Adrenal Disorders
Key Points
- Primary adrenal insufficiency (Addison’s disease) causes deficient cortisol and aldosterone, with major fluid-electrolyte and perfusion consequences.
- Addisonian crisis is a life-threatening emergency often triggered by physiologic stress and requires rapid hemodynamic and hormone replacement support.
- Typical Addisonian electrolyte pattern is hyponatremia with hyperkalemia, often with hypotension and arrhythmia risk.
- Cushing’s disease/syndrome reflects excess adrenal-glucocorticoid effect and commonly presents with hypertension, central weight gain, skin fragility, and infection susceptibility.
- Core management differs by direction of dysfunction: hormone replacement for adrenal insufficiency versus cortisol-reduction or source-control therapy for hypercortisol states.
- The most common Cushing syndrome cause is prolonged high-dose glucocorticoid exposure; endogenous tumor-driven cortisol excess also occurs.
- Adrenal crisis can progress to shock rapidly and carries substantial mortality risk without immediate treatment.
- In published acute-care references, adrenal-crisis mortality remains high, reinforcing immediate escalation needs.
- Nutrition counseling is disorder-specific: sodium support in Addison pathways and lower-refined-carbohydrate plans in Cushing pathways can improve metabolic stability.
Pathophysiology
Adrenal physiology integrates cortex and medulla outputs. The cortex produces glucocorticoids (for example cortisol), mineralocorticoids (for example aldosterone), and androgens; the medulla produces catecholamines that mediate acute fight-or-flight responses. In stress adaptation, early alarm-phase response is catecholamine dominant, while prolonged stress burden drives hypothalamic-pituitary-adrenal signaling and cortisol-mediated resistance/exhaustion effects.
Addison’s disease is adrenal hypofunction with inadequate cortisol and aldosterone production. Reported etiologies include autoimmune adrenal destruction, infectious processes (including tuberculosis, HIV, syphilis, and histoplasmosis), bilateral adrenalectomy, inherited disorders, adrenal trauma, metastatic malignancy, selected cortisol-suppressing medications (for example ketoconazole, etomidate, and phenytoin), and abrupt cessation after prolonged high-dose glucocorticoid exposure.
Low aldosterone reduces sodium and water retention and contributes to potassium retention, leading to hypovolemia, hypotension, hyponatremia, and hyperkalemia risk. Low cortisol also impairs stress adaptation and metabolic stability.
Cushing’s patterns represent adrenal hormone excess. The most common Cushing syndrome pattern is exogenous from prolonged high-dose glucocorticoid or corticosteroid therapy. Endogenous hypercortisol states can also occur when adrenal tumors or ectopic ACTH-producing tumors drive excess cortisol production. Excess glucocorticoid and mineralocorticoid effect contributes to fluid retention, blood-pressure elevation, glucose dysregulation, skin and muscle catabolism, and infection risk. Cushing physiology also includes increased gluconeogenesis and insulin-resistance burden with impaired protein synthesis and reduced tissue-repair capacity, which worsens wound healing, bone integrity, and immune resilience.
Classification
- Chronic adrenal insufficiency (Addison’s disease): Persistent adrenal hormone deficiency with multisystem manifestations.
- Acute adrenal crisis (Addisonian crisis): Emergency decompensation when cortisol/aldosterone demand exceeds available supply during stress (for example infection, surgery, or trauma).
- Cushing’s disease: Endogenous adrenal hormone excess associated with adrenal tumor-driven hypersecretion.
- Pituitary-driven Cushing’s disease subtype: ACTH-secreting pituitary adenoma drives most endogenous Cushing pathways in major reports.
- Cushing’s syndrome: Hypercortisol state caused by glucocorticoid medication exposure for another condition.
Nursing Assessment
NCLEX Focus
Prioritize hemodynamic instability and electrolyte-danger cues in suspected adrenal crisis.
- Assess for fatigue, weakness, weight loss, salt craving, hypotension, dizziness, and syncope.
- Screen for GI findings including abdominal pain, nausea, vomiting, diarrhea, and anorexia.
- Assess endocrine and reproductive cues such as hypoglycemia, amenorrhea or oligomenorrhea, decreased libido, and hair-loss changes.
- Assess for progressive hyperpigmentation and, in affected female patients, reduced axillary or pubic hair as chronic adrenal-insufficiency clues.
- Assess dermatologic and neuropsychiatric findings including hyperpigmentation, easy bruising, mood change, lethargy, irritability, and concentration difficulties.
- In adrenal-insufficiency patterns, inspect common hyperpigmentation zones (for example palmar creases, lips, knuckles, posterior neck, areola, and gingival mucosa) and assess for vitiligo.
- Distinguish slow chronic progression from rapid deterioration suggestive of adrenal crisis.
- In crisis concern, prioritize hypotension, dehydration, sodium decline, potassium elevation, and arrhythmia symptoms.
- In crisis concern, also prioritize hypoglycemia progression and altered mental-status burden.
- For Cushing’s patterns, assess for hypertension, central weight gain, posterior cervical fat pad, round facial appearance, muscle weakness/wasting, and skin thinning with bruising or striae.
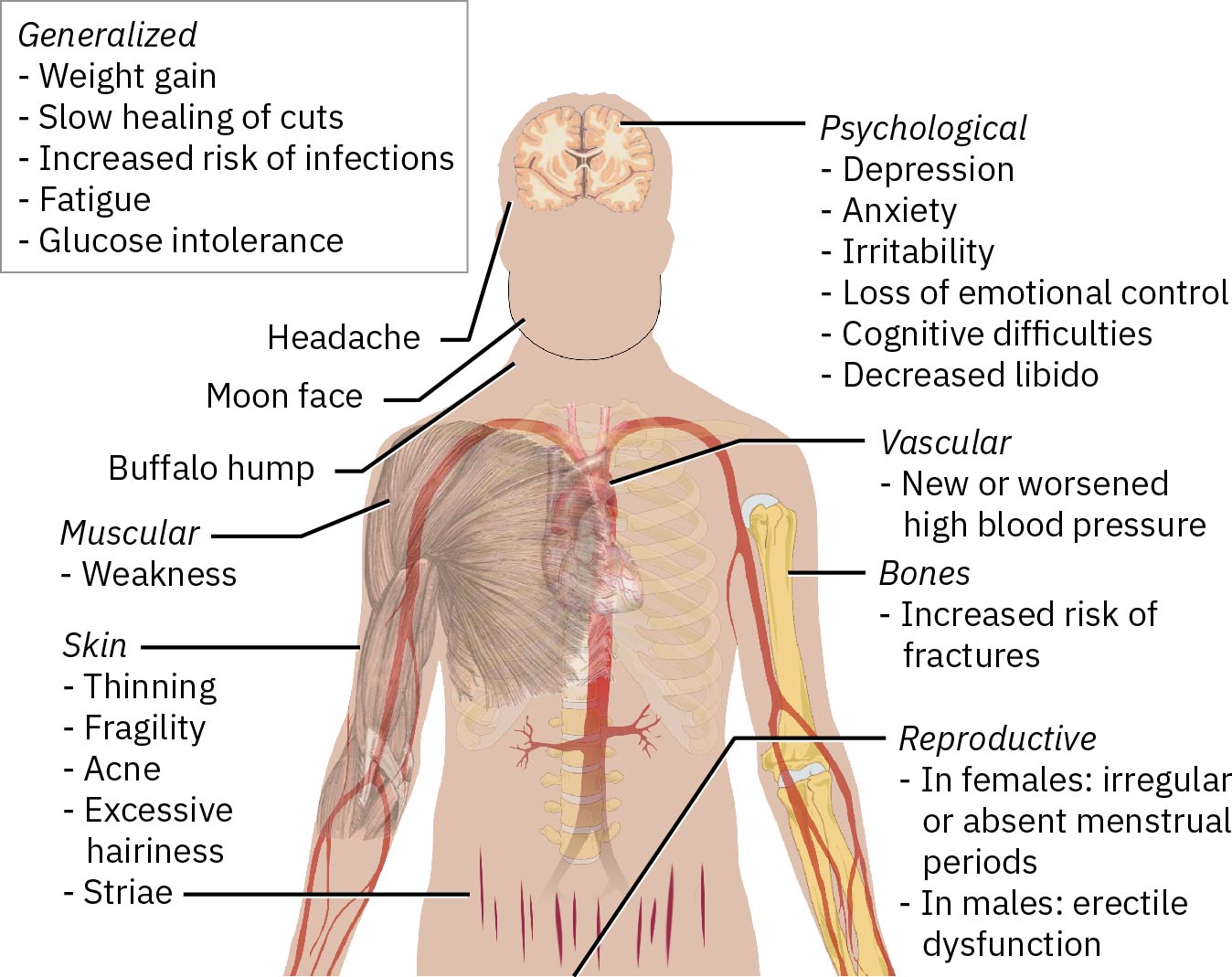 Illustration reference: OpenStax Clinical Nursing Skills Ch.22.1.
Illustration reference: OpenStax Clinical Nursing Skills Ch.22.1.
- Screen for mood or cognitive changes, infection susceptibility, delayed wound healing, edema, peptic-ulcer symptoms, and reproductive changes (menstrual irregularity, hirsutism, reduced libido, impotence).
Nursing Interventions
- Escalate suspected Addisonian crisis immediately and support emergency care workflow.
- Support rapid IV isotonic saline administration for hypovolemia correction and blood-pressure stabilization.
- In crisis pathways, support rapid dextrose plus hydrocortisone administration with continuous reassessment of perfusion and mentation.
- Administer prescribed corticosteroid replacement (for example hydrocortisone) and monitor response.
- Support urgent hyperkalemia management and continuous cardiac monitoring when potassium is elevated.
- Trend glucose and electrolyte trajectories closely; consider telemetry monitoring when dysrhythmia or severe volume-electrolyte instability is present.
- Reinforce medication adherence and stress-dose education for illness, injury, and other high-stress periods.
- Teach emergency-alert identification use (medical alert bracelet or necklace) and when to seek urgent care.
- Teach self-administration of emergency hydrocortisone during high-stress illness when prescribed.
- For Cushing’s states, support source-control treatment planning (tumor-directed surgery or radiation when indicated).
- Support medication strategy for cortisol reduction or glucocorticoid tapering when ordered, and monitor blood pressure and electrolyte response.
- In Addison pathways, reinforce sodium-adequate meal planning and salt-containing options when ordered to reduce hyponatremia risk.
- Reinforce sodium-aware nutrition guidance, weight-management education, activity planning, infection-prevention habits, and psychological support for chronic body-image and coping burden.
- In Cushing pathways, reinforce lower-refined-carbohydrate plans (often around 25-50 g/day in specialist-directed plans) and avoidance of sugary, highly processed, and refined-fat food patterns.
- In Cushing care, implement pressure-injury prevention and fall precautions proactively because weakness, poor healing, and fatigue increase safety risk.
- If appetite is poor in adrenal-insufficiency pathways, support frequent small meals and collaborative nutrition planning.
- Monitor for adrenal-crisis transition when glucocorticoid therapy is reduced or stopped; abrupt withdrawal can trigger severe decompensation.
- Coordinate diagnostic preparation and follow-up for endocrine imaging and hormone-testing workflows used to localize excess-cortisol sources.
Adrenal Crisis
Hyponatremia, hyperkalemia, and hypotension in adrenal insufficiency can progress quickly to life-threatening instability and require immediate escalation.
Laboratory and Diagnostic Testing
- Trend serum cortisol and aldosterone as core hormone markers in suspected adrenal insufficiency.
- In suspected primary adrenal insufficiency, assess morning cortisol (for example values below about 3 mcg/dL), ACTH-stimulation response, and renin-activity trends.
- Trend serum-sodium and serum-potassium because low aldosterone is associated with hyponatremia and hyperkalemia.
- Correlate electrolyte abnormalities with perfusion status, rhythm findings, and symptom progression.
- Include glucose and calcium trends when adrenal-insufficiency severity is being characterized.
- Review TSH pattern in context because secondary elevation can appear with low-cortisol states.
- Use targeted imaging and selected biopsy pathways to evaluate infection, adrenal mass, calcification, or malignancy when indicated.
- In hypercortisol states, monitor for hypernatremia and hypokalemia patterns associated with excess aldosterone effect.
- In Cushing evaluation, include dexamethasone-suppression testing and 24-hour urine free-cortisol assessment when ordered.
- In Cushing-lab patterns, review glucose elevation and low-potassium/low-calcium trends alongside clinical findings.
- In Cushing patterns, correlate cortisol with ACTH trends and review MRI/CT findings for adrenal, pituitary, or ectopic tumor sources; pituitary-driven Cushing disease is a major endogenous subtype.
Pharmacology
| Drug Class | Examples | Key Nursing Considerations |
|---|---|---|
| glucocorticoid replacement | hydrocortisone, prednisone, dexamethasone | Replace deficient cortisol in adrenal insufficiency; illness/stress can require dose escalation. |
| mineralocorticoid replacement | fludrocortisone | Supports sodium-water balance and potassium regulation when aldosterone is deficient. |
| cortisol-suppression therapy | aminoglutethimide, ketoconazole, mifepristone | Used in hypercortisol states; monitor response with symptom, glucose, blood-pressure, and lab trends. |
| mineralocorticoid antagonism | spironolactone | Supports blood-pressure and electrolyte control in excess-aldosterone presentations. |
| rate/pressure control | beta-blockers | Adjunct for tachycardia or hypertension in hyperfunction presentations as ordered. |
| hyperkalemia-directed therapy | protocol-based potassium-lowering medications | Used during Addisonian crisis when potassium elevation increases arrhythmia risk. |
| adjunct complication control | potassium supplements, diuretics, antihypertensives, bisphosphonates | Used in selected Cushing pathways to manage electrolyte, volume-pressure, and bone-loss complications. |
Clinical Judgment Application
Clinical Scenario
A patient with known Addison’s disease develops vomiting, weakness, hypotension, and worsening dizziness during an acute infection.
- Recognize Cues: Infection trigger plus adrenal-insufficiency history and perfusion decline.
- Analyze Cues: High concern for Addisonian crisis with possible sodium-potassium derangement.
- Prioritize Hypotheses: Immediate risk is hemodynamic collapse and dysrhythmia from electrolyte shift.
- Generate Solutions: Urgent escalation, IV saline support, corticosteroid replacement, and cardiac-electrolyte monitoring.
- Take Action: Implement emergency protocol and reassess blood pressure, symptoms, and lab trends.
- Evaluate Outcomes: Perfusion improves and electrolyte abnormalities move toward safer range.
Related Concepts
- endocrine-system - Adrenal cortex and medulla physiology context.
- corticosteroids - Glucocorticoid therapy and adrenal-axis suppression considerations.
- serum-sodium - Hyponatremia trend interpretation in aldosterone deficiency.
- serum-potassium - Hyperkalemia risk and arrhythmia surveillance.
- potassium-balance-disorders - Hyperkalemia management pathways used during crisis care.
- thyroid-medications - Thyroid-hormone replacement requires adrenal-insufficiency screening to avoid crisis precipitation.
Self-Check
- Why does aldosterone deficiency in Addison’s disease cause both hyponatremia and hyperkalemia?
- Which assessment cues should trigger immediate suspicion for Addisonian crisis?
- Why are stress-dose glucocorticoids emphasized during acute illness in adrenal insufficiency?