after interventions.
- Screen modifiable respiratory-risk practices during health history, including smoking/vaping, home or occupational pollution exposure, vaccine refusal, inactivity, high-weight pattern, and poor fruit-vegetable intake.
- Inspect chest configuration and symmetry, tracheal midline, accessory-muscle use, retractions, and nasal flaring.
- Treat new tracheal deviation as a high-risk cue for unilateral volume loss or pleural-space pathology and escalate promptly.
- Inspect for chest-wall contour abnormalities such as pectus excavatum or pectus carinatum, and correlate with thoracic expansion quality.
- During inspection, compare anteroposterior with transverse diameter; expected adult AP:transverse ratio is about 1:2.
- Count respiratory rate over a full minute and classify expected adult baseline as about 12-20 breaths/min with nonlabored regular rhythm.
- Use resting SpO2 context with respiratory rate; expected adult resting saturation is commonly about 94-100%, while values below baseline (especially in chronic COPD) require trend-based interpretation and follow-up.
- Assess depth and inspiratory-expiratory timing; quiet breathing is typically inspiration shorter than expiration, while active breathing may approach 1:1.
- Use surface landmarks (sternal angle/second rib and thoracic reference lines) to localize auscultation points and intercostal spaces consistently.
- Apply thoracic reference lines consistently for telemetry-lead placement and other chest procedures requiring precise anatomic localization.
- Check for tripod positioning and inability to speak full sentences as bedside respiratory-distress cues.
- Treat prolonged expiration and tripod positioning as high-risk bedside findings that can reflect obstructive air-trapping or significant respiratory effort.
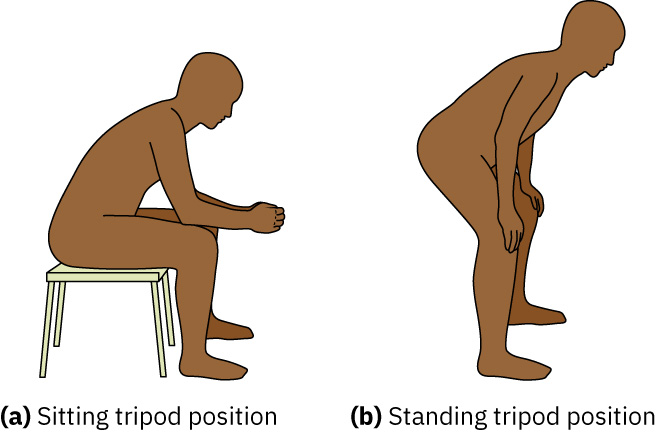 Illustration reference: OpenStax Clinical Nursing Skills Ch.23.2.
Illustration reference: OpenStax Clinical Nursing Skills Ch.23.2.
- Inspect for chronic hypoxia morphology such as fingertip clubbing and increased anterior-posterior chest diameter (barrel chest); AP:transverse near 1:1 supports hyperinflation patterns often seen with COPD.
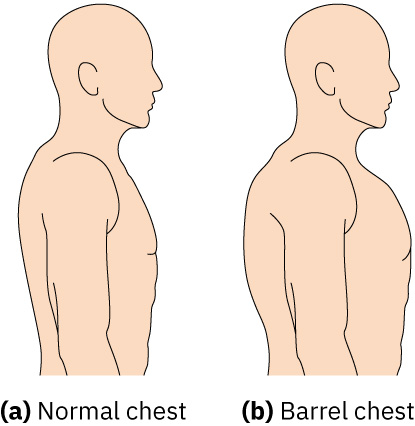 Illustration reference: OpenStax Clinical Nursing Skills Ch.23.2.
Illustration reference: OpenStax Clinical Nursing Skills Ch.23.2.
- Inspect spinal posture (for example kyphosis in older adults) because thoracic-shape changes can alter expansion patterns and breathing mechanics.
- Characterize sputum by amount, color, odor, viscosity, and blood presence; yellow/green purulent sputum supports infection-focused follow-up.
- Treat rusty/brown sputum as possible old blood and red/pink sputum (hemoptysis) as an escalation cue requiring prompt clinical correlation.
- Palpate for tenderness, masses, asymmetric expansion, and subcutaneous crepitus when indicated.
- If clavicular/chest-wall swelling is noted after recent airway instrumentation, palpate for subcutaneous emphysema (crepitus) and escalate new findings.
- After chest-tube placement or removal, palpate near insertion site for crepitus or air-leak clues and escalate new findings.
- Check symmetric chest expansion by placing hands at equal chest level and observing equal thumb movement during inhalation.
- Treat unequal unilateral expansion as an abnormal cue for possible pneumonia or thoracic trauma (including rib fracture or pneumothorax).
- Use percussion to differentiate expected lung-field resonance from abnormal dullness that can suggest consolidation or pleural-fluid accumulation.
- Auscultate anterior and posterior lung fields systematically and compare side-to-side.
- During auscultation, place the stethoscope directly on skin (not over clothing or hair) and avoid bony surfaces, clavicles/scapulae, and breast tissue when possible.
- Use an upright position for best sound transmission; if the patient cannot sit upright, turn side-to-side in bed and continue paired left-right comparisons.
- Use the diaphragm, coach deep mouth breathing, and listen through the full inspiratory-expiratory cycle at each site.
- If focal consolidation or pleural disease is suspected, add voice-transmission maneuvers (whisper pectoriloquy, egophony, bronchophony) and correlate with percussion and symptom findings.
- Escalate quickly when distress signs cluster: pursed-lip breathing, nasal flaring, audible breathing, intercostal retractions, anxiety, and accessory-muscle recruitment.
- Evaluate breathing-pattern abnormalities with clinical context: bradypnea (<12/min adult), tachypnea (>20/min adult), Cheyne-Stokes cyclical apnea-hyperventilation, Kussmaul deep rapid metabolic-acidosis pattern, orthopnea, dyspnea/PND, apnea, and agonal respirations.
- In older-adult trend interpretation, recognize Cheyne-Stokes patterns can occur without acute pathology, then correlate with oxygenation and neurologic findings before escalation.
- Review medication-related respiratory cues during history (for example ACE inhibitor-associated dry cough or NSAID-sensitive bronchospasm patterns).
- Include concurrent lung-assessment parameters: skin color (cyanosis/pallor), pleuritic pain, accessory-muscle use, and finger clubbing.
- In newborns and infants, count respirations for a full minute while the child is calm; periodic breathing pauses up to about 10 seconds can occur in the first weeks of life and are not abnormal unless distress signs are present.
- For infants and young children, prioritize early detection of nasal flaring/retractions and account for developmental mechanics (obligate nose breathing in early infancy; AP:transverse often near 1:1 until thoracic musculature matures around age six).
- In infants and young children, collect subjective respiratory-history details from caregivers when developmental stage limits direct symptom reporting.
- In infants and young children, recognize that cough may be relatively nonproductive and respiratory-muscle endurance is limited, so secretion retention and fatigue can progress quickly.
- In preterm or otherwise vulnerable neonates, maintain heightened surveillance for surfactant-related atelectatic patterns during respiratory illness.
- In pediatric respiratory illness, screen multisystem cues of deterioration (decreased oral intake, fewer wet diapers/urine output, sunken fontanel in infants, tachycardia, altered mentation, fatigue, cyanosis, and dry mucous membranes or poor skin turgor).
- For older adults with shallow breathing or respiratory-muscle fatigue, split deep-breath auscultation into shorter segments with rest periods (for example anterior lung fields first, then posterior) to reduce fatigue-related assessment error.
- Use finding clusters to narrow likely complications: unilateral diminished sounds/expansion lag (atelectasis or pneumothorax), cough plus wheeze/URI history (bronchitis), crackles with pink frothy sputum/peripheral edema (pulmonary edema), and diminished sounds with pleuritic pain/fever (pleural effusion).
Nursing Interventions
- Escalate increased work of breathing, severe pattern abnormalities, and new high-risk adventitious sounds promptly.
- Position to optimize ventilation, support oxygen therapy per orders, and reduce exertional demand.
- Apply the smoking-cessation Five As for patients ready to quit: Ask, Advise, Assess readiness, Assist with counseling/pharmacotherapy, Arrange early follow-up.
- Reassess after interventions and report objective trend changes in effort, sounds, and oxygenation.
- Monitor respiratory rate, rhythm, depth, and effort as ongoing trend data rather than isolated values.
- Track chest movement symmetry, accessory-muscle use, and intercostal or supraclavicular retractions.
- Auscultate for decreased or absent ventilation and adventitious sounds, then map changes by location and severity.
- Use oxygen-saturation monitoring with appropriate alarms in clients at risk for hypoxia per policy.
- Use ABG data when indicated for severe compromise, recognizing the test is invasive and interpreted with bedside trends.
- In sedated clients, prioritize continuous pulse-oximetry trend monitoring and prompt alarm response.
- Continue close trend monitoring even after short-term oxygen-saturation improvement because acute respiratory distress can recur rapidly.
- In adults without chronic low baseline, escalate new SpO2 below 92%; treat SpO2 below 88% as severe hypoxia requiring urgent intervention.
- If rapid shallow breathing, crackles, and reduced air movement appear together, escalate promptly for urgent imaging and treatment of possible severe pulmonary infection.
- Treat new chest pain with respiratory decline, or worsening dyspnea with irritability/restlessness/LOC change, as immediate escalation findings.
- Evaluate suctioning need by correlating coarse airway sounds (rhonchi) with secretion burden and cough effectiveness.
- Monitor for hypoxia progression cues such as restlessness, anxiety, and air hunger.
- Include new confusion or LOC change in early hypoxia cue surveillance.
- Document onset, duration, and characteristics of cough, plus secretion amount and quality.
- Track dyspnea triggers/relievers and include chest-imaging findings in longitudinal respiratory surveillance.
- Include chest X-ray findings in trend review and verify pregnancy status before imaging escalation per policy.
- When escalation workup is unclear, anticipate expanded respiratory diagnostics such as CT/MRI, V/Q scan, or pulmonary-function testing per clinical context.
- If chest pain is present with pressure quality, jaw/arm radiation, dyspnea, dizziness, or nausea, escalate as an emergency before routine focused pain workup.
- Modify planned interventions to current dyspnea severity; if distress worsens, pause exertional tasks (for example ambulation) and escalate for RT consult or medication adjustment as ordered.
- Document intervention effectiveness using reassessment trends in heart rate, respiratory rate, pulse oximetry, lung sounds, and patient-reported dyspnea score.
- Document baseline and reassessment in a complete lung-assessment format: respiratory symptoms/history, thorax symmetry and AP:transverse ratio, respiratory rate/effort/pattern, retractions or accessory-muscle use, palpation findings, bilateral auscultation map, and room-air or oxygen-supported SpO2.
Respiratory Failure Risk
Retractions, stridor, agonal pattern, or rapidly worsening dyspnea are emergency findings requiring immediate escalation.
Pharmacology
| Drug Class | Examples | Key Nursing Considerations |
|---|---|---|
| bronchodilators | Short-acting and long-acting agents | Evaluate post-treatment breath-sound and work-of-breathing response. |
| antibiotics | Bacterial respiratory infection contexts | Correlate regimen effectiveness with fever, sputum, and auscultation trends. |
Clinical Judgment Application
Clinical Scenario
A patient with COPD develops tachypnea, intercostal retractions, and new diffuse wheezing.
- Recognize Cues: Increased respiratory effort with new adventitious findings.
- Analyze Cues: Airflow limitation is worsening and may progress to respiratory failure.
- Prioritize Hypotheses: Immediate priority is oxygenation and ventilation support.
- Generate Solutions: Initiate escalation, optimize positioning, and prepare ordered respiratory interventions.
- Take Action: Communicate objective findings and monitor trend response closely.
- Evaluate Outcomes: Work of breathing and breath sounds improve after treatment.
Related Concepts
- respiratory-system - Core anatomy and gas-exchange framework for focused respiratory assessment.
- pulmonary-function-testing-and-pefr-zones - Supports trend interpretation of obstructive respiratory changes.
- oxygen-therapy-device-selection-and-monitoring - Device selection and reassessment principles during respiratory decline.
- documenting-and-reporting-data - Objective pattern documentation improves escalation quality.
Self-Check
- Which assessment findings indicate immediate respiratory escalation?
- How do adventitious sounds help narrow likely pathophysiology?
- Why must respiratory-pattern changes be trended instead of treated as isolated findings?